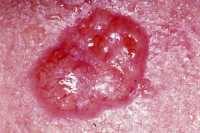
15 Dec Surgery Versus 5% Imiquimod for Nodular and Superficial Basal Cell Carcinoma
MedicalResearch.com Interview with:
Hywel C. Williams DSc, FMedSci, NIHR Senior Investigator
Director of the NIHR Health Technology Assessment Programme
http://www.nets.nihr.ac.uk/programmes/hta
Professor of Dermato-Epidemiology and Co-Director of the Centre of Evidence-Based Dermatology,
http://www.nottingham.ac.uk/research/groups/cebd/index.aspx
University of Nottingham,
Queen’s Medical Centre,
Nottingham University Hospitals NHS Trust,
Nottingham UK
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Our clinical trial of 5% imiquimod cream versus surgery for low risk basal carcinoma (BCC) of the skin was first prompted by a shocking lack of randomised controlled clinical trials for what is the commonest form of human cancer.
We had conducted a Cochrane systematic review prior to starting the study and found very few long term studies. An emerging literature on imiquimod cream at the time suggested that it might have a clinically useful effect for low risk BCC. All the studies were short term and industry supported, so with the support of Cancer Research UK (UK largest cancer charity), we undertook a large independent study to see how the cream compared to the reference standard of excision surgery with a 4mm margin for low risk superficial and nodular BCC.
Our three year results, published in Lancet Oncology, showed that surgery is clearly superior to imiquimod cream, with a success rate (absence of initial failure and no signs of recurrence at 3 years) of 98.4% compared to 83.6% for imiquimod. Nevertheless, the 83.6% success rate is still potentially useful, so we wanted to see whether these 3 year results were sustained. So we followed up your study participants for a total of 5 years and found that the response rates at 5 years were almost the same as those at 3 years (97.7% and 82.5% for surgery and imiquimod respectively).
Most treatment failures with imiquimod occurred early on ie in the first year of treatment. Our study shows that if initial treatment works, the benefits are sustained.
MedicalResearch.com: What should readers take away from your report?
Response: That surgery offers the best chance of treatment success for BCC. But, if a person did not want to have surgery, then imiquimod cream offers a reasonable response rate for low risk BCC. And if it fails, the surgery can always be done later. At least we now have reasonable precise and independent estimates of the benefits and possible harms of both forms of treatment that can be used in a shared decision making discussion between patient and doctor.
MedicalResearch.com: What recommendations do you have for future research as a result of this study?
Response: Given that Basal Cell Skin Cancer is increasing all the time due to an ageing fair skin population with lots of previous sun exposure, one strategy that could be tested in a future pragmatic study is to see whether a policy of treating all low risk BCC in the community with topical imiquimod (followed by referral to secondary care for treatment failure or recurrence) would be cost effective and acceptable to patients as compared to the current pathway of referring nearly all people with suspected basal cell skin cancer to dermatologists who are struggling to cope with the demand – at least in the UK. We hope that the modest success rates of topical imiquimod will stimulate the development of other more potent topical preparations that can boost the body’s immune system to get rid of basal cell skin cancer.
MedicalResearch.com: Is there anything else you would like to add?
Response: Only to emphasise that this research, as all our research conducted at the Centre of Evidence-Based Dermatology, is completely independent of any for profit organisations.
MedicalResearch.com: Thank you for your contribution to the MedicalResearch.com community.
Citation:
Surgery Versus 5% Imiquimod for Nodular and Superficial Basal Cell Carcinoma: 5-Year Results of the SINS Randomized Controlled Trial Hywel C. Williams1 , Fiona Bath-Hextall1,2 , Mara Ozolins1,3 , Sarah J. Armstrong4 , Graham B. Colver5 , William Perkins6 and Paul S.J. Miller4 , on behalf of the Surgery Versus Imiquimod for Nodular and Superficial Basal Cell Carcinoma (SINS) Study Group
Published online December 05, 2016
DOI: http://dx.doi.org/10.1016/j.jid.2016.10.019
Note: Content is Not intended as medical advice. Please consult your health care provider regarding your specific medical condition and questions.
More Medical Research Interviews on MedicalResearch.com
[wysija_form id=”5″]
Last Updated on December 15, 2016 by Marie Benz MD FAAD