Allergies, Asthma, Author Interviews / 15.09.2022
Does Pollen Cause Your Asthma Here’s How To Handle It
Knowing about pollen forecasts might help you understand how much pollen will be released in your area and when....
Knowing about pollen forecasts might help you understand how much pollen will be released in your area and when....
 Dr. Bacharier[/caption]
Leonard B. Bacharier, MD
Janie Robinson and John Moore Lee Chair in Pediatrics
Professor of Pediatrics
Director - Center for Pediatric Asthma Research
Scientific Director - Center for Clinical and Translational Research
Section Chief - Pediatric Allergy and Immunology
Division of Allergy, Immunology and Pulmonary Medicine
Monroe Carell Jr. Children’s Hospital at Vanderbilt University Medical Center
MedicalResearch.com: What is the background for this study? Is Dupilumab used for other atopic conditions, ie eczema/atopic dermatitis?
Response: Many children with moderate-severe asthma continue to experience asthma exacerbations and poor asthma control despite use of controller therapies. Dupilumab has been shown to reduce asthma exacerbations in adolescents and adults, as well as to improve atopic dermatitis in children and adults.
Dr. Bacharier[/caption]
Leonard B. Bacharier, MD
Janie Robinson and John Moore Lee Chair in Pediatrics
Professor of Pediatrics
Director - Center for Pediatric Asthma Research
Scientific Director - Center for Clinical and Translational Research
Section Chief - Pediatric Allergy and Immunology
Division of Allergy, Immunology and Pulmonary Medicine
Monroe Carell Jr. Children’s Hospital at Vanderbilt University Medical Center
MedicalResearch.com: What is the background for this study? Is Dupilumab used for other atopic conditions, ie eczema/atopic dermatitis?
Response: Many children with moderate-severe asthma continue to experience asthma exacerbations and poor asthma control despite use of controller therapies. Dupilumab has been shown to reduce asthma exacerbations in adolescents and adults, as well as to improve atopic dermatitis in children and adults. Prof. Brightling[/caption]
Professor Chris Brightling, NIHR Senior Investigator
Department of Respiratory Sciences
University of Leicester
Leicester, UK
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Risankizumab is an anti-IL23 monoclonal antibody. It is a very effective licensed therapy for plaque psoriasis. IL23 has been implicated in asthma and therefore we chose to study the effect of risankizumab in people with severe asthma.
The time to first asthma worsening was earlier and the frequency of asthma worsenings was higher in those treated with risankizumab versus placebo.
We found that the gene expression of key molecules involved in the response to infection were decreased in airway samples in those treated with risankizumab. It is possible that the increased asthma worsening following risankizumab was related to this suppression of anti-microbial immunity.
Prof. Brightling[/caption]
Professor Chris Brightling, NIHR Senior Investigator
Department of Respiratory Sciences
University of Leicester
Leicester, UK
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Risankizumab is an anti-IL23 monoclonal antibody. It is a very effective licensed therapy for plaque psoriasis. IL23 has been implicated in asthma and therefore we chose to study the effect of risankizumab in people with severe asthma.
The time to first asthma worsening was earlier and the frequency of asthma worsenings was higher in those treated with risankizumab versus placebo.
We found that the gene expression of key molecules involved in the response to infection were decreased in airway samples in those treated with risankizumab. It is possible that the increased asthma worsening following risankizumab was related to this suppression of anti-microbial immunity.  Dr. Salciccioli[/caption]
Justin Salciccioli, MBBS, MA
Research Fellow in Medicine
[caption id="attachment_57392" align="alignleft" width="150"]
Dr. Salciccioli[/caption]
Justin Salciccioli, MBBS, MA
Research Fellow in Medicine
[caption id="attachment_57392" align="alignleft" width="150"] Dr. Israel[/caption]
Elliot Israel, MD
Professor of Medicine, Harvard Medical School
Pulmonary and Critical Care, Rheumatology, Medicine
Brigham and Women's Hospital
MedicalResearch.com: What is the background for this study?
Response: Asthma attacks account for almost 50% of the cost of asthma care, which costs $80 billion each year in the United States. Asthma is more severe in African-American/Black and Hispanic/Latinx patients, with these groups having double the rates of attacks and hospitalizations as the general population. The PREPARE study is an ongoing national clinical trial for African American/Black and Hispanic/Latinx adults with moderate-to-severe asthma from different U.S. cities in which reporting of asthma control and asthma exacerbations was monitored entirely remotely.
With the arrival of the Covid19 pandemic, several studies suggested that asthma exacerbations may have decreased during the pandemic. However, multiple reports have suggested people were avoiding health services because of the pandemic, making it difficult to tell whether exacerbations truly decreased or whether people were simply avoiding their doctors. This is the first study done to assess asthma exacerbations before and during the pandemic that is unlikely to be impacted by patient healthcare avoidance.
Dr. Israel[/caption]
Elliot Israel, MD
Professor of Medicine, Harvard Medical School
Pulmonary and Critical Care, Rheumatology, Medicine
Brigham and Women's Hospital
MedicalResearch.com: What is the background for this study?
Response: Asthma attacks account for almost 50% of the cost of asthma care, which costs $80 billion each year in the United States. Asthma is more severe in African-American/Black and Hispanic/Latinx patients, with these groups having double the rates of attacks and hospitalizations as the general population. The PREPARE study is an ongoing national clinical trial for African American/Black and Hispanic/Latinx adults with moderate-to-severe asthma from different U.S. cities in which reporting of asthma control and asthma exacerbations was monitored entirely remotely.
With the arrival of the Covid19 pandemic, several studies suggested that asthma exacerbations may have decreased during the pandemic. However, multiple reports have suggested people were avoiding health services because of the pandemic, making it difficult to tell whether exacerbations truly decreased or whether people were simply avoiding their doctors. This is the first study done to assess asthma exacerbations before and during the pandemic that is unlikely to be impacted by patient healthcare avoidance.
 Yun-Han Wang[/caption]
Yun-Han Wang MSc
PhD student, Karolinska Institute
MedicalResearch.com: What is the background for this study?
