
01 Jun Photoaged Skin Therapy with Adipose-Derived Stem Cells
MedicalResearch.com Interview with:

Dr. Charles-de-Sá
Charles-de-SáM.D., Ph.D.
Rio de Janeiro, Brazil
MedicalResearch.com: What is the background for this study?
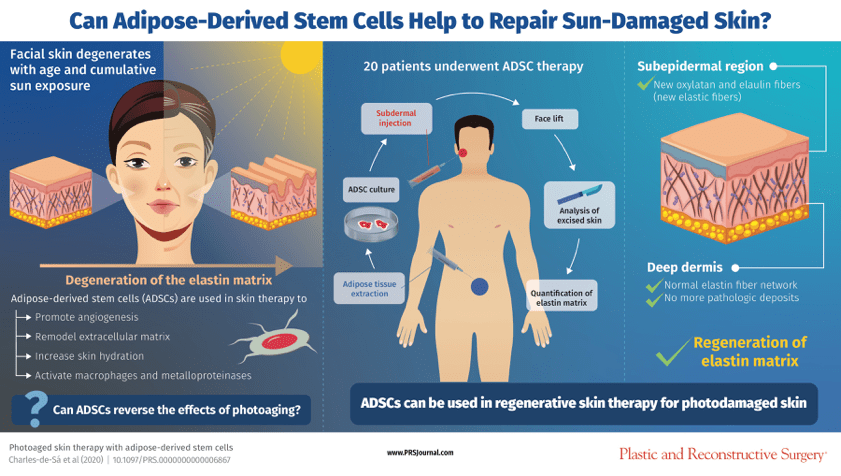
Response: Our clinical trial was based on our clinical skin observations in areas submitted to a lipotransfer previously, an ordinary practice in plastic surgery. These clinical observations lead us to investigate what will be the key element played in these findings. Our scientific support investigation addressed the Dardick1and Zuk, P2 studies, that demonstrated fibroblastic-like cells in adipose tissue with regenerative ability. Our clinical trial proposal is to investigate the adipose-derived stem cell (ADSC) role in the photoaged skin. The direct endpoint of the study was to assess the histological benefits provided by the subdermal ADSC injection. Mesenchymal stem cells were obtained from lipoaspirates, expanded in vitro, and introduced into the facial skin of 20 patients submitted after three to four months to a face-lifting surgery. In the retrieved skin, immunocytochemical and ultrastructural analysis quantified elastic matrix components, cathepsin-K, metalloprotease MMP-12, and the macrophage M2 markers: CD68, CD206 and heme-oxygenase-1.An overview of the trial steps is described in the infographic.
MedicalResearch.com: What are the main findings?
Response:A full de novo formation of oxytalan and elaunin fibers was observed in the subepidermal region, with a reconstitution of the papillary structure of the dermal-epidermal junction. Elastotic deposits in the deep dermis were substituted by a normal elastin fiber network. The coordinated removal of the pathologic deposits of ‘old’ elastic fibers and their substitution by the normal ones was concomitant with activation of cathepsin-K and MPP12, and with expansion of the M2 macrophage infiltration.
MedicalResearch.com: What should readers take away from your report?
Response: This study has demonstrated ADSC to remodeling the skin extra cellular matrix, mainly in the elastic system.
MedicalResearch.com: What recommendations do you have for future research as a result of this study?
Response: Based on these findings, the future of this research line aims to create new possibilities in regenerative cell therapy not only in skin diseases, but also in other clinical applicability in the case of organs and tissues with reduction and / or alteration in the elastic system (ex: aneurysms, cardiac valve disease and others), with a better understanding of the mechanisms involved and the control of these processes.
MedicalResearch.com: Is there anything else you would like to add? Any disclosures?
Response: It is interesting to be able, in future studies, to evaluate other mechanisms involved and the duration of effects regenerative effects on skin treated with ADSC. Another question could be considered: optimized ADSC (quantity) / area with the tissue effect found. We have not any to disclosure. This study was developed by federal university of Rio de Janeiro-Brasil and Verona University-Italy
Citation:
Charles-de-Sá, Luiz M.D., Ph.D.; Gontijo-de-Amorim, Natale Ferreira M.D., Ph.D.; Rigotti, Gino M.D., Ph.D.; Sbarbati, Andrea M.D., Ph.D.; Bernardi, Paolo Ph.D.; Benati, Donatella Ph.D.; Bizon Vieira Carias, Rosana Ph.D.; Maeda Takiya, Christina M.D., Ph.D.; Borojevic, Radovan Ph.D. Photoaged Skin Therapy with Adipose-Derived Stem Cells, Plastic and Reconstructive Surgery: June 2020 – Volume 145 – Issue 6 – p 1037e-1049e doi: 10.1097/PRS.0000000000006867
References:
- Dardick I, Polansky, WJ. Ultrastructural Observations on Differentiating Human Preadipocytes Cultured in Vitro. Tissue & Cell.1976.561-571
- Zuk Pa,Zhu M,Et Al. Multilineage Cells From Human Adipose Tissue: Implications For Cell-Based Therapies. Tissue Eng 2001; 7: 211-28
The information on MedicalResearch.com is provided for educational purposes only, and is in no way intended to diagnose, cure, or treat any medical or other condition. Always seek the advice of your physician or other qualified health and ask your doctor any questions you may have regarding a medical condition. In addition to all other limitations and disclaimers in this agreement, service provider and its third party providers disclaim any liability or loss in connection with the content provided on this website.
Last Updated on June 1, 2020 by Marie Benz MD FAAD
