29 Nov Tetravalent Vaccine Proves Promising In Protecting Children From Dengue
MedicalResearch.com Interview with:
Vianney Tricou DPhil
Takeda Vaccines Pte Ltd
Singapore
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Dengue fever is a painful, debilitating mosquito-borne disease caused by any one of the four closely related dengue virus serotypes. Forty percent of the world’s population lives under the threat of dengue, with approximately 390 million infections and 20,000 deaths occurring globally each year. Dengue virus can infect people of all ages and is a leading cause of serious illness among children in some countries in Latin America and Asia. Takeda is developing a dengue vaccine candidate to safely protect children and adults living in, or traveling to, endemic areas against all four dengue virus serotypes, regardless of previous dengue virus exposure.
Takeda’s tetravalent dengue vaccine candidate (TAK-003) is based on a live, attenuated dengue serotype 2 virus, which provides the genetic ‘backbone’ for all four vaccine viruses. Takeda’s ongoing Phase 2 DEN-204 study was designed to assess the safety and immunogenicity of different dose schedules of TAK-003 in approximately 1,800 healthy children and adolescents ages two through 17 years living in dengue-endemic countries in Latin America and Asia. Participants of the DEN-204 trial received either one primary dose of TAK-003, two primary doses of TAK-003 administered three months apart, one primary dose of TAK-003 followed by a booster dose one year later, or a placebo. Eighteen-month interim data showed that that TAK-003 is associated with a reduction in the incidence of dengue in the study participants. Data also showed that TAK-003 induced sustained antibody responses against all four serotypes of dengue virus, regardless of previous dengue exposure and dosing schedule.
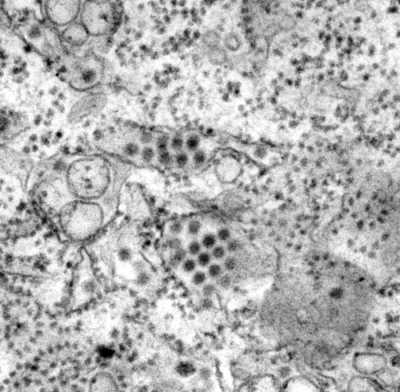
This transmission electron microscopic (TEM) image depicts a number of round, Dengue virus particles that were revealed in this tissue specimen.
These data were published in The Lancet Infectious Diseases in November 2017, and presented during the 66th Annual Meeting of The American Society of Tropical Medicine & Hygiene and the XVII Congreso Latinoamericano de Infectología Pediátrica – SLIPE 2017.
Febrile surveillance and assessment of safety and immunogenicity will continue through the 48-month study period of the trial. Final data collection is expected to be complete in July 2019.
MedicalResearch.com: What should clinicians and patients take away from your report?
Response: Eighteen-month interim data from DEN-204 showed that TAK-003 is associated with a reduction in the incidence of dengue in children and adolescents ages two through 17 years. Data also showed that TAK-003 induced sustained antibody responses against all four serotypes of dengue virus, regardless of previous dengue virus exposure and dosing schedule. These findings further support selection of a two-dose regimen, administered three months apart, for Takeda’s ongoing global pivotal Phase 3 efficacy trial. This regimen quickly achieves a high rate of response to all four dengue virus serotypes regardless of previous exposure to dengue virus.
Additionally, in this analysis, TAK-003 was found to be safe and well-tolerated in terms of solicited local reactions and systemic adverse events (AEs), relative to the placebo control group. This is consistent with data from previous Phase 1 and 2 trials, and occurrence of local reactions and all AEs was comparable to those observed in trials of other live, attenuated vaccines.
Ongoing follow-up will continue through the 48-month study period of the trial, and final data collection is expected to be complete in July 2019.
MedicalResearch.com: What recommendations do you have for future research as a result of this study?
Response: TAK-003 is currently under evaluation in the Tetravalent Immunization against Dengue Efficacy Study (TIDES), a large-scale Phase 3 efficacy trial being conducted in eight dengue-endemic countries. TIDES will build on DEN-204 and other previous studies in continuing to assess the tolerability, safety and immunogenicity of the vaccine against all four dengue serotypes in multiple age groups and to determine whether the vaccine prevents symptomatic dengue.
MedicalResearch.com: Is there anything else you would like to add?
Response: Takeda’s dengue vaccine candidate is an integral part of Takeda’s mission to apply world class expertise to develop and deliver innovative vaccines for infectious diseases that address important global public health needs.
Earlier this year, Takeda completed enrollment and vaccination of more than 20,000 children and adolescents ages 4 through 16 in our TIDES trial of TAK-003. This is a significant milestone for Takeda, as it is another step forward in the journey to develop and test a vaccine that is safe for and protects populations and individuals at risk of symptomatic dengue, including children, regardless of whether they have previously been exposed to the virus. This trial is ongoing, and data from TIDES will be available in late 2018.
MedicalResearch.com: Thank you for your contribution to the MedicalResearch.com community.
Citation:
Immunogenicity and safety of one versus two doses of tetravalent dengue vaccine in healthy children aged 2–17 years in Asia and Latin America: 18-month interim data from a phase 2, randomised, placebo-controlled study
Sáez-Llorens, Xavier et al.
The Lancet Infectious Diseases , November 6 2017
DOI: http://dx.doi.org/10.1016/S1473-3099(17)30632-1
Note: Content is Not intended as medical advice. Please consult your health care provider regarding your specific medical condition and questions.
[wysija_form id=”1″]
Last Updated on November 29, 2017 by Marie Benz MD FAAD