Response: The use of proton pump inhibitors (PPIs) in children has increased substantially in recent years, concurrently with emerging concerns that these drugs may increase the risk of asthma. Whether PPI use in the broad pediatric population is associated with increased risk of asthma is not known.
Yun-Han Wang[/caption]
Yun-Han Wang MSc
PhD student, Karolinska Institute
MedicalResearch.com: What is the background for this study?
Response: The use of proton pump inhibitors (PPIs) in children has increased substantially in recent years, concurrently with emerging concerns that these drugs may increase the risk of asthma. Whether PPI use in the broad pediatric population is associated with increased risk of asthma is not known.  Dr. Frank Trudo[/caption]
Frank Trudo, MD MBA
Vice President, US Medical, Respiratory & Immunology
AstraZeneca
MedicalResearch.com: What is the background for this study?
Response: PONENTE is a multicenter, open-label, single-arm, Phase IIIb trial to evaluate the efficacy and safety of reducing daily oral corticosteroids (OCS) use after initiation of 30 mg dose of FASENRA (benralizumab) administered subcutaneously in adult patients with severe eosinophilic asthma on high-dose inhaled corticosteroids plus long-acting beta2-agonist and long-term use of OCS therapy with or without additional asthma controller(s). The trial expands on OCS-sparing data previously seen in the ZONDA Phase III trial by using a faster steroid tapering schedule in patients who did not experience adrenal insufficiency to reduce OCS use from higher doses.
Compared to published trials of other biologics, PONENTE has a personalized OCS tapering schedule that allows for more rapid OCS tapering from higher OCS doses, followed by an assessment of the adrenal function as part of decision-making to manage the risk of adrenal insufficiency. PONENTE also has a longer maintenance phase (approximately 24-32 weeks), allowing assessment of the durability of OCS reduction.
FASENRA is a monoclonal antibody that binds directly to IL-5 receptor alpha on eosinophils and attracts natural killer cells to induce rapid and near-complete depletion of eosinophils via apoptosis (programmed cell death).
MedicalResearch.com: How is it administered?
Response: FASENRA is injected under your skin (subcutaneously) one time every 4 weeks for the first 3 doses, and then every 8 weeks. In 2019, FASENRA was approved in the US for self-administration in a single dose prefilled autoinjector, the FASENRA pen.
Dr. Frank Trudo[/caption]
Frank Trudo, MD MBA
Vice President, US Medical, Respiratory & Immunology
AstraZeneca
MedicalResearch.com: What is the background for this study?
Response: PONENTE is a multicenter, open-label, single-arm, Phase IIIb trial to evaluate the efficacy and safety of reducing daily oral corticosteroids (OCS) use after initiation of 30 mg dose of FASENRA (benralizumab) administered subcutaneously in adult patients with severe eosinophilic asthma on high-dose inhaled corticosteroids plus long-acting beta2-agonist and long-term use of OCS therapy with or without additional asthma controller(s). The trial expands on OCS-sparing data previously seen in the ZONDA Phase III trial by using a faster steroid tapering schedule in patients who did not experience adrenal insufficiency to reduce OCS use from higher doses.
Compared to published trials of other biologics, PONENTE has a personalized OCS tapering schedule that allows for more rapid OCS tapering from higher OCS doses, followed by an assessment of the adrenal function as part of decision-making to manage the risk of adrenal insufficiency. PONENTE also has a longer maintenance phase (approximately 24-32 weeks), allowing assessment of the durability of OCS reduction.
FASENRA is a monoclonal antibody that binds directly to IL-5 receptor alpha on eosinophils and attracts natural killer cells to induce rapid and near-complete depletion of eosinophils via apoptosis (programmed cell death).
MedicalResearch.com: How is it administered?
Response: FASENRA is injected under your skin (subcutaneously) one time every 4 weeks for the first 3 doses, and then every 8 weeks. In 2019, FASENRA was approved in the US for self-administration in a single dose prefilled autoinjector, the FASENRA pen.
 Beth Hahn, PhD, Director
U.S. Value Evidence and Outcomes,
GlaxoSmithKline,
Research Triangle Park, NC
MedicalResearch.com: What is the background for this study?
Response: Biologic therapies are increasing as a treatment for patients with severe asthma, with multiple therapies approved by the FDA. There is also an increasing understanding of the factors influencing preference for and adherence to biologic therapies for patients with severe asthma; however, little is known about why patients discontinue biologic therapy. In patients who have access to biologic treatment, understanding the circumstances and asthma characteristics associated with discontinuation of biologic therapy may allow for the identification of barriers to treatment success . The objective of this study of cross-sectional physician and patient survey data was to assess the patient characteristics and the given reasons for treatment discontinuation in a US patient cohort with severe asthma treated with biologic therapy.
A total of 117 physicians and 285 patients completed surveys with 70% of patients continuing biologic therapy (N=200). This study included a number of different FDA approved biologics. From the perspectives of the physicians included in the current study (85 providing a rationale for discontinuation), the majority reported a lack of symptom control, particularly shortness of breath (46%), exacerbations (26%) and other chronic symptoms (29%), as a key reason for discontinuing biologic therapy in severe asthma. Symptom control was also key for patients, with these three symptom categories among their top six reasons for biologic discontinuation. The cost of biologic treatment was also an important factor, cited as the 5th most common reason for discontinuation among physicians and the 3rd among patients.
Beth Hahn, PhD, Director
U.S. Value Evidence and Outcomes,
GlaxoSmithKline,
Research Triangle Park, NC
MedicalResearch.com: What is the background for this study?
Response: Biologic therapies are increasing as a treatment for patients with severe asthma, with multiple therapies approved by the FDA. There is also an increasing understanding of the factors influencing preference for and adherence to biologic therapies for patients with severe asthma; however, little is known about why patients discontinue biologic therapy. In patients who have access to biologic treatment, understanding the circumstances and asthma characteristics associated with discontinuation of biologic therapy may allow for the identification of barriers to treatment success . The objective of this study of cross-sectional physician and patient survey data was to assess the patient characteristics and the given reasons for treatment discontinuation in a US patient cohort with severe asthma treated with biologic therapy.
