07 Nov New Drug Class Holds Promise Against Antibiotic Resistant Gonorrhea
MedicalResearch.com Interview with:

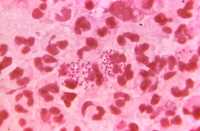
Intracellular Gram-negative, Neisseria gonorrhoeae diplococcal bacteria, – CDC image
Edward W. Hook, III, MD
University of Alabama at Birmingham
Medicine / Infectious Diseases
Birmingham, AL
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Zoliflodacin represents a new class of antibiotics (spiropyrimidinetriones) with in vitro activity against Neisseria gonorrhoeae, as well as other STD pathogens (Chlamydia trachomatis and Mycoplasma genitalium). Because of this promising data and the fact that the manufacturer (Entasis Pharmaceuticals) was willing to pursue the possibility of using this drug to treat gonorrhea, a Phase II trial was conducted which showed he drug to be 96% effective for genital or rectal infections. The drug was well tolerated as well making it a promising drug for gonorrhea treatment which might help to combat the increasing problem of antibiotic resistant gonorrhea.
MedicalResearch.com: What should readers take away from your report?
Response: Zoliflodacin holds promise as a possible new drug for gonorrhea treatment and efforts to combat the problem of progressive antibiotic resistant gonorrhea.
MedicalResearch.com: What recommendations do you have for future research as a result of this work?
Response: Based on the study outcome, planning is underway for a larger, multi-center Phase III study with the goal of obtaining approval from the U.S. FDA, as well as in South Africa, Thailand and the European Union.
Citation:
[wysija_form id=”3″]
[last-modified]
The information on MedicalResearch.com is provided for educational purposes only, and is in no way intended to diagnose, cure, or treat any medical or other condition. Always seek the advice of your physician or other qualified health and ask your doctor any questions you may have regarding a medical condition. In addition to all other limitations and disclaimers in this agreement, service provider and its third party providers disclaim any liability or loss in connection with the content provided on this website.
Last Updated on November 7, 2018 by Marie Benz MD FAAD
