Cannabis / 27.04.2026
Declines and pronounced state-level disparities in tetrahydrocannabinol (THC) prescribing to Medicare patients
[caption id="attachment_73447" align="alignleft" width="142"] Dr. Edward Liu[/caption]
MedicalResearch.com Interview with:
Edward Liu, MD
Geisinger College of Health Sciences
Scranton, PA 18509
Medicalresearch.com: What is the background for this study?
Response: Prescription drugs have high levels of uniformity that plant-based products cannot achieve. Given the liberalization of state-laws regarding medical marijuana1 over the past three-decades and increasing evidence of evidence of cannabis for conditions like chronic pain,2 we were interested in the use of the prescription formulation of delta(Δ)9-tetrahydrocannabinol (THC). A prior pharmacoepidemiology report found that prescription THC (dronabinol) to Medicaid patients decreased from 2016 to 2020. There were also pronounced state-level disparities in prescribing with a 130-fold difference when correcting for population between the highest and lowest states.
There was no research on this topic among Medicare patients. To address this gap, we obtained prescription numbers nationally and at a state level from 2014 to 2019 for Medicare Part D patients.
Dr. Edward Liu[/caption]
MedicalResearch.com Interview with:
Edward Liu, MD
Geisinger College of Health Sciences
Scranton, PA 18509
Medicalresearch.com: What is the background for this study?
Response: Prescription drugs have high levels of uniformity that plant-based products cannot achieve. Given the liberalization of state-laws regarding medical marijuana1 over the past three-decades and increasing evidence of evidence of cannabis for conditions like chronic pain,2 we were interested in the use of the prescription formulation of delta(Δ)9-tetrahydrocannabinol (THC). A prior pharmacoepidemiology report found that prescription THC (dronabinol) to Medicaid patients decreased from 2016 to 2020. There were also pronounced state-level disparities in prescribing with a 130-fold difference when correcting for population between the highest and lowest states.
There was no research on this topic among Medicare patients. To address this gap, we obtained prescription numbers nationally and at a state level from 2014 to 2019 for Medicare Part D patients.
 Dr. Edward Liu[/caption]
MedicalResearch.com Interview with:
Edward Liu, MD
Geisinger College of Health Sciences
Scranton, PA 18509
Medicalresearch.com: What is the background for this study?
Response: Prescription drugs have high levels of uniformity that plant-based products cannot achieve. Given the liberalization of state-laws regarding medical marijuana1 over the past three-decades and increasing evidence of evidence of cannabis for conditions like chronic pain,2 we were interested in the use of the prescription formulation of delta(Δ)9-tetrahydrocannabinol (THC). A prior pharmacoepidemiology report found that prescription THC (dronabinol) to Medicaid patients decreased from 2016 to 2020. There were also pronounced state-level disparities in prescribing with a 130-fold difference when correcting for population between the highest and lowest states.
There was no research on this topic among Medicare patients. To address this gap, we obtained prescription numbers nationally and at a state level from 2014 to 2019 for Medicare Part D patients.
Dr. Edward Liu[/caption]
MedicalResearch.com Interview with:
Edward Liu, MD
Geisinger College of Health Sciences
Scranton, PA 18509
Medicalresearch.com: What is the background for this study?
Response: Prescription drugs have high levels of uniformity that plant-based products cannot achieve. Given the liberalization of state-laws regarding medical marijuana1 over the past three-decades and increasing evidence of evidence of cannabis for conditions like chronic pain,2 we were interested in the use of the prescription formulation of delta(Δ)9-tetrahydrocannabinol (THC). A prior pharmacoepidemiology report found that prescription THC (dronabinol) to Medicaid patients decreased from 2016 to 2020. There were also pronounced state-level disparities in prescribing with a 130-fold difference when correcting for population between the highest and lowest states.
There was no research on this topic among Medicare patients. To address this gap, we obtained prescription numbers nationally and at a state level from 2014 to 2019 for Medicare Part D patients.









