Author Interviews, COVID -19 Coronavirus / 09.09.2020
Digital Droplet Technology Can Reduce False Negative COVID-19 Tests
MedicalResearch.com Interview with:
Claudia Alteri, PhD
Assistant Professor (RTD-B) in Microbiology And Clinical Microbiology
Department of Oncology and Hemato-Oncology
University of Milano
MedicalResearch.com: What is the background for this study?
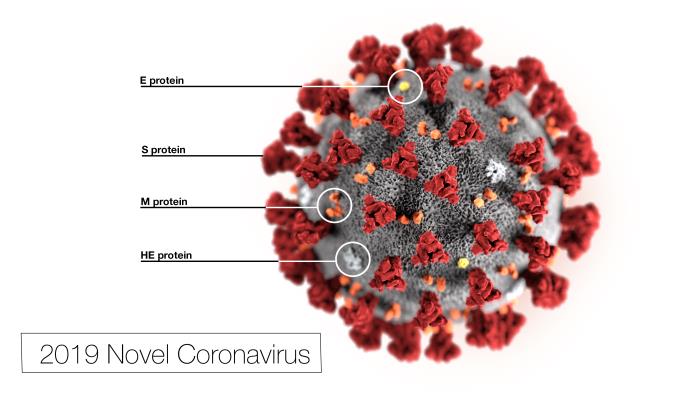
Response: In the context of SARS-CoV-2 diagnosis, laboratories play a critical role in confirming the initial clinical suspicion of this disease, as confirmation of SARS-CoV-2 presence is essential to ensure the prompt initiation of containment and treatment protocols. This is of utmost importance to avoid further spread of the pandemic, and to assure the best clinical and therapeutic management of the infected patients in the hospital setting. Unfortunately, currently used rtPCR assays lack of the necessary sensitivity to identify all cases of SARS-CoV-2 infection (20% of false negative results [Li D et all, Korean J Rad 2020; Zhao et all CID 2020]). Complementary laboratory assays are therefore strongly needed. Droplet Digital PCR (ddPCR) is a highly sensitive assay for the direct detection and quantification of DNA and RNA targets. It has been increasingly used in infectious disease settings, especially thanks to its ability to consistently and reliably detect down to few copies of viral genomes. Standing the necessity of a limitation (as much as possible) of false negative results in COVID-19 diagnosis, the use of ddPCR could provide a critical support. In the context of COVID-19 diagnosis, two recent studies highlight the good performances of ddPCR in detecting low viral load samples (Suo T MedRxiv 2020; You F MedRxiv 2020)





 Dr. Bin Cao[/caption]
Bin Cao, Yeming Wang, Guohui Fan,
Lianghan Shang, Jiuyang Xu, DingyuZhang, Chen Wang
on behalf of LOTUS-China Study Group
China-Japan Friendship Hospital; Wuhan Jintinyan Hospital;
Institute of Respiratory Medicine, Chinese Academy of Medical Science
MedicalResearch.com: What is the background for this study?
Response: In the past two months, the outbreak of Coronavirus Disease 2019 (COVID-19) has been spreading rapidly across the world. Science and technology is the most powerful weapon for human to fight against diseases, especially in such a pandemic setting. Seeking for effective antiviral medication is the most critical and urgent among the many scientific tasks in the pandemic.
At the most critical moment in the fight against COVID-19, Chinese clinical scientists have stepped forward under extremely difficult research conditions to carry out clinical trials in antiviral treatment including lopinavir–ritonavir and remdesivir, in a swift, decisive and effective manner. These trials have attracted worldwide attention.
Recently, the Lopinavir–ritonavir Trial for suppression of SARS-CoV-2 in China (LOTUS-China) has been completed, which, with great clinical significance, can provide strong evidence for the treatment of COVID-19 both in China and around the world.
Dr. Bin Cao[/caption]
Bin Cao, Yeming Wang, Guohui Fan,
Lianghan Shang, Jiuyang Xu, DingyuZhang, Chen Wang
on behalf of LOTUS-China Study Group
China-Japan Friendship Hospital; Wuhan Jintinyan Hospital;
Institute of Respiratory Medicine, Chinese Academy of Medical Science
MedicalResearch.com: What is the background for this study?
Response: In the past two months, the outbreak of Coronavirus Disease 2019 (COVID-19) has been spreading rapidly across the world. Science and technology is the most powerful weapon for human to fight against diseases, especially in such a pandemic setting. Seeking for effective antiviral medication is the most critical and urgent among the many scientific tasks in the pandemic.
At the most critical moment in the fight against COVID-19, Chinese clinical scientists have stepped forward under extremely difficult research conditions to carry out clinical trials in antiviral treatment including lopinavir–ritonavir and remdesivir, in a swift, decisive and effective manner. These trials have attracted worldwide attention.
Recently, the Lopinavir–ritonavir Trial for suppression of SARS-CoV-2 in China (LOTUS-China) has been completed, which, with great clinical significance, can provide strong evidence for the treatment of COVID-19 both in China and around the world.

