Author Interviews, Ophthalmology, Technology / 16.01.2025
SING IMT for Late-Stage, Age-Related Macular Degeneration from Samsara Vision
MedicalResearch.com Interview with:
Prof Alfonso Savastano
Ospedale Generale Regionale
“F. Miulli” - Acquaviva delle Fonti (BA) and
Libera Università Mediterranea Degennaro (L.U.M.)- Casamassima (BA)
MedicalResearch.com: What is the background for this study?
Response: The SING IMT® (Smaller-Incision New-Generation Implantable Miniature Telescope) is a novel, intraocular device for people blinded in the central vision by late-stage, age-related macular degeneration (AMD), the leading cause of unpreventable blindness. It is approved for use in CE referenced countries and under investigation in the United States.
[caption id="attachment_65965" align="aligncenter" width="500"]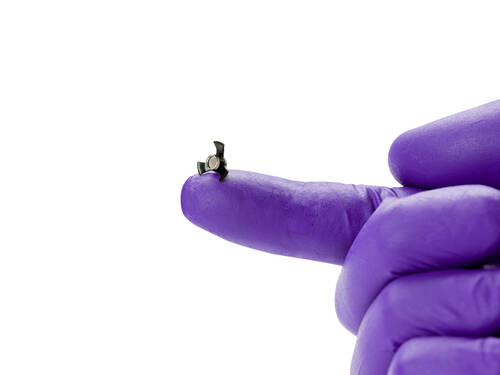 SING IMT for Late-Stage, Age-Related Macular Degeneration from Samsara Vision[/caption]
Our study evaluated the intermediate-term visual and safety outcomes of the SING IMT in patients (n=35) 55 years and older at 6 months post-surgery and found that SING IMT implantation improved distance and near vision, with an expected impact on the corneal endothelium cell density and manageable safety outcomes. Key findings include:
SING IMT for Late-Stage, Age-Related Macular Degeneration from Samsara Vision[/caption]
Our study evaluated the intermediate-term visual and safety outcomes of the SING IMT in patients (n=35) 55 years and older at 6 months post-surgery and found that SING IMT implantation improved distance and near vision, with an expected impact on the corneal endothelium cell density and manageable safety outcomes. Key findings include:
 SING IMT for Late-Stage, Age-Related Macular Degeneration from Samsara Vision[/caption]
Our study evaluated the intermediate-term visual and safety outcomes of the SING IMT in patients (n=35) 55 years and older at 6 months post-surgery and found that SING IMT implantation improved distance and near vision, with an expected impact on the corneal endothelium cell density and manageable safety outcomes. Key findings include:
SING IMT for Late-Stage, Age-Related Macular Degeneration from Samsara Vision[/caption]
Our study evaluated the intermediate-term visual and safety outcomes of the SING IMT in patients (n=35) 55 years and older at 6 months post-surgery and found that SING IMT implantation improved distance and near vision, with an expected impact on the corneal endothelium cell density and manageable safety outcomes. Key findings include:
- At six months post-surgery, at least 1-, 2-, and 3-line gains in best-corrected distance (BCDVA) were achieved in 97.1 percent, 68.6 percent and 51.4 percent of operated eyes, respectively
- The percentage of patients able to read at near distance increased from 28.6 percent at baseline to 97.1 percent at six months.
- The study also found that corrected near visual acuity was also significantly improved by ⁓3 lines at 6 months post-surgery.
- The mean (SD) change from baseline in corneal endothelial cell density (ECD) at six months in operated eyes was -280.7 (315.9) cells/mm2 (-11.4 %). This is a result similar to that seen with standard cataract surgery.
- The most frequent adverse event was corneal edema, and all cases were resolved with topical medications.