 Dr. Piper[/caption]
Brian J. Piper, PhD
Department of Medical Education
Center for Pharmacy Innovation & Outcomes
Geisinger College of Health Sciences
Scranton, PA 18509
MedicalResearch.com: What is the background for this study?
Dr. Piper: Tapentadol has an unusual mechanism of action. This opioid is similar to morphine in that it activates mu, the main opiate receptor which is important for pain. This drug also acts similar to an antidepressant like duloxetine (Cymbalta) with equal ability to block both the norepinephrine and the serotonin transporters (Figure 1). The combination of opioid and monoaminergic activity may influence both therapeutic and side effect profiles.
[caption id="attachment_68512" align="aligncenter" width="192"]
Dr. Piper[/caption]
Brian J. Piper, PhD
Department of Medical Education
Center for Pharmacy Innovation & Outcomes
Geisinger College of Health Sciences
Scranton, PA 18509
MedicalResearch.com: What is the background for this study?
Dr. Piper: Tapentadol has an unusual mechanism of action. This opioid is similar to morphine in that it activates mu, the main opiate receptor which is important for pain. This drug also acts similar to an antidepressant like duloxetine (Cymbalta) with equal ability to block both the norepinephrine and the serotonin transporters (Figure 1). The combination of opioid and monoaminergic activity may influence both therapeutic and side effect profiles.
[caption id="attachment_68512" align="aligncenter" width="192"] Figure 1. Biological mechanism of action of the atypical opioid tapentadol involves binding to the mu opiate receptor, blocking the norepinephrine transporter (NET), and blocking the serotonin transporter (SERT) [1].[/caption]Tapentadol is approved in the US for moderate to severe acute pain, moderate to severe chronic pain, and neuropathic pain associated with diabetic peripheral neuropathy. However, tapentadol is not generally considered first-line for any of these indications. Although this agent has a high potential for abuse and is classified as a Schedule II substance, a manufacturer of extended release tapentadol (Grunenthal) has conducted a misinformation campaign claiming tapentadol has a “minimum potential of abuse”, does not cause respiratory depression (a well-known opioid effect), does not cause any euphoric feelings, has no street value, and is not an opioid [2]. Prior pharmacoepidemiological studies have examined the use patterns of other prescription opioids that can be misused like oxycodone [3], fentanyl [4], meperidine [5] and methadone [6] but there is limited national information about the use of tapentadol.
Figure 1. Biological mechanism of action of the atypical opioid tapentadol involves binding to the mu opiate receptor, blocking the norepinephrine transporter (NET), and blocking the serotonin transporter (SERT) [1].[/caption]Tapentadol is approved in the US for moderate to severe acute pain, moderate to severe chronic pain, and neuropathic pain associated with diabetic peripheral neuropathy. However, tapentadol is not generally considered first-line for any of these indications. Although this agent has a high potential for abuse and is classified as a Schedule II substance, a manufacturer of extended release tapentadol (Grunenthal) has conducted a misinformation campaign claiming tapentadol has a “minimum potential of abuse”, does not cause respiratory depression (a well-known opioid effect), does not cause any euphoric feelings, has no street value, and is not an opioid [2]. Prior pharmacoepidemiological studies have examined the use patterns of other prescription opioids that can be misused like oxycodone [3], fentanyl [4], meperidine [5] and methadone [6] but there is limited national information about the use of tapentadol.

 Luke Cavanah B.S.[/caption]
Luke Cavanah, BS
Department of Medical Education
Geisinger College of Health Sciences
Scranton, PA, 18509
MedicalResearch.com: What is the background for this study?
Response: Almost a third of patients with medication-treated major depressive disorder will fail at least two depressants—many patients failing more.1 Esketamine is an intranasal medication that was FDA-approved in 2019 for such patients when used in conjunction with an oral antidepressant.2 Esketamine also is unique from the more traditional oral antidepressants in its mechanism of action, quick onset, and evidence for treatment of acute suicidality.3 While esketamine has been of much interest for these last few years, a recent study found low and geographically variable use of this medication within the Medicaid population.4 We were interested in investigating esketamine treatment utilization among Medicare patients in 2019 and 2020.
