Laboratories, Pharmacology / 25.05.2026
LNP technology in drug delivery – how lipid nanoparticles became the backbone of modern RNA therapeutics
 For most of the history of RNA research, the molecule's therapeutic promise was undermined by a fundamental problem: RNA is unstable, immunogenic, and unable to cross cell membranes on its own. You could design a sequence that encoded exactly the right protein, synthesise it cleanly — and watch it degrade before reaching its target. The biology was understood; the delivery was the bottleneck.
Lipid nanoparticles solved that problem. Not all at once, and not simply — but the arc of LNP development over six decades produced the delivery technology that made RNA therapeutics clinically viable. The mRNA vaccines of 2020–2021 were the most visible expression of that achievement. They were not, however, the end of the story. They were the proof of concept that opened the pipeline.
For most of the history of RNA research, the molecule's therapeutic promise was undermined by a fundamental problem: RNA is unstable, immunogenic, and unable to cross cell membranes on its own. You could design a sequence that encoded exactly the right protein, synthesise it cleanly — and watch it degrade before reaching its target. The biology was understood; the delivery was the bottleneck.
Lipid nanoparticles solved that problem. Not all at once, and not simply — but the arc of LNP development over six decades produced the delivery technology that made RNA therapeutics clinically viable. The mRNA vaccines of 2020–2021 were the most visible expression of that achievement. They were not, however, the end of the story. They were the proof of concept that opened the pipeline.



 Dr. Tauscher-Wisniewski,[/caption]
Sitra Tauscher-Wisniewski, MD
Vice President Clinical Development & Analytics
Novartis Gene Therapies
MedicalResearch.com: What is the background for this study? Would you briefly describe the condition of Spinal muscular atrophy (SMA)?
Response: At the 2023 Muscular Dystrophy Association Conference, we presented new data from two of our Long-Term Follow-Up (LTFU) studies, LT001 and LT002, which show the continued efficacy and durability of Zolgensma across a range of patient populations, with an overall benefit-risk profile that remains favorable. LT001 is a 15-year ongoing observational LTFU study following the Phase 1 START patients, who were the very first patients to receive our gene replacement therapy. LT-002 is a voluntary Phase 4 15-year ongoing follow-up safety and efficacy study of Zolgensma IV and investigational intrathecal (IT) OAV101 in patients previously treated in the Phase 3 IV studies (STR1VE-US, STR1VE-EU, STR1VE-AP, SPR1NT) and the Phase 1 IT study (STRONG).
Spinal muscular atrophy (SMA) is a rare, devastating genetic disease that leads to progressive muscle weakness, paralysis, and when left untreated in one of its most severe forms (SMA Type 1), permanent ventilation or death in 90% of cases by age 2. It is caused by a lack of a functional survival motor neuron 1 (SMN1) gene, and in the most severe forms results in the rapid and irreversible loss of motor neurons, affecting muscle functions, including breathing, swallowing and basic movement.
Dr. Tauscher-Wisniewski,[/caption]
Sitra Tauscher-Wisniewski, MD
Vice President Clinical Development & Analytics
Novartis Gene Therapies
MedicalResearch.com: What is the background for this study? Would you briefly describe the condition of Spinal muscular atrophy (SMA)?
Response: At the 2023 Muscular Dystrophy Association Conference, we presented new data from two of our Long-Term Follow-Up (LTFU) studies, LT001 and LT002, which show the continued efficacy and durability of Zolgensma across a range of patient populations, with an overall benefit-risk profile that remains favorable. LT001 is a 15-year ongoing observational LTFU study following the Phase 1 START patients, who were the very first patients to receive our gene replacement therapy. LT-002 is a voluntary Phase 4 15-year ongoing follow-up safety and efficacy study of Zolgensma IV and investigational intrathecal (IT) OAV101 in patients previously treated in the Phase 3 IV studies (STR1VE-US, STR1VE-EU, STR1VE-AP, SPR1NT) and the Phase 1 IT study (STRONG).
Spinal muscular atrophy (SMA) is a rare, devastating genetic disease that leads to progressive muscle weakness, paralysis, and when left untreated in one of its most severe forms (SMA Type 1), permanent ventilation or death in 90% of cases by age 2. It is caused by a lack of a functional survival motor neuron 1 (SMN1) gene, and in the most severe forms results in the rapid and irreversible loss of motor neurons, affecting muscle functions, including breathing, swallowing and basic movement.
 Dr. Mapara[/caption]
Markus Y Mapara, MD
Professor of Medicine
Director of the Blood and Marrow Transplantation
Columbia University Medical Center
MedicalResearch.com: What is the background for this study? What are the main findings?
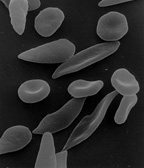
Response: Sickle cell disease is caused by a point mutation in the beta-globin gene of hemoglobin resulting in the production of abnormal hemoglobin which leads to formation of sickle-shaped RBC under conditions of low oxygen. Sickle cell disease affects about 100,000 patients in the US which are predominantly African American. The only curative approach is to perform an allogeneic bone marrow transplant which is however fraught with significant treatment-related risks if a matched sibling donor is not available.
The current study describes the successful application of a novel gene therapy to treat patients with sickle cell disease. The strategy is based on a gene-addition approach to introduce the genetic information for a Hemoglobin F-like molecule termed HgAT87Q into hematopoietic stem cells. The expression of this novel hemoglobin prevents polymerization of HgbS and has now been demonstrated to prevent the occurrence of vaso-occlusive pain crises in sickle cell disease patients.
Dr. Mapara[/caption]
Markus Y Mapara, MD
Professor of Medicine
Director of the Blood and Marrow Transplantation
Columbia University Medical Center
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Sickle cell disease is caused by a point mutation in the beta-globin gene of hemoglobin resulting in the production of abnormal hemoglobin which leads to formation of sickle-shaped RBC under conditions of low oxygen. Sickle cell disease affects about 100,000 patients in the US which are predominantly African American. The only curative approach is to perform an allogeneic bone marrow transplant which is however fraught with significant treatment-related risks if a matched sibling donor is not available.
The current study describes the successful application of a novel gene therapy to treat patients with sickle cell disease. The strategy is based on a gene-addition approach to introduce the genetic information for a Hemoglobin F-like molecule termed HgAT87Q into hematopoietic stem cells. The expression of this novel hemoglobin prevents polymerization of HgbS and has now been demonstrated to prevent the occurrence of vaso-occlusive pain crises in sickle cell disease patients.