A total of 117 physicians and 285 patients completed surveys with 70% of patients continuing biologic therapy (N=200). This study included a number of different FDA approved biologics. From the perspectives of the physicians included in the current study (85 providing a rationale for discontinuation), the majority reported a lack of symptom control, particularly shortness of breath (46%), exacerbations (26%) and other chronic symptoms (29%), as a key reason for discontinuing biologic therapy in severe asthma. Symptom control was also key for patients, with these three symptom categories among their top six reasons for biologic discontinuation. The cost of biologic treatment was also an important factor, cited as the 5th most common reason for discontinuation among physicians and the 3rd among patients.
 Christos V. Chalitsios[/caption]
Christos Chalitsios B.Sc, M.Sc PhD student
Funded by British Medical Association (BMA)
School of Medicine
Division of Respiratory Medicine
University of Nottingham
City Hospital,Nottingham
MedicalResearch.com: What is the background for this study?
Response: Inhaled (ICS) and oral (OCS) corticosteroids play a crucial role in the control of airway inflammation in asthma. Given that the use of ICS in asthma is likely to increase with the recent change in GINA guidance recommending combined long-acting-β2-agonists with ICS at step 1 and the upward trend in prescribing of OCS, we sought to clarify the link between steroids, osteoporosis and FF in patients with asthma, stratifying the risk by dose, number of courses and type of steroids. The prevalence of patients receiving at least one bisphosphonate was also calculated.
Christos V. Chalitsios[/caption]
Christos Chalitsios B.Sc, M.Sc PhD student
Funded by British Medical Association (BMA)
School of Medicine
Division of Respiratory Medicine
University of Nottingham
City Hospital,Nottingham
MedicalResearch.com: What is the background for this study?
Response: Inhaled (ICS) and oral (OCS) corticosteroids play a crucial role in the control of airway inflammation in asthma. Given that the use of ICS in asthma is likely to increase with the recent change in GINA guidance recommending combined long-acting-β2-agonists with ICS at step 1 and the upward trend in prescribing of OCS, we sought to clarify the link between steroids, osteoporosis and FF in patients with asthma, stratifying the risk by dose, number of courses and type of steroids. The prevalence of patients receiving at least one bisphosphonate was also calculated.
 Response: Asthma is a chronic, progressive disease driven in part by underlying inflammation and requires long-term control of symptoms. Over time, this chronic inflammation can lead to a decline in lung function.
The Phase 3 open-label extension trial evaluated long-term safety and efficacy of Dupixent treatment in adults and adolescents with moderate-to-severe asthma who had previously participated in a controlled Dupixent clinical trial, including three pivotal trials that lasted between 24 and 52 weeks. With more than 2,200 patients enrolled, the Phase 3 LIBERTY ASTHMA TRAVERSE open-label extension trial is the largest of a biologic medicine ever conducted in asthma.
Dupixent is a biologic therapy that works differently from existing therapies that treat asthma. Dupixent is a fully-human monoclonal antibody that inhibits the signaling of the interleukin-4 (IL-4) and interleukin-13 (IL-13) proteins. Data from Dupixent clinical trials have shown that interleukin-4 (IL-4) and interleukin-13 (IL-13) are key drivers of the type 2 inflammation that plays a major role in asthma. It is the only biologic to demonstrate sustained improvements in lung function and asthma exacerbations across a broad patient population with type 2 inflammation. Dupixent is not an immunosuppressant.
Response: Asthma is a chronic, progressive disease driven in part by underlying inflammation and requires long-term control of symptoms. Over time, this chronic inflammation can lead to a decline in lung function.
The Phase 3 open-label extension trial evaluated long-term safety and efficacy of Dupixent treatment in adults and adolescents with moderate-to-severe asthma who had previously participated in a controlled Dupixent clinical trial, including three pivotal trials that lasted between 24 and 52 weeks. With more than 2,200 patients enrolled, the Phase 3 LIBERTY ASTHMA TRAVERSE open-label extension trial is the largest of a biologic medicine ever conducted in asthma.
Dupixent is a biologic therapy that works differently from existing therapies that treat asthma. Dupixent is a fully-human monoclonal antibody that inhibits the signaling of the interleukin-4 (IL-4) and interleukin-13 (IL-13) proteins. Data from Dupixent clinical trials have shown that interleukin-4 (IL-4) and interleukin-13 (IL-13) are key drivers of the type 2 inflammation that plays a major role in asthma. It is the only biologic to demonstrate sustained improvements in lung function and asthma exacerbations across a broad patient population with type 2 inflammation. Dupixent is not an immunosuppressant.
 Dr. Frank Trudo[/caption]
Frank Trudo, MD MBA
Vice President, US Medical Affairs
Respiratory & Immunology
AstraZeneca
MedicalResearch.com: What is the background for this study?
Dr. Frank Trudo[/caption]
Frank Trudo, MD MBA
Vice President, US Medical Affairs
Respiratory & Immunology
AstraZeneca
MedicalResearch.com: What is the background for this study?
 Dr. Martin[/caption]
Ubaldo Martin MD
VP Clinical Respiratory
RIA Late Stage Development
AstraZeneca
MedicalResearch.com: What is the background for this study?
Response: BORA was an extension study evaluating the long-term safety and specific aspects of efficacy in patients who had previously been in the benralizumab pivotal studies. After the patients completed the pivotal studies (Calima, Sirocco and Zonda), they were eligible to join BORA which followed adults for an additional year and adolescent for an additional 2 years. All patients receive one of two dosages of benralizumab. The abstract reports the outcomes of adolescents in the BORA study who were followed for approximately 3 years in total.
Dr. Martin[/caption]
Ubaldo Martin MD
VP Clinical Respiratory
RIA Late Stage Development
AstraZeneca
MedicalResearch.com: What is the background for this study?
Response: BORA was an extension study evaluating the long-term safety and specific aspects of efficacy in patients who had previously been in the benralizumab pivotal studies. After the patients completed the pivotal studies (Calima, Sirocco and Zonda), they were eligible to join BORA which followed adults for an additional year and adolescent for an additional 2 years. All patients receive one of two dosages of benralizumab. The abstract reports the outcomes of adolescents in the BORA study who were followed for approximately 3 years in total.