Luke Cavanah B.S.[/caption]
Luke Cavanah, BS
Department of Medical Education
Geisinger College of Health Sciences
Scranton, PA, 18509
MedicalResearch.com: What is the background for this study?
Response: Almost a third of patients with medication-treated major depressive disorder will fail at least two depressants—many patients failing more.1 Esketamine is an intranasal medication that was FDA-approved in 2019 for such patients when used in conjunction with an oral antidepressant.2 Esketamine also is unique from the more traditional oral antidepressants in its mechanism of action, quick onset, and evidence for treatment of acute suicidality.3 While esketamine has been of much interest for these last few years, a recent study found low and geographically variable use of this medication within the Medicaid population.4 We were interested in investigating esketamine treatment utilization among Medicare patients in 2019 and 2020.

 Parita K. Ray[/caption]
Parita K. Ray[/caption]
 Elena Stains[/caption]
Elena Stains
Medical Student
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
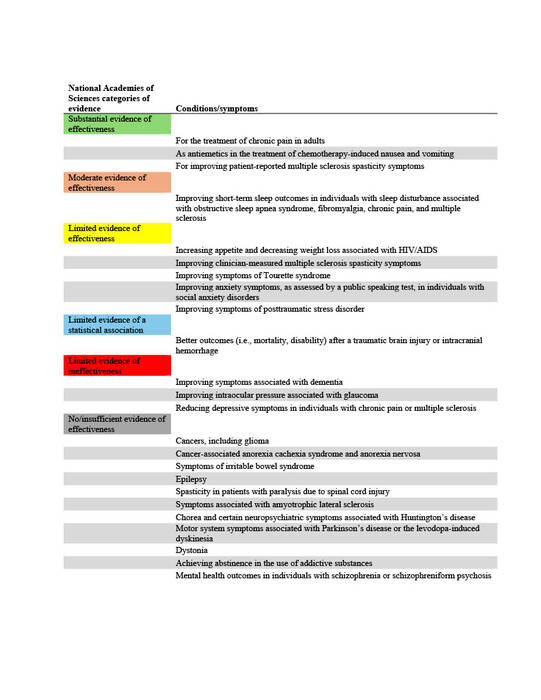
Response: In 2019 to 2020, 2.5% of Americans reported using cannabis for medical needs, compared to 1.2% in 2013-2014, representing a 12.9% annual increase1. Forty states and the District of Columbia have legislation for some form of medical cannabis (MC) in 2024. Because MC is not federally legalized, each state creates its own legislation on the conditions that qualify a person for MC, without any standardized process to determine what qualifying conditions (QC) are proven to be aided by MC. Thus, the QCs chosen by states vary widely. Common QCs include cancer, dementia, and PTSD.
The National Academies of Sciences, Engineering, and Medicine (NAS) published a report in 2017 on the evidence for the therapeutic effects of cannabis and cannabinoids for over twenty conditions2. This report reviews the evidence of effectiveness of medical cannabis for the most common QCs chosen by states. The researchers at Geisinger Commonwealth School of Medicine aimed to compare the evidence found by the NAS report with the QCs of 38 states (including the District of Columbia) in both 2017 and 2024. QCs were categorized based on NAS-established level of evidence: limited, moderate, or substantial/conclusive evidence of effectiveness, limited evidence of ineffectiveness, or no/insufficient evidence to support or refute effectiveness (Table 1).
Elena Stains[/caption]
Elena Stains
Medical Student
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: In 2019 to 2020, 2.5% of Americans reported using cannabis for medical needs, compared to 1.2% in 2013-2014, representing a 12.9% annual increase1. Forty states and the District of Columbia have legislation for some form of medical cannabis (MC) in 2024. Because MC is not federally legalized, each state creates its own legislation on the conditions that qualify a person for MC, without any standardized process to determine what qualifying conditions (QC) are proven to be aided by MC. Thus, the QCs chosen by states vary widely. Common QCs include cancer, dementia, and PTSD.
