Allergies, Author Interviews, Dermatology / 06.12.2025
ACAAI: Remibrutinib As Oral option for Patients with Chronic Spontaneous Urticaria who have failed high dose antihistamines
[caption id="attachment_71677" align="alignleft" width="200"] Dr. Gogate[/caption]
MedicalResearch.com Interview with:
Dr. Shaila Gogate, MD
Colorado Allergy & Asthma Centers
Allergist
REMIX-1/-2 Investigator
MedicalResearch.com: What is the background for this study? What are the main findings?
A: The Phase III REMIX-1/-2 studies were randomized, double-blind, placebo-controlled studies assessing the efficacy and safety of oral remibrutinib 25 mg twice daily or placebo over a 24-week double-blind period, followed by a 28-week open-label remibrutinib treatment period.
The purpose of this pooled analysis was to examine the early and long-term efficacy data of remibrutinib vs. placebo over time, looking at the mean percentage change from baseline (CFB) in weekly scores of disease activity (UAS7), itch severity (ISS7) and hives severity (HSS7) over 52 weeks. This analysis demonstrated that remibrutinib showed reductions in urticaria symptoms vs. placebo as early as week 1:
Dr. Gogate[/caption]
MedicalResearch.com Interview with:
Dr. Shaila Gogate, MD
Colorado Allergy & Asthma Centers
Allergist
REMIX-1/-2 Investigator
MedicalResearch.com: What is the background for this study? What are the main findings?
A: The Phase III REMIX-1/-2 studies were randomized, double-blind, placebo-controlled studies assessing the efficacy and safety of oral remibrutinib 25 mg twice daily or placebo over a 24-week double-blind period, followed by a 28-week open-label remibrutinib treatment period.
The purpose of this pooled analysis was to examine the early and long-term efficacy data of remibrutinib vs. placebo over time, looking at the mean percentage change from baseline (CFB) in weekly scores of disease activity (UAS7), itch severity (ISS7) and hives severity (HSS7) over 52 weeks. This analysis demonstrated that remibrutinib showed reductions in urticaria symptoms vs. placebo as early as week 1:
 Dr. Gogate[/caption]
MedicalResearch.com Interview with:
Dr. Shaila Gogate, MD
Colorado Allergy & Asthma Centers
Allergist
REMIX-1/-2 Investigator
MedicalResearch.com: What is the background for this study? What are the main findings?
A: The Phase III REMIX-1/-2 studies were randomized, double-blind, placebo-controlled studies assessing the efficacy and safety of oral remibrutinib 25 mg twice daily or placebo over a 24-week double-blind period, followed by a 28-week open-label remibrutinib treatment period.
The purpose of this pooled analysis was to examine the early and long-term efficacy data of remibrutinib vs. placebo over time, looking at the mean percentage change from baseline (CFB) in weekly scores of disease activity (UAS7), itch severity (ISS7) and hives severity (HSS7) over 52 weeks. This analysis demonstrated that remibrutinib showed reductions in urticaria symptoms vs. placebo as early as week 1:
Dr. Gogate[/caption]
MedicalResearch.com Interview with:
Dr. Shaila Gogate, MD
Colorado Allergy & Asthma Centers
Allergist
REMIX-1/-2 Investigator
MedicalResearch.com: What is the background for this study? What are the main findings?
A: The Phase III REMIX-1/-2 studies were randomized, double-blind, placebo-controlled studies assessing the efficacy and safety of oral remibrutinib 25 mg twice daily or placebo over a 24-week double-blind period, followed by a 28-week open-label remibrutinib treatment period.
The purpose of this pooled analysis was to examine the early and long-term efficacy data of remibrutinib vs. placebo over time, looking at the mean percentage change from baseline (CFB) in weekly scores of disease activity (UAS7), itch severity (ISS7) and hives severity (HSS7) over 52 weeks. This analysis demonstrated that remibrutinib showed reductions in urticaria symptoms vs. placebo as early as week 1:
-
- 38.4% vs. 10.3% in UAS7
- 37.0% vs. 9.8% in ISS7
- 39.8% vs. 10.1% HSS7 Greater reductions vs. placebo were seen over 24 weeks and results were observed in the open-label period through week 52 in all remibrutinib-treated patients (including those transitioned from placebo).


 Dr. Mosnaim[/caption]
Dr. Giselle Mosnaim MD MS FACAAI
Allergist
Division of Allergy and Immunology
Department of Medicine
Northshore University Health System
REMIX-1/-2 Investigator
MedicalResearch.com: What is the background for this study? What are the main findings?
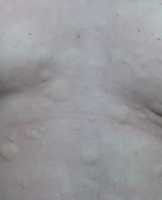
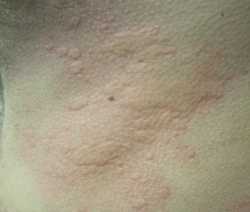
Response: Chronic spontaneous urticaria is a debilitating condition significantly affecting patient quality of life and is characterized by the presence of itch, hives, and/or angioedema (swelling) lasting for greater than 6 weeks, without identifiable trigger. Second-generation H1-antihistamines at standard doses are recommended as first-line treatment for patients with chronic spontaneous urticaria, and can be up-dosed to 4 times the standard dose. However, over 50% of patients continue to have symptoms despite this therapy. Both Remibrutinib and Dupilumab are FDA-approved for the treatment of chronic spontaneous urticaria in adult patients (Remibrutinib ≥ 18 years old and Dupilumab ≥ 12 years old) who remain symptomatic despite second-generation H1-antihistamine treatment.
The RECLAIM study is an ongoing head-to-head randomized controlled trial comparing the efficacy of Remibrutinib, an oral Bruton’s tyrosine kinase inhibitor, and Dupilumab, an injectable anti-interleukin-4 and anti-interleukin-13 inhibitor, at early timepoints (4 weeks and earlier) when administered as an add-on therapy in adults with moderate to severe chronic spontaneous urticaria inadequately controlled by second-generation H1-antihistamines.
Dr. Mosnaim[/caption]
Dr. Giselle Mosnaim MD MS FACAAI
Allergist
Division of Allergy and Immunology
Department of Medicine
Northshore University Health System
REMIX-1/-2 Investigator
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Chronic spontaneous urticaria is a debilitating condition significantly affecting patient quality of life and is characterized by the presence of itch, hives, and/or angioedema (swelling) lasting for greater than 6 weeks, without identifiable trigger. Second-generation H1-antihistamines at standard doses are recommended as first-line treatment for patients with chronic spontaneous urticaria, and can be up-dosed to 4 times the standard dose. However, over 50% of patients continue to have symptoms despite this therapy. Both Remibrutinib and Dupilumab are FDA-approved for the treatment of chronic spontaneous urticaria in adult patients (Remibrutinib ≥ 18 years old and Dupilumab ≥ 12 years old) who remain symptomatic despite second-generation H1-antihistamine treatment.
The RECLAIM study is an ongoing head-to-head randomized controlled trial comparing the efficacy of Remibrutinib, an oral Bruton’s tyrosine kinase inhibitor, and Dupilumab, an injectable anti-interleukin-4 and anti-interleukin-13 inhibitor, at early timepoints (4 weeks and earlier) when administered as an add-on therapy in adults with moderate to severe chronic spontaneous urticaria inadequately controlled by second-generation H1-antihistamines.