 Olga Ryan, DrPH, MPH, MBA
Regional Clinical Account Director, Southwest
AstraZeneca
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Asthma is common and imparts a substantial societal burden. It is well documented that asthma prevalence varies between males and females. Before puberty, more boys have asthma. Following puberty, a greater proportion of women suffer with asthma. We also have observed that women experience greater morbidity from the illness, greater healthcare resource utilization and suboptimal response for guideline recommended therapies (ICS, ICS/LABA).
Rationale for this study focused on describing asthma related outcomes between a well characterized severe asthma cohort, with intent in delineating differences among the sexes. With the advent of targeted biological medicines for severe asthma, as well as apparent gaps in knowledge, we wanted to understand potential sex-specific disease indicators in a well characterized severe asthma cohort.
Olga Ryan, DrPH, MPH, MBA
Regional Clinical Account Director, Southwest
AstraZeneca
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Asthma is common and imparts a substantial societal burden. It is well documented that asthma prevalence varies between males and females. Before puberty, more boys have asthma. Following puberty, a greater proportion of women suffer with asthma. We also have observed that women experience greater morbidity from the illness, greater healthcare resource utilization and suboptimal response for guideline recommended therapies (ICS, ICS/LABA).
Rationale for this study focused on describing asthma related outcomes between a well characterized severe asthma cohort, with intent in delineating differences among the sexes. With the advent of targeted biological medicines for severe asthma, as well as apparent gaps in knowledge, we wanted to understand potential sex-specific disease indicators in a well characterized severe asthma cohort.
 Yen Chung, PharmD
Payer Evidence Director
US Medical Affairs, AstraZeneca
MedicalResearch.com: What is the background for this study?
Response: Among patients with persistent asthma, use of systemic corticosteroids (SCS) is typically reserved for treatment of asthma exacerbations and as a supplemental maintenance therapy for patients whose disease remains uncontrolled with maximum maintenance controller therapies. However, SCS therapy comes with known risks for acute and chronic complications. It is well established that patients with severe asthma are responsible for a disproportionate amount of the economic burden of asthma; however, less clear is the extent to which systemic corticosteroids use and its consequences specifically contributes to the cost burden of asthma.
The purpose of this study was to use administrative claims to follow asthma patients with and without SCS treatment for up to 3 years and compare their complication rates, health care resource utilization, and costs.
Yen Chung, PharmD
Payer Evidence Director
US Medical Affairs, AstraZeneca
MedicalResearch.com: What is the background for this study?
Response: Among patients with persistent asthma, use of systemic corticosteroids (SCS) is typically reserved for treatment of asthma exacerbations and as a supplemental maintenance therapy for patients whose disease remains uncontrolled with maximum maintenance controller therapies. However, SCS therapy comes with known risks for acute and chronic complications. It is well established that patients with severe asthma are responsible for a disproportionate amount of the economic burden of asthma; however, less clear is the extent to which systemic corticosteroids use and its consequences specifically contributes to the cost burden of asthma.
The purpose of this study was to use administrative claims to follow asthma patients with and without SCS treatment for up to 3 years and compare their complication rates, health care resource utilization, and costs.  Dr. Han[/caption]
Joseph Han, MD FARS, FAAOA FAAAAI
Medical Director for the Division of Allergy
Eastern Virginia Medical School
MedicalResearch.com: What is the background for this study?
Response: Chronic rhinosinusitis with nasal polyps (CRSwNP) is a type 2 inflammatory disease of the nasal passages and sinuses associated with a high symptom burden due to the chronic, relapsing nature of the disease. If you ask CRSwNP patients about the most important disease symptoms, they would say nasal congestion and decrease in smell. However, many CRSwNP patients would say that losing their sense of smell is particularly troublesome.
This study was conducted to better understand the effect of dupilumab, which is approved in the U.S. for adults with uncontrolled CRSwNP, on sense of smell.
Dr. Han[/caption]
Joseph Han, MD FARS, FAAOA FAAAAI
Medical Director for the Division of Allergy
Eastern Virginia Medical School
MedicalResearch.com: What is the background for this study?
Response: Chronic rhinosinusitis with nasal polyps (CRSwNP) is a type 2 inflammatory disease of the nasal passages and sinuses associated with a high symptom burden due to the chronic, relapsing nature of the disease. If you ask CRSwNP patients about the most important disease symptoms, they would say nasal congestion and decrease in smell. However, many CRSwNP patients would say that losing their sense of smell is particularly troublesome.
This study was conducted to better understand the effect of dupilumab, which is approved in the U.S. for adults with uncontrolled CRSwNP, on sense of smell.  Dr. Aquino[/caption]
Marcella Aquino, M.D.
Hasbro Children's Hospital
Department of Pediatrics
Division of Allergy & Immunology
Associate Professor of Pediatrics
[caption id="attachment_53397" align="alignleft" width="107"]
Dr. Aquino[/caption]
Marcella Aquino, M.D.
Hasbro Children's Hospital
Department of Pediatrics
Division of Allergy & Immunology
Associate Professor of Pediatrics
[caption id="attachment_53397" align="alignleft" width="107"] Dr. Koinis-Mitchell[/caption]
Daphne Koinis-Mitchell PhD
Professor (Research) in the Departments of Psychiatry and
Human Behavior and Pediatrics
Warren Alpert Medical School of Brown University
Providence, Rhode Island 02903
MedicalResearch.com: What is the background for this study?
Response: Urban minority children with asthma are at increased risk for sleep loss and poorer sleep quality secondary to socio-contextual stressors (poverty, stressors of urban living) and the underlying challenges related to following possibly complex asthma treatment regimens. Atopic dermatitis (AD) is very frequently seen in children with asthma and increases the risk for poor quality sleep, for example difficulty falling asleep, awakenings during the night, difficulty awakening in the morning, and/or daytime sleepiness.