The National Academies of Sciences, Engineering, and Medicine (NAS) published a report in 2017 on the evidence for the therapeutic effects of cannabis and cannabinoids for over twenty conditions2. This report reviews the evidence of effectiveness of medical cannabis for the most common QCs chosen by states. The researchers at Geisinger Commonwealth School of Medicine aimed to compare the evidence found by the NAS report with the QCs of 38 states (including the District of Columbia) in both 2017 and 2024. QCs were categorized based on NAS-established level of evidence: limited, moderate, or substantial/conclusive evidence of effectiveness, limited evidence of ineffectiveness, or no/insufficient evidence to support or refute effectiveness (Table 1).





 Elena Stains
Medical Student
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: Opioid use has been an increasing problem since the early 2000s in the United States (US) with a surge around 2010. Twenty-five percent of those having abused pain relievers in 2013 and 2014 got those drugs from physicians1. Physicians are particularly well-known for fueling the opioid crisis in Florida in the 2000s. Of the United States’ top 100 opioid prescribing physicians in 2010, an astounding 98 were prescribing in Florida2. Florida taking the main stage of the opioid crisis can be attributed to several factors, including ability of physicians to dispense opioids directly from their offices to patients (i.e. without pharmacists) and the presence of many infamous “pill mills” in the state3–6.
The researchers at Geisinger Commonwealth School of Medicine aimed to analyze the amount of hydrocodone (including brand names of Vicodin and Lortab) and oxycodone (OxyContin and Percocet) distributed in Florida from 2006 to 2021, paying close attention to the peak year of the opioid crisis, 2010. The team used the Washington Post and the US Drug Enforcement Administration’s Automation of Reports and Consolidated Orders System (ARCOS) databases to compile this compelling information.
Elena Stains
Medical Student
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: Opioid use has been an increasing problem since the early 2000s in the United States (US) with a surge around 2010. Twenty-five percent of those having abused pain relievers in 2013 and 2014 got those drugs from physicians1. Physicians are particularly well-known for fueling the opioid crisis in Florida in the 2000s. Of the United States’ top 100 opioid prescribing physicians in 2010, an astounding 98 were prescribing in Florida2. Florida taking the main stage of the opioid crisis can be attributed to several factors, including ability of physicians to dispense opioids directly from their offices to patients (i.e. without pharmacists) and the presence of many infamous “pill mills” in the state3–6.
The researchers at Geisinger Commonwealth School of Medicine aimed to analyze the amount of hydrocodone (including brand names of Vicodin and Lortab) and oxycodone (OxyContin and Percocet) distributed in Florida from 2006 to 2021, paying close attention to the peak year of the opioid crisis, 2010. The team used the Washington Post and the US Drug Enforcement Administration’s Automation of Reports and Consolidated Orders System (ARCOS) databases to compile this compelling information.


 Amy Kennalley[/caption]
Amy Kennalley, MBS
First Year Medical Student
Department of Medical Education
Geisinger Commonwealth School of Medicine
MedicalResearch.com: What is the background for this study
Response: The legalization of medical marijuana (MMJ) and the number of qualifying conditions are expanding across the USA, emphasizing the need to understand the implications of MMJ dispensary distribution for equitable access. Pennsylvania (PA) legalized MMJ in 2016, with the first dispensary opening its doors in 2018. The state currently recognizes 24 medical conditions for MMJ use, including six for which there is insufficient or no evidence for their efficacy as a treatment. Prior research suggests that there is a link between proximity to dispensaries and overall MMJ use. However, a gap exists in our understanding of how dispensary locations might be associated with the specific qualifying conditions for which individuals receive their certification.
In response to this knowledge gap, our study delved into the medical marijuana dispensary access in PA and explored associations with both MMJ certifications and the community demographics. Utilizing data from the Pennsylvania Department of Health, we investigated how proximity to MMJ dispensaries related to the proportion of individuals certified for MMJ use within a specific area or Zip Code Tabulation Area. Additionally, we analyzed the proportion of certifications for conditions with varying levels of evidence supporting the efficacy of MMJ.
