AI and HealthCare, Author Interviews, Cannabis, Pharmacology, Technology / 28.08.2025
When the Math Doesn’t Add Up, Can AI Do the First Pass to Improve Biomedical Research?
[caption id="attachment_70470" align="alignleft" width="150"] Dr. Dobbins, PharmD[/caption]
MedicalResearch.com Interview with:
Duncan Dobbins, PharmD, MHI
Geisinger College of Health Sciences
Scranton, Pennsylvania
MedicalResearch.com: What prompted this commentary, and what did you find?
Response: In theory, there could be a drug interaction between immunotherapy and medical cannabis. A small (N=102) observational report from Israel appeared to find that immunotherapies worked much less well in cancer patients who also used medical cannabis.1 However, a follow up report2 took about two weeks and involved manually rechecking the math and data-analysis. Several discrepancies emerged between the methods and results. Two-tailed tests were listed in the methods yet one-tailed p values appeared in the results. Arithmetic errors, some traceable to unconventional “floor” rounding, affected key percentages. Multiple p values in Table 1 (21 out of 22) could not be reproduced with the stated tests. Finally, smoking status, a key confound, was not reported. Taken together, these issues complicate interpretation and highlight how small computational slips can cascade into larger inferential uncertainty. For this follow-up report, I was asked, “Do you think AI could have double checked this math?”
Dr. Dobbins, PharmD[/caption]
MedicalResearch.com Interview with:
Duncan Dobbins, PharmD, MHI
Geisinger College of Health Sciences
Scranton, Pennsylvania
MedicalResearch.com: What prompted this commentary, and what did you find?
Response: In theory, there could be a drug interaction between immunotherapy and medical cannabis. A small (N=102) observational report from Israel appeared to find that immunotherapies worked much less well in cancer patients who also used medical cannabis.1 However, a follow up report2 took about two weeks and involved manually rechecking the math and data-analysis. Several discrepancies emerged between the methods and results. Two-tailed tests were listed in the methods yet one-tailed p values appeared in the results. Arithmetic errors, some traceable to unconventional “floor” rounding, affected key percentages. Multiple p values in Table 1 (21 out of 22) could not be reproduced with the stated tests. Finally, smoking status, a key confound, was not reported. Taken together, these issues complicate interpretation and highlight how small computational slips can cascade into larger inferential uncertainty. For this follow-up report, I was asked, “Do you think AI could have double checked this math?”
 Dr. Dobbins, PharmD[/caption]
MedicalResearch.com Interview with:
Duncan Dobbins, PharmD, MHI
Geisinger College of Health Sciences
Scranton, Pennsylvania
MedicalResearch.com: What prompted this commentary, and what did you find?
Response: In theory, there could be a drug interaction between immunotherapy and medical cannabis. A small (N=102) observational report from Israel appeared to find that immunotherapies worked much less well in cancer patients who also used medical cannabis.1 However, a follow up report2 took about two weeks and involved manually rechecking the math and data-analysis. Several discrepancies emerged between the methods and results. Two-tailed tests were listed in the methods yet one-tailed p values appeared in the results. Arithmetic errors, some traceable to unconventional “floor” rounding, affected key percentages. Multiple p values in Table 1 (21 out of 22) could not be reproduced with the stated tests. Finally, smoking status, a key confound, was not reported. Taken together, these issues complicate interpretation and highlight how small computational slips can cascade into larger inferential uncertainty. For this follow-up report, I was asked, “Do you think AI could have double checked this math?”
Dr. Dobbins, PharmD[/caption]
MedicalResearch.com Interview with:
Duncan Dobbins, PharmD, MHI
Geisinger College of Health Sciences
Scranton, Pennsylvania
MedicalResearch.com: What prompted this commentary, and what did you find?
Response: In theory, there could be a drug interaction between immunotherapy and medical cannabis. A small (N=102) observational report from Israel appeared to find that immunotherapies worked much less well in cancer patients who also used medical cannabis.1 However, a follow up report2 took about two weeks and involved manually rechecking the math and data-analysis. Several discrepancies emerged between the methods and results. Two-tailed tests were listed in the methods yet one-tailed p values appeared in the results. Arithmetic errors, some traceable to unconventional “floor” rounding, affected key percentages. Multiple p values in Table 1 (21 out of 22) could not be reproduced with the stated tests. Finally, smoking status, a key confound, was not reported. Taken together, these issues complicate interpretation and highlight how small computational slips can cascade into larger inferential uncertainty. For this follow-up report, I was asked, “Do you think AI could have double checked this math?”

