24 Jun Antibody Points Way Toward Norovirus Vaccine
MedicalResearch.com Interview with:

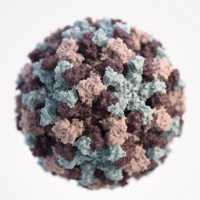
CDC Image
Based on electron microscopic (EM) imagery, this three-dimensional (3D) illustration provides a graphical representation of a single norovirus virion, set against a white background. Though subtle, the different colors represent different regions of the organism’s outer protein shell, or capsid. Illustrator: Alissa Eckert, MS
Lisa Lindesmith, MS
Research specialist
Ralph S. Baric, PhD
Professor, Departments of Epidemiology, Microbiology and Immunology
Lineberger Comprehensive Cancer Center
Gillings School of Global Public Health
University of North Carolina
MedicalResearch.com: What is the background for this study? Would you briefly explain the types of outbreaks caused by Norovirus infections?
Response: Noroviruses cause about 20% of endemic and 50% of food-borne acute gastroenteritis, infecting all age groups, globally. While may different strains of norovirus cause outbreaks primarily in community settings, since the mid-1990’s the GII.4 strains of norovirus have caused waves of pandemic disease every 2-7 years. These pandemics are associated with emergence of a GII.4 strain that has changed key viral domains rendering the virus less susceptible to recognition by and protection from a person’s immune system. For a vaccine to be efficacious against pandemic GII.4 strains, it must be able to train the immune system to focus on the part of the GII.4 virus that does not change over time.
MedicalResearch.com: What are the main findings?
Response: In this study, using samples collected from human volunteers vaccinated with a candidate norovirus vaccine, we identified key viral domains that shape the antibody response to GII.4 noroviruses following infection and vaccination. Most importantly, we identified a human antibody able to inhibit binding of diverse GII.4 strains that circulated over three decades. We then used that antibody binding site to map the location on the virus surface that is conserved across GII.4 strains, identifying a key target for vaccine and drug design that would provide cross-protection. Further, we identified other antibodies that bind to parts of the virus that change frequently, facilitating escape from pre-existing immunity. Monitoring of these hyper-variable regions on the virus will inform decisions on vaccine reformulation and public health systems preparation, as until a vaccine is available, behavioral modifications including hand washing are the best protection from norovirus infection.
MedicalResearch.com: What should readers take away from your report?
Response: Norovirus is a significant human pathogen causing ~700 million illnesses and >200,000 deaths annually, primarily in young children and the elderly. Extensive diversity within noroviruses has hampered development of a vaccine. Studies like this one, that define the virus landscape with respect to immunity, can guide design of vaccine candidates that preferentially promote targeting of virus domains that remain constant, and are most likely to generate longterm, broadly protective immune responses. Moreover, our studies reveal a potent antibody that can be used to treat life-threatening GII.4 infections in children and the elderly. The approaches used here are easily applicable to other human pathogens.
MedicalResearch.com: What recommendations do you have for future research as a result of this work?
Response: This study analyzed in detail antibodies from a small number of vaccinated adults. More individuals, including the elderly and children, key target populations for a norovirus vaccine, would provide needed information on the complex relationship between noroviruses and human immunity that could than be applied to design of vaccine candidates. Finally, we will need to prove that this potent cross reactive antibody can protect humans from norovirus gastroenteritis and infection.
Any disclosures?
Lisa Lindesmith and Ralph Baric have consulted and collaborated with Takeda on norovirus vaccines. We are also inventors on patents relevant to norovirus vaccines.
Citation:
Sera Antibody Repertoire Analyses Reveal Mechanisms of Broad and Pandemic Strain Neutralizing Responses after Human Norovirus Vaccination
Lisa C. Lindesmith, Jonathan R. McDaniel , Anita Changela, Peter D. Kwong, George Georgiou, Ralph S. Baric
Immunity: DOI :https://doi.org/10.1016/j.immuni.2019.05.007
[wysija_form id=”3″]
[last-modified]
The information on MedicalResearch.com is provided for educational purposes only, and is in no way intended to diagnose, cure, or treat any medical or other condition. Always seek the advice of your physician or other qualified health and ask your doctor any questions you may have regarding a medical condition. In addition to all other limitations and disclaimers in this agreement, service provider and its third party providers disclaim any liability or loss in connection with the content provided on this website.
Last Updated on June 24, 2019 by Marie Benz MD FAAD