Dr. Koinis-Mitchell[/caption]
Daphne Koinis-Mitchell PhD
Professor (Research) in the Departments of Psychiatry and
Human Behavior and Pediatrics
Warren Alpert Medical School of Brown University
Providence, Rhode Island 02903
MedicalResearch.com: What is the background for this study?
Response: Urban minority children with asthma are at increased risk for sleep loss and poorer sleep quality secondary to socio-contextual stressors (poverty, stressors of urban living) and the underlying challenges related to following possibly complex asthma treatment regimens. Atopic dermatitis (AD) is very frequently seen in children with asthma and increases the risk for poor quality sleep, for example difficulty falling asleep, awakenings during the night, difficulty awakening in the morning, and/or daytime sleepiness.  Dr. Zasłona and Dr. ONeill[/caption]
Zbigniew Zasłona PhD
Luke A. J. O’Neill PhD
Professor (Chair of Biochemistry)
School of Biochemistry and Immunology
Trinity Biomedical Sciences Institute
Trinity College Dublin, Dublin, Ireland
MedicalResearch.com: What is the background for this study?
Response: Asthma is the most common disease in childhood and the most common respiratory condition in Ireland. It is a disease of environmental and genetic components. It is important to point out that although Ireland has very good air quality, asthma prevalence is very high (the second highest in Europe), and although asthma is not a single gene disease (such as cystic fibrosis) it is very important to study genetic variations in Irish population.
Therefore in this study we put emphasis on the genetic component of asthma, rather than environmental factors, especially given that asthma heritability has been estimated as high as 60%. Prevention of asthma by reducing exposure to common risk factors, such as air pollution, will not stop the asthma epidemic in Ireland, as inferior air quality is not an issue.
Dr. Zasłona and Dr. ONeill[/caption]
Zbigniew Zasłona PhD
Luke A. J. O’Neill PhD
Professor (Chair of Biochemistry)
School of Biochemistry and Immunology
Trinity Biomedical Sciences Institute
Trinity College Dublin, Dublin, Ireland
MedicalResearch.com: What is the background for this study?
Response: Asthma is the most common disease in childhood and the most common respiratory condition in Ireland. It is a disease of environmental and genetic components. It is important to point out that although Ireland has very good air quality, asthma prevalence is very high (the second highest in Europe), and although asthma is not a single gene disease (such as cystic fibrosis) it is very important to study genetic variations in Irish population.
Therefore in this study we put emphasis on the genetic component of asthma, rather than environmental factors, especially given that asthma heritability has been estimated as high as 60%. Prevention of asthma by reducing exposure to common risk factors, such as air pollution, will not stop the asthma epidemic in Ireland, as inferior air quality is not an issue.
 Dr. Litonjua[/caption]
Augusto A. Litonjua, M.D., M.P.H.
Professor - Department of Pediatrics, Pulmonology
Interim Chief - Department of Medicine , Pulmonary Diseases and Critical Care
Professor - Department of Medicine , Pulmonary Diseases and Critical Care
University of Rochester
MedicalResearch.com: What is the background for this study?
Response: Vitamin D deficiency and insufficiency is prevalent worldwide. Prior observational studies have shown that low vitamin D levels have been associated with the development of asthma. Animal studies have reported that antenatal vitamin D is important for lung development in utero. Thus, we conducted a randomized, double-blind, controlled trial of vitamin D supplementation in pregnant women to see if we could prevent the development of asthma and wheezing illnesses in young children. The initial report of the trial results showed that children born to mothers in the vitamin D supplementation arm had lower risks for developing either asthma or recurrent wheezing episodes over the first 3 years, but this was not statistically significant (p=0.051)(Litonjua et al. JAMA 2016).
Dr. Litonjua[/caption]
Augusto A. Litonjua, M.D., M.P.H.
Professor - Department of Pediatrics, Pulmonology
Interim Chief - Department of Medicine , Pulmonary Diseases and Critical Care
Professor - Department of Medicine , Pulmonary Diseases and Critical Care
University of Rochester
MedicalResearch.com: What is the background for this study?
Response: Vitamin D deficiency and insufficiency is prevalent worldwide. Prior observational studies have shown that low vitamin D levels have been associated with the development of asthma. Animal studies have reported that antenatal vitamin D is important for lung development in utero. Thus, we conducted a randomized, double-blind, controlled trial of vitamin D supplementation in pregnant women to see if we could prevent the development of asthma and wheezing illnesses in young children. The initial report of the trial results showed that children born to mothers in the vitamin D supplementation arm had lower risks for developing either asthma or recurrent wheezing episodes over the first 3 years, but this was not statistically significant (p=0.051)(Litonjua et al. JAMA 2016). Dr. Chawes[/caption]
Bo Chawes, MD, PhD, DMSc
Associate Professor
Copenhagen Prospective Studies on Asthma in Childhood
Herlev and Gentofte Hospital
University of Copenhagen
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: There has been a global surge in vitamin D deficiency happening in parallel with an increase in prevalence of childhood asthma, which suggests that low maternal vitamin D levels during pregnancy may increase asthma risk in the child.
Due to that we conducted a randomized double-blinded controlled trial within the Danish COPSAC2010 cohort (www.copsac.com) of 7-fold (2,800 IU/d) vs. standard dose (400 IU/d) of vitamin D supplementation from pregnancy week 24 aiming to reduce offspring asthma risk. At age 3, we observed a non-significant 24% reduced risk of recurrent asthma-like symptoms, ie. recurrent wheeze, in the high-dose vitamin D group. In the current study, we followed 545 of the 581 children in the study till age 6, where an asthma diagnosis can be established and observed no effect of the high-dose vitamin D supplement on the child's risk of asthma.
Dr. Chawes[/caption]
Bo Chawes, MD, PhD, DMSc
Associate Professor
Copenhagen Prospective Studies on Asthma in Childhood
Herlev and Gentofte Hospital
University of Copenhagen
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: There has been a global surge in vitamin D deficiency happening in parallel with an increase in prevalence of childhood asthma, which suggests that low maternal vitamin D levels during pregnancy may increase asthma risk in the child.