 Elena Stains[/caption]
Elena Stains
Medical Student
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
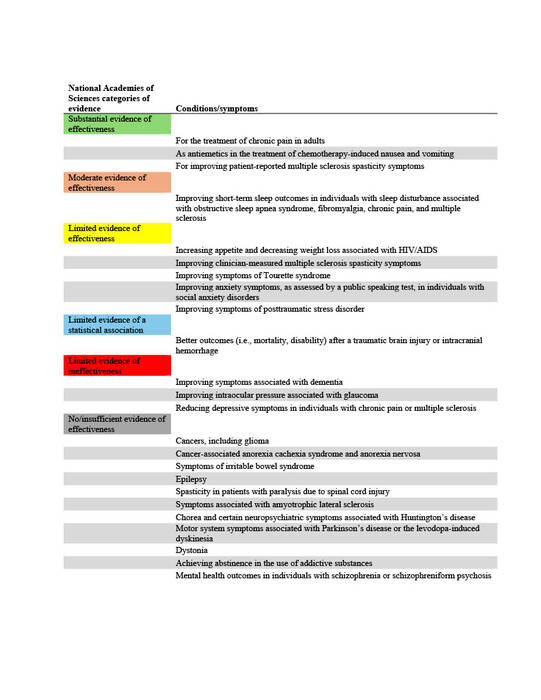
Response: In 2019 to 2020, 2.5% of Americans reported using cannabis for medical needs, compared to 1.2% in 2013-2014, representing a 12.9% annual increase1. Forty states and the District of Columbia have legislation for some form of medical cannabis (MC) in 2024. Because MC is not federally legalized, each state creates its own legislation on the conditions that qualify a person for MC, without any standardized process to determine what qualifying conditions (QC) are proven to be aided by MC. Thus, the QCs chosen by states vary widely. Common QCs include cancer, dementia, and PTSD.
The National Academies of Sciences, Engineering, and Medicine (NAS) published a report in 2017 on the evidence for the therapeutic effects of cannabis and cannabinoids for over twenty conditions2. This report reviews the evidence of effectiveness of medical cannabis for the most common QCs chosen by states. The researchers at Geisinger Commonwealth School of Medicine aimed to compare the evidence found by the NAS report with the QCs of 38 states (including the District of Columbia) in both 2017 and 2024. QCs were categorized based on NAS-established level of evidence: limited, moderate, or substantial/conclusive evidence of effectiveness, limited evidence of ineffectiveness, or no/insufficient evidence to support or refute effectiveness (Table 1).
Elena Stains[/caption]
Elena Stains
Medical Student
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: In 2019 to 2020, 2.5% of Americans reported using cannabis for medical needs, compared to 1.2% in 2013-2014, representing a 12.9% annual increase1. Forty states and the District of Columbia have legislation for some form of medical cannabis (MC) in 2024. Because MC is not federally legalized, each state creates its own legislation on the conditions that qualify a person for MC, without any standardized process to determine what qualifying conditions (QC) are proven to be aided by MC. Thus, the QCs chosen by states vary widely. Common QCs include cancer, dementia, and PTSD.
The National Academies of Sciences, Engineering, and Medicine (NAS) published a report in 2017 on the evidence for the therapeutic effects of cannabis and cannabinoids for over twenty conditions2. This report reviews the evidence of effectiveness of medical cannabis for the most common QCs chosen by states. The researchers at Geisinger Commonwealth School of Medicine aimed to compare the evidence found by the NAS report with the QCs of 38 states (including the District of Columbia) in both 2017 and 2024. QCs were categorized based on NAS-established level of evidence: limited, moderate, or substantial/conclusive evidence of effectiveness, limited evidence of ineffectiveness, or no/insufficient evidence to support or refute effectiveness (Table 1).






 Lauren C. Davis, MBS
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA 19409
MedicalResearch.com: What is the background for this study?
Response: Financial conflicts of interest (COIs) resulting from ties between academia and industry have been under scrutiny for their potential to hinder the integrity of medical research. COIs can lead to implicit bias, compromise the research process, and erode public trust (1-6). The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders (DSM), standardizes symptom criteria and codifies psychiatric disorders. This manual contributes to the approval of new drugs, extensions of patent exclusivity, and can influence payers and mental health professionals seeking third-party reimbursements. Given the implications of the DSM on public health, it is paramount that it is free of industry influence. Previous research has shown a high prevalence of industry ties among panel and task force members of the DSM-IV-TR and DSM-5, despite the implementation of a disclosure policy for the DSM-5 (7,8). This study (9) determined the extent and type of COIs received by panel and task-force members of the DSM-5-TR (2022) (10). As the DSM-5-TR did not disclose COI, we used the Center for Medicare and Medicaid Services Open Payments (OP) database (11) to quantify them.
Lauren C. Davis, MBS
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA 19409
MedicalResearch.com: What is the background for this study?
