Author Interviews, CDC, Infections / 20.05.2024
NIH Study Suggests Prion Disease Unlikely to Spread from Deer to Humans
MedicalResearch.com Interview with:
[caption id="attachment_61743" align="alignleft" width="150"] Dr. Haigh[/caption]
Cathryn Haigh, Ph.D.
Chief Prion Cell Biology Unit
Laboratory of Neurological Infections and Immunity
National Institute of Allergy and Infectious Diseases
Division of Intramural Research, Rocky Mountain Laboratories
National Institutes of Health
Hamilton, MT 59840
MedicalResearch.com: What is the background for this study, ie what are prions/prion-related diseases? Where are prions found?
Response: Prion diseases are infectious neurodegenerative diseases of humans and animals. In humans these diseases often manifest as rapidly progressing dementias but are rarely caused by a known exposure to the infectious agents (prions). More commonly they are sporadic (no known cause) or hereditary.
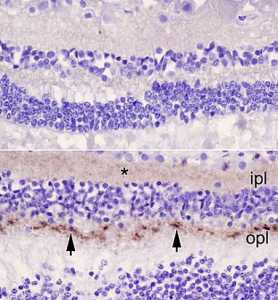
One form of human disease is believed to have arisen from eating beef contaminated with bovine spongiform encephalopathy (as known as mad cow disease). This has resulted in concerns that chronic wasting disease (CWD), a prion disease affecting deer, elk and moose, might also have the potential to cross the species barrier and cause disease in humans. To date, transmissions of CWD prions to cynomolgus macaques have been negative, a good sign that crossing the species barrier would not be easy, but macaques are not human so we wanted to test whether CWD could infect human brain tissue.
To do this we used a human cerebral organoid model (mini human brain tissues grown from skin cells in a laboratory) and directly exposed the organoids to prions from the brains of animals that had died of CWD.
Dr. Haigh[/caption]
Cathryn Haigh, Ph.D.
Chief Prion Cell Biology Unit
Laboratory of Neurological Infections and Immunity
National Institute of Allergy and Infectious Diseases
Division of Intramural Research, Rocky Mountain Laboratories
National Institutes of Health
Hamilton, MT 59840
MedicalResearch.com: What is the background for this study, ie what are prions/prion-related diseases? Where are prions found?
Response: Prion diseases are infectious neurodegenerative diseases of humans and animals. In humans these diseases often manifest as rapidly progressing dementias but are rarely caused by a known exposure to the infectious agents (prions). More commonly they are sporadic (no known cause) or hereditary.
One form of human disease is believed to have arisen from eating beef contaminated with bovine spongiform encephalopathy (as known as mad cow disease). This has resulted in concerns that chronic wasting disease (CWD), a prion disease affecting deer, elk and moose, might also have the potential to cross the species barrier and cause disease in humans. To date, transmissions of CWD prions to cynomolgus macaques have been negative, a good sign that crossing the species barrier would not be easy, but macaques are not human so we wanted to test whether CWD could infect human brain tissue.
To do this we used a human cerebral organoid model (mini human brain tissues grown from skin cells in a laboratory) and directly exposed the organoids to prions from the brains of animals that had died of CWD.
 Dr. Haigh[/caption]
Cathryn Haigh, Ph.D.
Chief Prion Cell Biology Unit
Laboratory of Neurological Infections and Immunity
National Institute of Allergy and Infectious Diseases
Division of Intramural Research, Rocky Mountain Laboratories
National Institutes of Health
Hamilton, MT 59840
MedicalResearch.com: What is the background for this study, ie what are prions/prion-related diseases? Where are prions found?
Response: Prion diseases are infectious neurodegenerative diseases of humans and animals. In humans these diseases often manifest as rapidly progressing dementias but are rarely caused by a known exposure to the infectious agents (prions). More commonly they are sporadic (no known cause) or hereditary.
One form of human disease is believed to have arisen from eating beef contaminated with bovine spongiform encephalopathy (as known as mad cow disease). This has resulted in concerns that chronic wasting disease (CWD), a prion disease affecting deer, elk and moose, might also have the potential to cross the species barrier and cause disease in humans. To date, transmissions of CWD prions to cynomolgus macaques have been negative, a good sign that crossing the species barrier would not be easy, but macaques are not human so we wanted to test whether CWD could infect human brain tissue.
To do this we used a human cerebral organoid model (mini human brain tissues grown from skin cells in a laboratory) and directly exposed the organoids to prions from the brains of animals that had died of CWD.
Dr. Haigh[/caption]
Cathryn Haigh, Ph.D.
Chief Prion Cell Biology Unit
Laboratory of Neurological Infections and Immunity
National Institute of Allergy and Infectious Diseases
Division of Intramural Research, Rocky Mountain Laboratories
National Institutes of Health
Hamilton, MT 59840
MedicalResearch.com: What is the background for this study, ie what are prions/prion-related diseases? Where are prions found?
Response: Prion diseases are infectious neurodegenerative diseases of humans and animals. In humans these diseases often manifest as rapidly progressing dementias but are rarely caused by a known exposure to the infectious agents (prions). More commonly they are sporadic (no known cause) or hereditary.
One form of human disease is believed to have arisen from eating beef contaminated with bovine spongiform encephalopathy (as known as mad cow disease). This has resulted in concerns that chronic wasting disease (CWD), a prion disease affecting deer, elk and moose, might also have the potential to cross the species barrier and cause disease in humans. To date, transmissions of CWD prions to cynomolgus macaques have been negative, a good sign that crossing the species barrier would not be easy, but macaques are not human so we wanted to test whether CWD could infect human brain tissue.
To do this we used a human cerebral organoid model (mini human brain tissues grown from skin cells in a laboratory) and directly exposed the organoids to prions from the brains of animals that had died of CWD.