MedicalResearch.com Interview with:
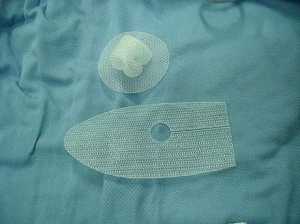
[caption id="attachment_44618" align="alignleft" width="200"] Polypropylene (PP) mesh is currently used in both hernia and pelvic organ prolapses - Wikipedia Image[/caption]
Professor David Taylor
Trinity Centre for Bioengineering
Trinity College Dublin
The University of Dublin, Ireland
MedicalResearch.com: What is the background for this study?
Response: The study was motivated by the emerging clinical problem of the failure of surgical mesh products used for transvaginal operations. Increasing numbers of women are experiencing the failure of mesh used to treated vaginal prolapse, urinary incontinence and other pelvic organ conditions.
I carried out a survey of all existing research, including medical case reports, meta studies of clinical outcomes and biomechanics research on the material, which is a knitted plastic mesh. A phenomenon known as "mesh erosion" causes damage to surrounding tissues and organs and can leave the person in severe and lasting pain.
Polypropylene (PP) mesh is currently used in both hernia and pelvic organ prolapses - Wikipedia Image[/caption]
Professor David Taylor
Trinity Centre for Bioengineering
Trinity College Dublin
The University of Dublin, Ireland
MedicalResearch.com: What is the background for this study?
Response: The study was motivated by the emerging clinical problem of the failure of surgical mesh products used for transvaginal operations. Increasing numbers of women are experiencing the failure of mesh used to treated vaginal prolapse, urinary incontinence and other pelvic organ conditions.
I carried out a survey of all existing research, including medical case reports, meta studies of clinical outcomes and biomechanics research on the material, which is a knitted plastic mesh. A phenomenon known as "mesh erosion" causes damage to surrounding tissues and organs and can leave the person in severe and lasting pain.
 Polypropylene (PP) mesh is currently used in both hernia and pelvic organ prolapses - Wikipedia Image[/caption]
Professor David Taylor
Trinity Centre for Bioengineering
Trinity College Dublin
The University of Dublin, Ireland
MedicalResearch.com: What is the background for this study?
Response: The study was motivated by the emerging clinical problem of the failure of surgical mesh products used for transvaginal operations. Increasing numbers of women are experiencing the failure of mesh used to treated vaginal prolapse, urinary incontinence and other pelvic organ conditions.
I carried out a survey of all existing research, including medical case reports, meta studies of clinical outcomes and biomechanics research on the material, which is a knitted plastic mesh. A phenomenon known as "mesh erosion" causes damage to surrounding tissues and organs and can leave the person in severe and lasting pain.
Polypropylene (PP) mesh is currently used in both hernia and pelvic organ prolapses - Wikipedia Image[/caption]
Professor David Taylor
Trinity Centre for Bioengineering
Trinity College Dublin
The University of Dublin, Ireland
MedicalResearch.com: What is the background for this study?
Response: The study was motivated by the emerging clinical problem of the failure of surgical mesh products used for transvaginal operations. Increasing numbers of women are experiencing the failure of mesh used to treated vaginal prolapse, urinary incontinence and other pelvic organ conditions.
I carried out a survey of all existing research, including medical case reports, meta studies of clinical outcomes and biomechanics research on the material, which is a knitted plastic mesh. A phenomenon known as "mesh erosion" causes damage to surrounding tissues and organs and can leave the person in severe and lasting pain.