Response: Financial conflicts of interest (COIs) resulting from ties between academia and industry have been under scrutiny for their potential to hinder the integrity of medical research. COIs can lead to implicit bias, compromise the research process, and erode public trust (1-6). The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders (DSM), standardizes symptom criteria and codifies psychiatric disorders. This manual contributes to the approval of new drugs, extensions of patent exclusivity, and can influence payers and mental health professionals seeking third-party reimbursements. Given the implications of the DSM on public health, it is paramount that it is free of industry influence. Previous research has shown a high prevalence of industry ties among panel and task force members of the DSM-IV-TR and DSM-5, despite the implementation of a disclosure policy for the DSM-5 (7,8). This study (9) determined the extent and type of COIs received by panel and task-force members of the DSM-5-TR (2022) (10). As the DSM-5-TR did not disclose COI, we used the Center for Medicare and Medicaid Services Open Payments (OP) database (11) to quantify them.


 Luke Cavanah[/caption]
Luke Cavanah, BS
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: It is well-known that schedule II stimulants, which are those that are highly addictive and include amphetamine, methylphenidate, and lisdexamfetamine, have had increasing use and misuse in the US.
Despite understanding the presence of this phenomenon, the reason for it is poorly understood. The purpose of this study was to see if rising rates of schedule II stimulants are related to the legalization of medical marijuana. We were interested in this because schedule II stimulants are primarily used as the treatment for attention-deficit hyperactivity disorder (ADHD), chronic cannabis use has been demonstrated to cause neurocognitive deficits resembling that of ADHD, and the conditions have been shown to affect similar brain regions.
Luke Cavanah[/caption]
Luke Cavanah, BS
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: It is well-known that schedule II stimulants, which are those that are highly addictive and include amphetamine, methylphenidate, and lisdexamfetamine, have had increasing use and misuse in the US.
Despite understanding the presence of this phenomenon, the reason for it is poorly understood. The purpose of this study was to see if rising rates of schedule II stimulants are related to the legalization of medical marijuana. We were interested in this because schedule II stimulants are primarily used as the treatment for attention-deficit hyperactivity disorder (ADHD), chronic cannabis use has been demonstrated to cause neurocognitive deficits resembling that of ADHD, and the conditions have been shown to affect similar brain regions.



 Edward Liu[/caption]
Edward Liu, BA
Second year medical student
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA
Medicalresearch.com: What is the background for this study?
Response: The United States’ opioid epidemic continues to rise because of increasing opioid use and availability, contributing to prescription opioid misuse, mortality, and rising cost.1 The worsening health and economic impact of opioid use disorder in the US warrants further attention on the adverse effects and distribution pattern of commonly prescribed opioids like oxycodone (OxyContin), fentanyl (Duragesic), and morphine (MS-Contin). Using the Automated Reports and Consolidated Ordering System (ARCOS) database,2 a comprehensive data collection system of pharmacies and hospitals distribution of Schedule II and III controlled substances in the US with the FDA Adverse Event Reporting System (FAERS)3 has never been done before. This approach may provide a more complete picture of the risks of prescription opioids which can include drowsiness, nausea, and potentially fatal respiratory depression.
Edward Liu[/caption]
Edward Liu, BA
Second year medical student
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA
Medicalresearch.com: What is the background for this study?
Response: The United States’ opioid epidemic continues to rise because of increasing opioid use and availability, contributing to prescription opioid misuse, mortality, and rising cost.1 The worsening health and economic impact of opioid use disorder in the US warrants further attention on the adverse effects and distribution pattern of commonly prescribed opioids like oxycodone (OxyContin), fentanyl (Duragesic), and morphine (MS-Contin). Using the Automated Reports and Consolidated Ordering System (ARCOS) database,2 a comprehensive data collection system of pharmacies and hospitals distribution of Schedule II and III controlled substances in the US with the FDA Adverse Event Reporting System (FAERS)3 has never been done before. This approach may provide a more complete picture of the risks of prescription opioids which can include drowsiness, nausea, and potentially fatal respiratory depression. 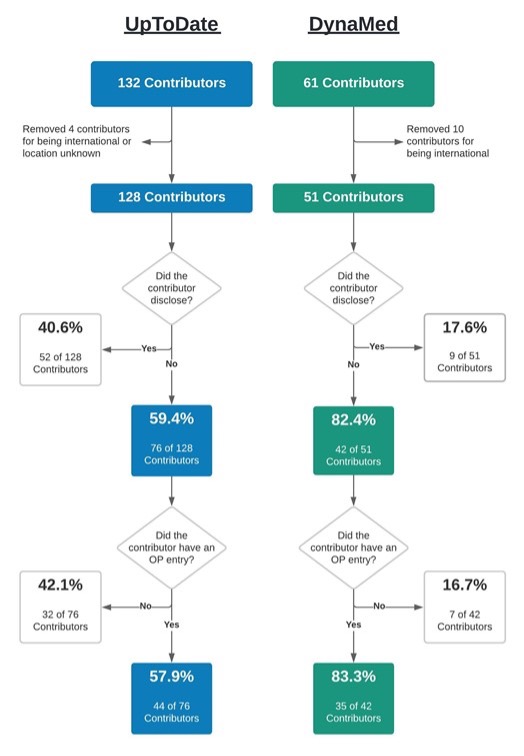
 SooYoung VanDeMar[/caption]
SooYoung VanDeMark, MBS
Geisinger Commonwealth School of Medicine
Scranton, Pennsylvania
MedicalResearch.com: What is the background for this study?
