MedicalResearch.com Interview with:
[caption id="attachment_53780" align="alignleft" width="164"]

Dr. Gefen[/caption]
Amit Gefen PhD
Professor of Biomedical Engineering
The Herbert J. Berman Chair in Vascular Bioengineering
Department of Biomedical Engineering, Faculty of Engineering
Tel Aviv University, Tel Aviv, Israel
MedicalResearch.com: What is the background for this study?
Response: Although we are witnessing continuous progress in medical technologies, the design of many of the most commonly used medical devices e.g. oxygen masks or cervical collars has changed very little over a period of decades. Not surprisingly, these devices are also the ones which are frequently associated with device-related pressure ulcers (DRPUs). These DRPUs are frequently a hospital-acquired injury which involves risk of infections (including e.g. sepsis and antibiotic-resistant bacteria), scarring with serious psychological consequences, additional and significant healthcare costs and a basis for liability suits and litigation. The problem is massive in Europe and the US and is most frequently encountered in clinical environments where devices are used intensively, such as in operation theatres, intensive care units and emergency care settings (in both adult and pediatric medicine), but also, in elderly care facilities where patients often have fragile skin. With the current pandemic spread of the coronavirus, facilities worldwide are experiencing a considerable rise in usage of emergency and intensive care equipment, which will very likely considerably escalate the incidence of DRPUs.
Early in 2019, a committee of global experts which I have chaired, has met for two days of intensive deliberation in London UK, to start developing the first-ever international consensus document on device-related pressure ulcers . After a rigorous review process by an international review committee of other experts, this consensus report has been published as a Special Edition of the
Journal of Wound Care in February 2019 (
https://doi.org/10.12968/jowc.2020.29.Sup2a.S1), under the name "
Device-related pressure ulcers: SECURE prevention". The publisher has kindly made this publication freely downloadable and thereby accessible and available to anyone, including all professionals who may need guidance in this regard, including clinicians, industry, regulators and academic researches.


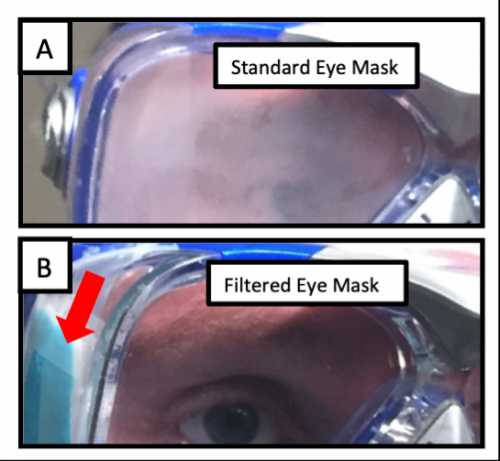

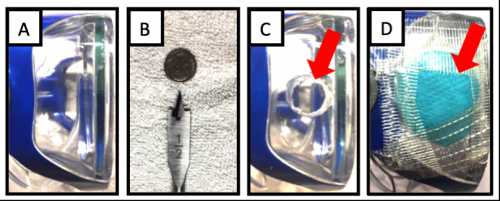 The building of the filtered eye mask prototype.[/caption]
The building of the filtered eye mask prototype.[/caption]
