
25 Feb Bladder and Colon Cancer: E. coli Bacteria Breakdown Protein Driver of Cancer Progression
MedicalResearch.com Interview with:

Catharina Svanborg M.D., Ph.D.
Professor at Lund University Department of Laboratory Medicine,
Division of Microbiology, Immunology and Glycobiology
Founder/Chairman of the Board at HAMLET Pharma
MedicalResearch.com: What is the background for this study?
 Response: Like many unexpected scientific developments, this finding was serendipitous. In our search for the molecular basis of host susceptibility to infection, we discovered that infection directly affects MYC levels.
Response: Like many unexpected scientific developments, this finding was serendipitous. In our search for the molecular basis of host susceptibility to infection, we discovered that infection directly affects MYC levels.
Gene expression analysis revealed that MYC itself was inhibited and that genes regulated by MYC were affected in children with acute kidney infection. Rapid reductions in MYC levels was further confirmed by infecting human kidney cells with the pathogenic E. coli bacteria isolated from patients with acute pyelonephritis, allowing us to formulate the hypothesis that bacteria regulate host MYC levels during acute infection and to investigate the mechanism leading to this inhibition. This work was conducted by the Laboratory Medicine group at Lund University in Sweden led by Professor Catharina Svanborg.
MedicalResearch.com: Would you explain the significance of the MYC protein?
Response: Cancer cells grow too fast, outcompete normal cells and spread to distant
sites, where they cause metastases. Understanding what makes cancer cells
so efficient and threatening is critically important and stopping them has
always been the goal of cancer research. Early studies identified so-called
‘’oncogenes’’; genes that that normally control cell growth but when mutated
are responsible for the creation of cancer cells and explain their competitive
advantages. This is why former President Nixon, when he declared the ”war
on cancer’’, intended for science to find the oncogenes and develop therapies
to prevent their destructive effects.
The transcription factor MYC has been named ‘’the quintessential oncogene’’
and is hyperactive in the majority of human cancers. Targeting MYC is
therefore highly desirable. MYC inhibition clearly arrests cancer progression
and restores tissue integrity in transgenic mouse models and a vast number
of technologies have been applied in attempts to target MYC and its different
co-factors in cancer cells. Still, finding c-MYC inhibitors for therapeutic use
has been problematic and MYC itself has long been viewed as “undruggable”.
MedicalResearch.com: What are the main findings?
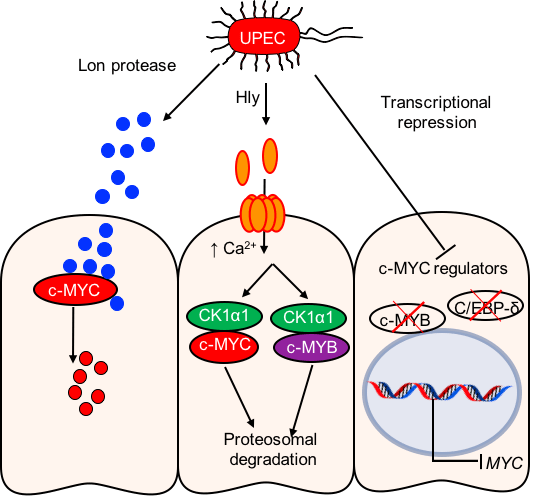
- Certain pathogenic bacteria degrade MYC and inhibit MYC expression.
- Several parallel mechanisms were identified.
- A specific protease released by the bacteria is taken up by human cells, where it binds to and degrades MYC.
- The purified protease has been used successful in treating different cancers in animal models.
- The protease was effective in murine models of bladder and colon cancer and was shown to reduce tumor progression and increase survival.
- Tissue levels of MYC returned to normal levels in treated mice, without evidence of adverse effects.
MedicalResearch.com: How does the E. coli molecule affect the MYC protein?
Response: The Lon protease is taken up by host cells and degrades the MYC protein. We believe that the specificity to MYC is due to the high serine content of the protein since the “natural” role of Lon protease in bacteria is to degrade short-lived serine-rich proteins.
MedicalResearch.com: What should readers take away from your report?
Response: If you make room for serendipity in science you can make some new discoveries. Targeting MYC is highly desirable for cancer therapy but has proven extremely difficult and MYC has long been regarded as ‘’undruggable’’. Our discovery identifies a new, promising molecular solution for this problem. The long-term goal is to develop a drug that can be used by cancer patients. Reaching that goal requires further studies regarding routes of administration, which cancers to target and analyses of toxicity.
MedicalResearch.com: What recommendations do you have for future research as a result of this work?
Response: This is a discovery and as such, it opens up for a lot of questions and approaches. We would love for this to become a new field with many participants. The therapeutic benefits of this approach should be explored as soon as possible developed for future clinical trials. The long-term goal is to provide more specific and less toxic therapies for cancer patients, A patent has been filed to protect the use of bacterial MYC inhibition therapy. The mechanism of MYC inhibition by the Lon protease is a novel observation and warrants further detailed investigation.
MedicalResearch.com: Is there anything else you would like to add?
Response: Our group at Lund University also discovered the tumoricidal complex HAMLET and has successfully developed this molecule for clinical trials. In the recent Phase I/II bladder cancer study in collaboration with Motol University Hospital Prague, we observed a rapid increase in cell shedding and accelerated cell death in tumors in patients receiving Alpha1H treatment, a second-generation derivative of the HAMLET molecule, with no evident of side effects. We are currently initiating a second phase of the clinical trial which aims to improve the the current treatment options by using higher doses of Alpha1H than that was used before, alone or in combination with chemotherapy drugs. We hope to develop a most efficient therapeutic approach that can maximise the benefits of patients in regards of safety and efficacy. Our experience and contacts will hopefully be beneficial across all the work we do.
Citation:
Butler, D.S.C., Cafaro, C., Putze, J. et al. A bacterial protease depletes c-MYC and increases survival in mouse models of bladder and colon cancer. Nat Biotechnol (2021). https://doi.org/10.1038/s41587-020-00805-3
[subscribe]
[last-modified]
The information on MedicalResearch.com is provided for educational purposes only, and is in no way intended to diagnose, cure, endorse or treat any medical or other condition. Always seek the advice of your physician or other qualified health and ask your doctor any questions you may have regarding a medical condition. In addition to all other limitations and disclaimers in this agreement, service provider and its third party providers disclaim any liability or loss in connection with the content provided on this website.
Last Updated on March 9, 2021 by Marie Benz MD FAAD
