Immunotherapy, Pharmaceutical Companies / 26.03.2026
The Evolving Landscape of Immunogenicity Risk Assessment in Biologic Drug Development
[caption id="attachment_72949" align="aligncenter" width="500"] Photo by Daniel Dan on Unsplash[/caption]
As biologic therapeutics continue to reshape modern medicine — from monoclonal antibodies to gene therapies and fusion proteins — one of the most consequential challenges facing drug developers remains largely invisible to the public: the immune response a patient’s own body may mount against a therapeutic protein. Immunogenicity, defined as the propensity of a biologic to trigger an unwanted immune response, can range from a clinically silent laboratory finding to a life-threatening hypersensitivity reaction or a complete loss of therapeutic efficacy. Understanding and mitigating this risk has become a cornerstone of the modern drug development paradigm.
Photo by Daniel Dan on Unsplash[/caption]
As biologic therapeutics continue to reshape modern medicine — from monoclonal antibodies to gene therapies and fusion proteins — one of the most consequential challenges facing drug developers remains largely invisible to the public: the immune response a patient’s own body may mount against a therapeutic protein. Immunogenicity, defined as the propensity of a biologic to trigger an unwanted immune response, can range from a clinically silent laboratory finding to a life-threatening hypersensitivity reaction or a complete loss of therapeutic efficacy. Understanding and mitigating this risk has become a cornerstone of the modern drug development paradigm.
 Photo by Daniel Dan on Unsplash[/caption]
As biologic therapeutics continue to reshape modern medicine — from monoclonal antibodies to gene therapies and fusion proteins — one of the most consequential challenges facing drug developers remains largely invisible to the public: the immune response a patient’s own body may mount against a therapeutic protein. Immunogenicity, defined as the propensity of a biologic to trigger an unwanted immune response, can range from a clinically silent laboratory finding to a life-threatening hypersensitivity reaction or a complete loss of therapeutic efficacy. Understanding and mitigating this risk has become a cornerstone of the modern drug development paradigm.
Photo by Daniel Dan on Unsplash[/caption]
As biologic therapeutics continue to reshape modern medicine — from monoclonal antibodies to gene therapies and fusion proteins — one of the most consequential challenges facing drug developers remains largely invisible to the public: the immune response a patient’s own body may mount against a therapeutic protein. Immunogenicity, defined as the propensity of a biologic to trigger an unwanted immune response, can range from a clinically silent laboratory finding to a life-threatening hypersensitivity reaction or a complete loss of therapeutic efficacy. Understanding and mitigating this risk has become a cornerstone of the modern drug development paradigm.















 Lauren C. Davis, MBS
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA 19409
MedicalResearch.com: What is the background for this study?
Response: Financial conflicts of interest (COIs) resulting from ties between academia and industry have been under scrutiny for their potential to hinder the integrity of medical research. COIs can lead to implicit bias, compromise the research process, and erode public trust (1-6). The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders (DSM), standardizes symptom criteria and codifies psychiatric disorders. This manual contributes to the approval of new drugs, extensions of patent exclusivity, and can influence payers and mental health professionals seeking third-party reimbursements. Given the implications of the DSM on public health, it is paramount that it is free of industry influence. Previous research has shown a high prevalence of industry ties among panel and task force members of the DSM-IV-TR and DSM-5, despite the implementation of a disclosure policy for the DSM-5 (7,8). This study (9) determined the extent and type of COIs received by panel and task-force members of the DSM-5-TR (2022) (10). As the DSM-5-TR did not disclose COI, we used the Center for Medicare and Medicaid Services Open Payments (OP) database (11) to quantify them.
Lauren C. Davis, MBS
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA 19409
MedicalResearch.com: What is the background for this study?
Response: Financial conflicts of interest (COIs) resulting from ties between academia and industry have been under scrutiny for their potential to hinder the integrity of medical research. COIs can lead to implicit bias, compromise the research process, and erode public trust (1-6). The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders (DSM), standardizes symptom criteria and codifies psychiatric disorders. This manual contributes to the approval of new drugs, extensions of patent exclusivity, and can influence payers and mental health professionals seeking third-party reimbursements. Given the implications of the DSM on public health, it is paramount that it is free of industry influence. Previous research has shown a high prevalence of industry ties among panel and task force members of the DSM-IV-TR and DSM-5, despite the implementation of a disclosure policy for the DSM-5 (7,8). This study (9) determined the extent and type of COIs received by panel and task-force members of the DSM-5-TR (2022) (10). As the DSM-5-TR did not disclose COI, we used the Center for Medicare and Medicaid Services Open Payments (OP) database (11) to quantify them.