Due to that we conducted a randomized double-blinded controlled trial within the Danish COPSAC2010 cohort (www.copsac.com) of 7-fold (2,800 IU/d) vs. standard dose (400 IU/d) of vitamin D supplementation from pregnancy week 24 aiming to reduce offspring asthma risk. At age 3, we observed a non-significant 24% reduced risk of recurrent asthma-like symptoms, ie. recurrent wheeze, in the high-dose vitamin D group. In the current study, we followed 545 of the 581 children in the study till age 6, where an asthma diagnosis can be established and observed no effect of the high-dose vitamin D supplement on the child's risk of asthma.
 Sean O'Quinn MPH
Director, Patient Reported Outcomes
AstraZeneca
MedicalResearch.com: What is the background for this study? How does benralizumab differ from traditional medications for asthma?
Response: FASENRA™ (benralizumab 30mg for subcutaneous injection as add-on maintenance therapy in severe eosinophilic asthma for patients 12 years and older) has a strong clinical profile, including powerful efficacy against exacerbations and the ability to improve lung function. Benralizumab is a respiratory biologic that binds directly to the IL-5α receptor on eosinophils and attracts natural killer cells to induce rapid and near-complete depletion of eosinophils via apoptosis. (NOTE: The mechanism of action of FASENRA in asthma has not been definitively established.) Benralizumab is not indicated for treatment of other eosinophilic conditions or for relief of acute bronchospasm or status asthmaticus. The most common adverse reactions include headache and pharyngitis.
Dependence on rescue medications is indicative of poor asthma control. In the Phase III SIROCCO/CALIMA trials, patients with severe eosinophilic asthma had significantly reduced exacerbation frequency and improved lung function when treated with benralizumab 30mg Q8W (first three doses Q4W) vs. placebo.
Less was known about the effects of benralizumab on rescue medication usage—specifically daily total rescue medication use, daytime and nighttime rescue medication use, and nighttime awakenings requiring rescue medication use. The aim of this analysis was to understand the potential treatment effects of benralizumab on these parameters.
Sean O'Quinn MPH
Director, Patient Reported Outcomes
AstraZeneca
MedicalResearch.com: What is the background for this study? How does benralizumab differ from traditional medications for asthma?
Response: FASENRA™ (benralizumab 30mg for subcutaneous injection as add-on maintenance therapy in severe eosinophilic asthma for patients 12 years and older) has a strong clinical profile, including powerful efficacy against exacerbations and the ability to improve lung function. Benralizumab is a respiratory biologic that binds directly to the IL-5α receptor on eosinophils and attracts natural killer cells to induce rapid and near-complete depletion of eosinophils via apoptosis. (NOTE: The mechanism of action of FASENRA in asthma has not been definitively established.) Benralizumab is not indicated for treatment of other eosinophilic conditions or for relief of acute bronchospasm or status asthmaticus. The most common adverse reactions include headache and pharyngitis.
Dependence on rescue medications is indicative of poor asthma control. In the Phase III SIROCCO/CALIMA trials, patients with severe eosinophilic asthma had significantly reduced exacerbation frequency and improved lung function when treated with benralizumab 30mg Q8W (first three doses Q4W) vs. placebo.
Less was known about the effects of benralizumab on rescue medication usage—specifically daily total rescue medication use, daytime and nighttime rescue medication use, and nighttime awakenings requiring rescue medication use. The aim of this analysis was to understand the potential treatment effects of benralizumab on these parameters. MedicalResearch.com Interview with: Bryce Hoffman, MD Allergy & Immunology Fellow National Jewish Health MedicalResearch.com: What is the background for this study? What are the...
 Catarina Almqvist Malmros MD, PhD
Professor | Consultant Pediatrician
Dept of Medical Epidemiology and Biostatistics | Karolinska Institutet
Lung and Allergy Unit | Astrid Lindgren Children’s Hospital
Stockholm, Sweden
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: We have previously shown an association between growing up with dogs and a lower risk of childhood asthma (doi: 10.1001/jamapediatrics.2015.3219) but it has been unknown whether this link is modified by characteristics of the dog. Sex of the dog may have an effect on expressed allergens, and uncastrated male dogs release more of a certain allergen than castrated male dogs and female dogs. Some breeds are also described as ‘hypoallergenic’, but there is no scientific evidence whether they are more suitable for people with allergies.
We examined how variables such as sex, breed, number of dogs or size of dog are associated with the risk of asthma and allergy among children with a dog in their home during the first year of life. We included all Swedish children born between January 2001 and December 2004 whose parents had a registered dog in a dog-owner register and linked the data to the Swedish population- and health data registers.
Main findings are that children raised with only female dogs at home had a 16 per cent lower risk of asthma than those with male dogs, and that children living with two or more dogs had a 21 per cent lower risk of asthma than those with only one dog. Importantly, families with parental asthma or allergies had ‘hypoallergenic’ breeds more often than children whose parents did not have asthma or allergies; 11.7% compared to 7.6 . Exposure to these breeds was associated with a 27 per cent higher risk of allergy and no decreased risk of asthma.
Catarina Almqvist Malmros MD, PhD
Professor | Consultant Pediatrician
Dept of Medical Epidemiology and Biostatistics | Karolinska Institutet
Lung and Allergy Unit | Astrid Lindgren Children’s Hospital
Stockholm, Sweden
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: We have previously shown an association between growing up with dogs and a lower risk of childhood asthma (doi: 10.1001/jamapediatrics.2015.3219) but it has been unknown whether this link is modified by characteristics of the dog. Sex of the dog may have an effect on expressed allergens, and uncastrated male dogs release more of a certain allergen than castrated male dogs and female dogs. Some breeds are also described as ‘hypoallergenic’, but there is no scientific evidence whether they are more suitable for people with allergies.
We examined how variables such as sex, breed, number of dogs or size of dog are associated with the risk of asthma and allergy among children with a dog in their home during the first year of life. We included all Swedish children born between January 2001 and December 2004 whose parents had a registered dog in a dog-owner register and linked the data to the Swedish population- and health data registers.