Response: Health care providers utilize subscription-based, point-of-care databases such as DynaMed and UpToDate to provide clinical care guidance and remain current on the latest evidence-based findings. Both of these websites maintain this content through a cadre of physician contributors who write and edit articles for these sites. These physician contributors are required to self-report any conflicts of interest (COI) as outlined by the respective policies on each website. However, prior COI research into similarly self-regulated areas, such as medical and pharmacology textbooks, and clinical practice guidelines, has found both appreciable potential COI and inconsistencies between self-reported and industry mandated disclosures (1-3).
This study (4) explored the accuracy of physician contributors to DynaMed and UpToDate by comparing their self-reported disclosure status with the financial remunerations they received from the healthcare industry (e.g., pharmaceutical companies) as reported to the U.S. Centers for Medicare and Medicaid Services’ Open Payments database. Physician contributors who reported “nothing to disclose” on their respective article topic but had an entry on Open Payments for having received money from industry, were classified as discordant and, thus, as having the potential for a COI. Additionally, total remuneration, gender, and payment category were investigated more in depth for each database.
SooYoung VanDeMar[/caption]
SooYoung VanDeMark, MBS
Geisinger Commonwealth School of Medicine
Scranton, Pennsylvania
MedicalResearch.com: What is the background for this study?
Response: Health care providers utilize subscription-based, point-of-care databases such as DynaMed and UpToDate to provide clinical care guidance and remain current on the latest evidence-based findings. Both of these websites maintain this content through a cadre of physician contributors who write and edit articles for these sites. These physician contributors are required to self-report any conflicts of interest (COI) as outlined by the respective policies on each website. However, prior COI research into similarly self-regulated areas, such as medical and pharmacology textbooks, and clinical practice guidelines, has found both appreciable potential COI and inconsistencies between self-reported and industry mandated disclosures (1-3).
This study (4) explored the accuracy of physician contributors to DynaMed and UpToDate by comparing their self-reported disclosure status with the financial remunerations they received from the healthcare industry (e.g., pharmaceutical companies) as reported to the U.S. Centers for Medicare and Medicaid Services’ Open Payments database. Physician contributors who reported “nothing to disclose” on their respective article topic but had an entry on Open Payments for having received money from industry, were classified as discordant and, thus, as having the potential for a COI. Additionally, total remuneration, gender, and payment category were investigated more in depth for each database.

 Sneha Vaddadi[/caption]
Sneha Vaddadi, BS
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, Pennsylvania
MedicalResearch.com: What is the background for this study?
Response: The prescription stimulants methylphenidate, amphetamine, and lisdexamfetamine, classified as Schedule II substances, are sympathomimetic drugs with therapeutic use widely used in the US for Attention Deficit Hyperactivity Disorder. Changes in criteria for diagnosis of Attention Deficit Hyperactivity Disorder in 2013 and approval of lisdexamfetamine for binge eating disorder in 2015 may have impacted usage patterns.
The goal of this study1 was to extend upon past research2 to compare the pharmacoepidemiology of these stimulants in the United States from 2010–2017, including consideration to variation within geographic regions, the Hispanic population, and the Medicaid population.
Sneha Vaddadi[/caption]
Sneha Vaddadi, BS
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, Pennsylvania
MedicalResearch.com: What is the background for this study?
Response: The prescription stimulants methylphenidate, amphetamine, and lisdexamfetamine, classified as Schedule II substances, are sympathomimetic drugs with therapeutic use widely used in the US for Attention Deficit Hyperactivity Disorder. Changes in criteria for diagnosis of Attention Deficit Hyperactivity Disorder in 2013 and approval of lisdexamfetamine for binge eating disorder in 2015 may have impacted usage patterns.
The goal of this study1 was to extend upon past research2 to compare the pharmacoepidemiology of these stimulants in the United States from 2010–2017, including consideration to variation within geographic regions, the Hispanic population, and the Medicaid population.