 Dr. Mallbris[/caption]
Lotus Mallbris, M.D., Ph.D.,
Senior Vice President of Immunology Development
Dr. Mallbris[/caption]
Lotus Mallbris, M.D., Ph.D.,
Senior Vice President of Immunology Development
 Dr. Dubinsky[/caption]
Marla C. Dubinsky, MD
Professor of Pediatrics and Medicine
Icahn School of Medicine at Mount Sinai
Co- director, Susan and Leonard Feinstein IBD Clinical Center
Mount Sinai Health System
MedicalResearch.com: What is the background for this study? Would you briefly describe the condition of UC?
Response: Lucent 1 and Lucent 2 were the induction and maintenance registration trials studying the efficacy and safety of mirikizumab in patients 18 years and older with moderate to severely active ulcerative colitis. Mirikizumab is a monoclonal antibody targeting the p19 subunit of IL23. Lucent-3 is the open label extension arm for those meeting inclusion criteria after completing Lucent 2. This study evaluated the long term efficacy and safety of mirikizumab in patients with ulcerative colitis who completed a total of 104 weeks of active mirikizumab treatment.
Ulcerative colitis is a chronic incurable inflammatory condition of colon. Common symptoms include diarrhea, blood in the stool, abdominal cramping and bowel urgency. Bowel urgency is one of the most burdensome symptoms that a patient with you could experience.
Dr. Dubinsky[/caption]
Marla C. Dubinsky, MD
Professor of Pediatrics and Medicine
Icahn School of Medicine at Mount Sinai
Co- director, Susan and Leonard Feinstein IBD Clinical Center
Mount Sinai Health System
MedicalResearch.com: What is the background for this study? Would you briefly describe the condition of UC?
Response: Lucent 1 and Lucent 2 were the induction and maintenance registration trials studying the efficacy and safety of mirikizumab in patients 18 years and older with moderate to severely active ulcerative colitis. Mirikizumab is a monoclonal antibody targeting the p19 subunit of IL23. Lucent-3 is the open label extension arm for those meeting inclusion criteria after completing Lucent 2. This study evaluated the long term efficacy and safety of mirikizumab in patients with ulcerative colitis who completed a total of 104 weeks of active mirikizumab treatment.
Ulcerative colitis is a chronic incurable inflammatory condition of colon. Common symptoms include diarrhea, blood in the stool, abdominal cramping and bowel urgency. Bowel urgency is one of the most burdensome symptoms that a patient with you could experience.


 Dr. Tauscher-Wisniewski,[/caption]
Sitra Tauscher-Wisniewski, MD
Vice President Clinical Development & Analytics
Novartis Gene Therapies
MedicalResearch.com: What is the background for this study? Would you briefly describe the condition of Spinal muscular atrophy (SMA)?
Response: At the 2023 Muscular Dystrophy Association Conference, we presented new data from two of our Long-Term Follow-Up (LTFU) studies, LT001 and LT002, which show the continued efficacy and durability of Zolgensma across a range of patient populations, with an overall benefit-risk profile that remains favorable. LT001 is a 15-year ongoing observational LTFU study following the Phase 1 START patients, who were the very first patients to receive our gene replacement therapy. LT-002 is a voluntary Phase 4 15-year ongoing follow-up safety and efficacy study of Zolgensma IV and investigational intrathecal (IT) OAV101 in patients previously treated in the Phase 3 IV studies (STR1VE-US, STR1VE-EU, STR1VE-AP, SPR1NT) and the Phase 1 IT study (STRONG).
Spinal muscular atrophy (SMA) is a rare, devastating genetic disease that leads to progressive muscle weakness, paralysis, and when left untreated in one of its most severe forms (SMA Type 1), permanent ventilation or death in 90% of cases by age 2. It is caused by a lack of a functional survival motor neuron 1 (SMN1) gene, and in the most severe forms results in the rapid and irreversible loss of motor neurons, affecting muscle functions, including breathing, swallowing and basic movement.
Dr. Tauscher-Wisniewski,[/caption]
Sitra Tauscher-Wisniewski, MD
Vice President Clinical Development & Analytics
Novartis Gene Therapies
MedicalResearch.com: What is the background for this study? Would you briefly describe the condition of Spinal muscular atrophy (SMA)?
Response: At the 2023 Muscular Dystrophy Association Conference, we presented new data from two of our Long-Term Follow-Up (LTFU) studies, LT001 and LT002, which show the continued efficacy and durability of Zolgensma across a range of patient populations, with an overall benefit-risk profile that remains favorable. LT001 is a 15-year ongoing observational LTFU study following the Phase 1 START patients, who were the very first patients to receive our gene replacement therapy. LT-002 is a voluntary Phase 4 15-year ongoing follow-up safety and efficacy study of Zolgensma IV and investigational intrathecal (IT) OAV101 in patients previously treated in the Phase 3 IV studies (STR1VE-US, STR1VE-EU, STR1VE-AP, SPR1NT) and the Phase 1 IT study (STRONG).