Main findings are that children raised with only female dogs at home had a 16 per cent lower risk of asthma than those with male dogs, and that children living with two or more dogs had a 21 per cent lower risk of asthma than those with only one dog. Importantly, families with parental asthma or allergies had ‘hypoallergenic’ breeds more often than children whose parents did not have asthma or allergies; 11.7% compared to 7.6 . Exposure to these breeds was associated with a 27 per cent higher risk of allergy and no decreased risk of asthma.  Neil Graham, M.B.B.S., M.D., M.P.H
VP of Immunology & Inflammation
Regeneron
MedicalResearch.com: What is the background for this announcement?
Response: Patients with moderate-to-severe asthma often have uncontrolled, persistent symptoms despite standard-of-care therapy that may make them suitable for treatment with a biologic therapy. They live with coughing, wheezing and difficulty breathing, and are at risk of severe asthma attacks that may require emergency room visits or hospitalizations. [i],[ii] Oral corticosteroids can provide relief for severe, short-term symptoms. However, their chronic use is limited to the most severe patients due to the potential for serious side effects. [iii],[iv]
A particular type of inflammation contributes to the cause of uncontrolled symptoms in multiple inflammatory diseases such as asthma and atopic dermatitis.[v] Dupixent is a medicine that inhibits the overactive signaling of interleukin-4 (IL-4) and interleukin-13 (IL-13), two key proteins that contribute to this type of inflammation. This inhibits cytokine-induced inflammatory responses, including the release of proinflammatory cytokines, chemokines, nitric oxide, and IgE; however, the mechanism of action of Dupixent in asthma has not been definitively established.
Neil Graham, M.B.B.S., M.D., M.P.H
VP of Immunology & Inflammation
Regeneron
MedicalResearch.com: What is the background for this announcement?
Response: Patients with moderate-to-severe asthma often have uncontrolled, persistent symptoms despite standard-of-care therapy that may make them suitable for treatment with a biologic therapy. They live with coughing, wheezing and difficulty breathing, and are at risk of severe asthma attacks that may require emergency room visits or hospitalizations. [i],[ii] Oral corticosteroids can provide relief for severe, short-term symptoms. However, their chronic use is limited to the most severe patients due to the potential for serious side effects. [iii],[iv]
A particular type of inflammation contributes to the cause of uncontrolled symptoms in multiple inflammatory diseases such as asthma and atopic dermatitis.[v] Dupixent is a medicine that inhibits the overactive signaling of interleukin-4 (IL-4) and interleukin-13 (IL-13), two key proteins that contribute to this type of inflammation. This inhibits cytokine-induced inflammatory responses, including the release of proinflammatory cytokines, chemokines, nitric oxide, and IgE; however, the mechanism of action of Dupixent in asthma has not been definitively established. Tosh Butt[/caption]
Tosh Butt, MBA
VP Respiratory
AstraZeneca
MedicalResearch.com: What is the background for this study? How is benralizumab different from more traditional treatments for asthma?
Tosh Butt[/caption]
Tosh Butt, MBA
VP Respiratory
AstraZeneca
MedicalResearch.com: What is the background for this study? How is benralizumab different from more traditional treatments for asthma?
 Aivaras Cepelis[/caption]
Aivaras Cepelis, MSci
Department of Public Health and Nursing, Faculty of Medicine and Health Science
NTNU, Norwegian University of Science and Technology
Trondheim, Norway
MedicalResearch.com: What is the background for this study?
Response: Atrial fibrillation is the most common sustained, irregular and often rapid heart rate with a lifetime risk of 26%. The number of adults with atrial fibrillation is projected to double by 2050. Atrial fibrillation is also linked to adverse cardiovascular outcomes such as doubled risk of stroke and cardiovascular mortality. Therefore, we believe that research into the novel risk factors of the disease is highly warranted.
One of the potential condition that could play a role in the growing prevalence of atrial fibrillation is asthma. Asthma is a chronic inflammatory airway disease, affecting as many as 30 million children and adults in Europe. High levels of systemic inflammation biomarkers have been reported in both uncontrolled asthmatics and patients with atrial fibrillation. Furthermore, beta-agonists, the most common prescribed asthma control medication, has been shown to influence heart rate and increase the risk of irregular heartbeat.
However, research looking at asthma and atrial fibrillation link are lacking and no previous studies have assessed the dose-response relationship between levels of asthma control and atrial fibrillation. We utilized over 54 000 adults from a large well-defined Norwegian population cohort The Nord-Trøndelag Health Study (HUNT) to explore this association.
Aivaras Cepelis[/caption]
Aivaras Cepelis, MSci
Department of Public Health and Nursing, Faculty of Medicine and Health Science
NTNU, Norwegian University of Science and Technology
Trondheim, Norway
MedicalResearch.com: What is the background for this study?
Response: Atrial fibrillation is the most common sustained, irregular and often rapid heart rate with a lifetime risk of 26%. The number of adults with atrial fibrillation is projected to double by 2050. Atrial fibrillation is also linked to adverse cardiovascular outcomes such as doubled risk of stroke and cardiovascular mortality. Therefore, we believe that research into the novel risk factors of the disease is highly warranted.
One of the potential condition that could play a role in the growing prevalence of atrial fibrillation is asthma. Asthma is a chronic inflammatory airway disease, affecting as many as 30 million children and adults in Europe. High levels of systemic inflammation biomarkers have been reported in both uncontrolled asthmatics and patients with atrial fibrillation. Furthermore, beta-agonists, the most common prescribed asthma control medication, has been shown to influence heart rate and increase the risk of irregular heartbeat.
However, research looking at asthma and atrial fibrillation link are lacking and no previous studies have assessed the dose-response relationship between levels of asthma control and atrial fibrillation. We utilized over 54 000 adults from a large well-defined Norwegian population cohort The Nord-Trøndelag Health Study (HUNT) to explore this association.
 Dr. Panettieri[/caption]
Reynold A. Panettieri, Jr., M.D.
Professor of Medicine, Robert Wood Johnson Medical School
Vice Chancellor, Clinical & Translational Science
Director, Rutgers Institute for Translational Medicine & Science
Emeritus Professor of Medicine, University of Pennsylvania
Child Health Institute of New Jersey
Rutgers, The State University of New Jersey
New Brunswick, NJ 08901
MedicalResearch.com: What is the background for this study?
