29 Jul NEJM Study Compares Treatments for Uterine Fibroids
MedicalResearch.com Interview with:
Jane Daniels PhD
Professor of Clinical Trials, Faculty of Medicine & Health Sciences
School of Medicine
University of Nottingham
MedicalResearch.com: What is the background for this study? MedicalResearch.com: What are the main findings?

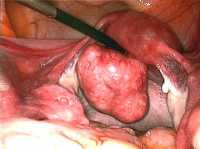
One example of a large fibroid-Wikipedia image
Response: Uterine fibroids are the most common non-cancerous tumour in women of childbearing age. They are associated with heavy bleeding, lower chances of having children and reduced quality of life. Traditional surgical options were either to remove the fibroids (myomectomy) or completely remove the womb. A newer approach, known as uterine artery embolization, involves blocking the blood supply to the fibroids in the womb.
Fibroids may be associated with infertility and problems during pregnancy, including miscarriage and preterm birth. As more women are having children at a later age, fibroids are becoming more of an issue for them and safe and effective fertility sparing treatments are needed.
Both treatments improve quality of life, but myomectomy will provide greater benefit to women on average.
MedicalResearch.com: What should readers take away from your report?
Response: This study is the largest to date comparing women treated with either myomectomy or uterine-artery embolization for their fibroids. It was completed in hospitals across the UK, and included a substantial number of women of African-Caribbean ethnicity, who are more likely have bothersome fibroids, so we can be confident that the findings are important and relevant.
Women, including those desiring a future pregnancy, should be provided with the evidence generated by the FEMME trial to enable to make a fully informed decision regarding their fibroid treatment.
MedicalResearch.com: What recommendations do you have for future research as a result of this work?
Response: There is no evidence to suggest either treatment is unsuitable for women wanting to get pregnant, but more research is needed in younger women.
No disclosures.
This study/project is funded by the National Institute for Health Research (NIHR) [Health Technology Assessment Programme (project reference 08/53/22)]. The views expressed are those of the author(s) and not necessarily those of the NIHR or the Department of Health and Social Care.
Citation:
Uterine-Artery Embolization or Myomectomy for Uterine Fibroids
Isaac Manyonda, Ph.D., Anna-Maria Belli, F.R.C.R., Mary-Ann Lumsden, M.D., Jonathan Moss, M.B., Ch.B., William McKinnon, Ph.D., Lee J. Middleton, M.Sc., Versha Cheed, M.Sc., Olivia Wu, Ph.D., Fusun Sirkeci, M.D., Jane P. Daniels, Ph.D., and Klim McPherson, Ph.D. for the FEMME Collaborative Group
July 30, 2020
N Engl J Med 2020; 383:440-451
DOI: 10.1056/NEJMoa1914735
[subscribe]
[last-modified]
The information on MedicalResearch.com is provided for educational purposes only, and is in no way intended to diagnose, cure, or treat any medical or other condition. Always seek the advice of your physician or other qualified health and ask your doctor any questions you may have regarding a medical condition. In addition to all other limitations and disclaimers in this agreement, service provider and its third party providers disclaim any liability or loss in connection with the content provided on this website.
Last Updated on July 29, 2020 by Marie Benz MD FAAD