Spinal muscular atrophy (SMA) is a rare, devastating genetic disease that leads to progressive muscle weakness, paralysis, and when left untreated in one of its most severe forms (SMA Type 1), permanent ventilation or death in 90% of cases by age 2. It is caused by a lack of a functional survival motor neuron 1 (SMN1) gene, and in the most severe forms results in the rapid and irreversible loss of motor neurons, affecting muscle functions, including breathing, swallowing and basic movement.




 SooYoung VanDeMar[/caption]
SooYoung VanDeMark, MBS
Geisinger Commonwealth School of Medicine
Scranton, Pennsylvania
MedicalResearch.com: What is the background for this study?
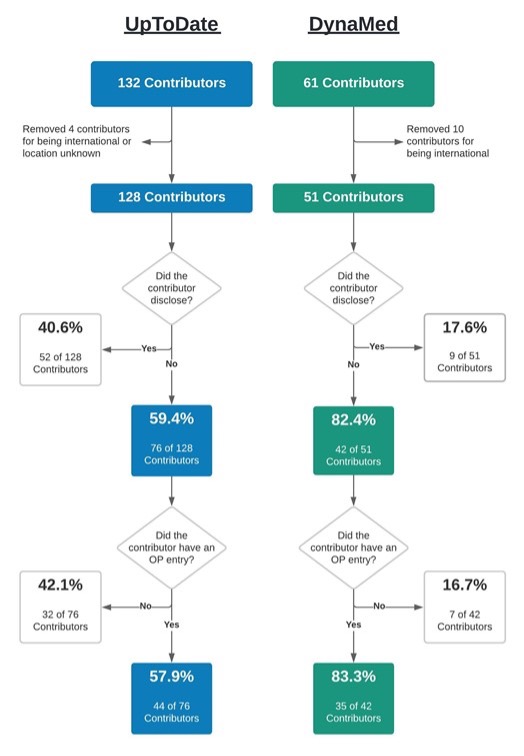
Response: Health care providers utilize subscription-based, point-of-care databases such as DynaMed and UpToDate to provide clinical care guidance and remain current on the latest evidence-based findings. Both of these websites maintain this content through a cadre of physician contributors who write and edit articles for these sites. These physician contributors are required to self-report any conflicts of interest (COI) as outlined by the respective policies on each website. However, prior COI research into similarly self-regulated areas, such as medical and pharmacology textbooks, and clinical practice guidelines, has found both appreciable potential COI and inconsistencies between self-reported and industry mandated disclosures (1-3).
This study (4) explored the accuracy of physician contributors to DynaMed and UpToDate by comparing their self-reported disclosure status with the financial remunerations they received from the healthcare industry (e.g., pharmaceutical companies) as reported to the U.S. Centers for Medicare and Medicaid Services’ Open Payments database. Physician contributors who reported “nothing to disclose” on their respective article topic but had an entry on Open Payments for having received money from industry, were classified as discordant and, thus, as having the potential for a COI. Additionally, total remuneration, gender, and payment category were investigated more in depth for each database.
SooYoung VanDeMar[/caption]
SooYoung VanDeMark, MBS
Geisinger Commonwealth School of Medicine
Scranton, Pennsylvania
MedicalResearch.com: What is the background for this study?
Response: Health care providers utilize subscription-based, point-of-care databases such as DynaMed and UpToDate to provide clinical care guidance and remain current on the latest evidence-based findings. Both of these websites maintain this content through a cadre of physician contributors who write and edit articles for these sites. These physician contributors are required to self-report any conflicts of interest (COI) as outlined by the respective policies on each website. However, prior COI research into similarly self-regulated areas, such as medical and pharmacology textbooks, and clinical practice guidelines, has found both appreciable potential COI and inconsistencies between self-reported and industry mandated disclosures (1-3).
This study (4) explored the accuracy of physician contributors to DynaMed and UpToDate by comparing their self-reported disclosure status with the financial remunerations they received from the healthcare industry (e.g., pharmaceutical companies) as reported to the U.S. Centers for Medicare and Medicaid Services’ Open Payments database. Physician contributors who reported “nothing to disclose” on their respective article topic but had an entry on Open Payments for having received money from industry, were classified as discordant and, thus, as having the potential for a COI. Additionally, total remuneration, gender, and payment category were investigated more in depth for each database.