Response: Severe asthma is characterized by Type 2 inflammation manifested by increases in IL-13, IL-4 and Il-5 levels in the airways that promotes airway hyperresponsiveness and in part irreversible airway obstruction. These clinical manifestations profoundly increase asthma morbidity and mortality.
To address an unmet therapeutic need, Tralokinumab was developed as a monoclonal antibody targeting soluble IL-13 with the goal of improving lung function and patient reported outcomes while decreasing annual exacerbation rates. Stratus 1 and 2 represent two identical randomized, double-blind, placebo-controlled, phase 3 clinical trials in severe asthma. These international trials enrolled approximately 2000 subjects with severe asthma and examined whether Tralokinumab decreased annualized exacerbation rates (AER) as compared with placebo (primary outcome).
Dr. Panettieri[/caption]
Reynold A. Panettieri, Jr., M.D.
Professor of Medicine, Robert Wood Johnson Medical School
Vice Chancellor, Clinical & Translational Science
Director, Rutgers Institute for Translational Medicine & Science
Emeritus Professor of Medicine, University of Pennsylvania
Child Health Institute of New Jersey
Rutgers, The State University of New Jersey
New Brunswick, NJ 08901
MedicalResearch.com: What is the background for this study?
Response: Severe asthma is characterized by Type 2 inflammation manifested by increases in IL-13, IL-4 and Il-5 levels in the airways that promotes airway hyperresponsiveness and in part irreversible airway obstruction. These clinical manifestations profoundly increase asthma morbidity and mortality.
To address an unmet therapeutic need, Tralokinumab was developed as a monoclonal antibody targeting soluble IL-13 with the goal of improving lung function and patient reported outcomes while decreasing annual exacerbation rates. Stratus 1 and 2 represent two identical randomized, double-blind, placebo-controlled, phase 3 clinical trials in severe asthma. These international trials enrolled approximately 2000 subjects with severe asthma and examined whether Tralokinumab decreased annualized exacerbation rates (AER) as compared with placebo (primary outcome).
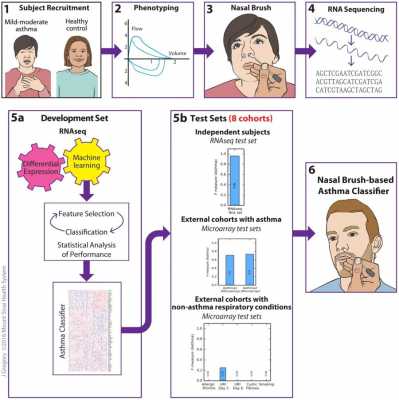 Asthma is a chronic respiratory disease that affects 10% of children and adults in the U.S. Mild to moderate asthma can be difficult to diagnose because symptoms change over time and can be complicated by other respiratory conditions. Given the high prevalence of asthma, there is high potential impact of improved diagnostic tools on reducing morbidity and mortality from asthma.
Current diagnostic tools for asthma, including spirometry and bronchoscopy, require specialized equipment and expertise to operate properly. Many individuals, particularly young children, have difficulty with pulmonary function testing because it requires, coordinated, forced breaths into a device. Spirometry results are unreliable when done with poor technique. Bronchoscopy is not practical for mild to moderate symptoms. For these reasons, asthma is often diagnosed and managed based on self-reporting of symptoms This can be unreliable, resulting in repeated doctor visits and even trips to the ER. Thus, a biomarker test for asthma that is easy to implement and interpret is highly desirable for the diagnosis and management of asthma.
Asthma is a chronic respiratory disease that affects 10% of children and adults in the U.S. Mild to moderate asthma can be difficult to diagnose because symptoms change over time and can be complicated by other respiratory conditions. Given the high prevalence of asthma, there is high potential impact of improved diagnostic tools on reducing morbidity and mortality from asthma.
Current diagnostic tools for asthma, including spirometry and bronchoscopy, require specialized equipment and expertise to operate properly. Many individuals, particularly young children, have difficulty with pulmonary function testing because it requires, coordinated, forced breaths into a device. Spirometry results are unreliable when done with poor technique. Bronchoscopy is not practical for mild to moderate symptoms. For these reasons, asthma is often diagnosed and managed based on self-reporting of symptoms This can be unreliable, resulting in repeated doctor visits and even trips to the ER. Thus, a biomarker test for asthma that is easy to implement and interpret is highly desirable for the diagnosis and management of asthma. Drr. Castro[/caption]
Mario Castro, M.D., M.P.H.
Alan A. and Edith L. Wolff Professor of Pulmonary and Critical Care Medicine,
Professor of Medicine, Pediatrics, and Radiology
Washington University School of Medicine
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: This is a confirmatory phase 3 pivotal study that assessed the efficacy and safety of dupilumab in a population of uncontrolled moderate to severe asthmatics.
This was the largest phase 3 placebo controlled trial conducted in this population evaluating a biologic. It enrolled patients without any minimum requirement for any type of biomarker such as blood eosinophils. It clearly confirmed the efficacy of dupilumab in reducing severe asthma exacerbations, improving lung function, asthma control and quality of life in the overall population. It also showed that patients with evidence of type 2 inflammation (increased blood eosinophils or exhaled NO) had a greater magnitude of effect.
Drr. Castro[/caption]
Mario Castro, M.D., M.P.H.
Alan A. and Edith L. Wolff Professor of Pulmonary and Critical Care Medicine,
Professor of Medicine, Pediatrics, and Radiology
Washington University School of Medicine
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: This is a confirmatory phase 3 pivotal study that assessed the efficacy and safety of dupilumab in a population of uncontrolled moderate to severe asthmatics.
This was the largest phase 3 placebo controlled trial conducted in this population evaluating a biologic. It enrolled patients without any minimum requirement for any type of biomarker such as blood eosinophils. It clearly confirmed the efficacy of dupilumab in reducing severe asthma exacerbations, improving lung function, asthma control and quality of life in the overall population. It also showed that patients with evidence of type 2 inflammation (increased blood eosinophils or exhaled NO) had a greater magnitude of effect.