This pioneering study represents the first of its kind in PA, shedding light on the association between MMJ dispensary locations and certifications. Likewise, it is the first in the US to investigate the link between dispensary locations and specific qualifying conditions. By examining these dynamics, we aim to contribute vital insights to inform policy and practice, ensuring equitable access to MMJ treatment for individuals with diverse medical needs.
Amy Kennalley[/caption]
Amy Kennalley, MBS
First Year Medical Student
Department of Medical Education
Geisinger Commonwealth School of Medicine
MedicalResearch.com: What is the background for this study
Response: The legalization of medical marijuana (MMJ) and the number of qualifying conditions are expanding across the USA, emphasizing the need to understand the implications of MMJ dispensary distribution for equitable access. Pennsylvania (PA) legalized MMJ in 2016, with the first dispensary opening its doors in 2018. The state currently recognizes 24 medical conditions for MMJ use, including six for which there is insufficient or no evidence for their efficacy as a treatment. Prior research suggests that there is a link between proximity to dispensaries and overall MMJ use. However, a gap exists in our understanding of how dispensary locations might be associated with the specific qualifying conditions for which individuals receive their certification.
In response to this knowledge gap, our study delved into the medical marijuana dispensary access in PA and explored associations with both MMJ certifications and the community demographics. Utilizing data from the Pennsylvania Department of Health, we investigated how proximity to MMJ dispensaries related to the proportion of individuals certified for MMJ use within a specific area or Zip Code Tabulation Area. Additionally, we analyzed the proportion of certifications for conditions with varying levels of evidence supporting the efficacy of MMJ.
This pioneering study represents the first of its kind in PA, shedding light on the association between MMJ dispensary locations and certifications. Likewise, it is the first in the US to investigate the link between dispensary locations and specific qualifying conditions. By examining these dynamics, we aim to contribute vital insights to inform policy and practice, ensuring equitable access to MMJ treatment for individuals with diverse medical needs.



 Lauren C. Davis, MBS
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA 19409
MedicalResearch.com: What is the background for this study?
Response: Financial conflicts of interest (COIs) resulting from ties between academia and industry have been under scrutiny for their potential to hinder the integrity of medical research. COIs can lead to implicit bias, compromise the research process, and erode public trust (1-6). The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders (DSM), standardizes symptom criteria and codifies psychiatric disorders. This manual contributes to the approval of new drugs, extensions of patent exclusivity, and can influence payers and mental health professionals seeking third-party reimbursements. Given the implications of the DSM on public health, it is paramount that it is free of industry influence. Previous research has shown a high prevalence of industry ties among panel and task force members of the DSM-IV-TR and DSM-5, despite the implementation of a disclosure policy for the DSM-5 (7,8). This study (9) determined the extent and type of COIs received by panel and task-force members of the DSM-5-TR (2022) (10). As the DSM-5-TR did not disclose COI, we used the Center for Medicare and Medicaid Services Open Payments (OP) database (11) to quantify them.
Lauren C. Davis, MBS
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA 19409
MedicalResearch.com: What is the background for this study?
Response: Financial conflicts of interest (COIs) resulting from ties between academia and industry have been under scrutiny for their potential to hinder the integrity of medical research. COIs can lead to implicit bias, compromise the research process, and erode public trust (1-6). The American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders (DSM), standardizes symptom criteria and codifies psychiatric disorders. This manual contributes to the approval of new drugs, extensions of patent exclusivity, and can influence payers and mental health professionals seeking third-party reimbursements. Given the implications of the DSM on public health, it is paramount that it is free of industry influence. Previous research has shown a high prevalence of industry ties among panel and task force members of the DSM-IV-TR and DSM-5, despite the implementation of a disclosure policy for the DSM-5 (7,8). This study (9) determined the extent and type of COIs received by panel and task-force members of the DSM-5-TR (2022) (10). As the DSM-5-TR did not disclose COI, we used the Center for Medicare and Medicaid Services Open Payments (OP) database (11) to quantify them.


 Luke Cavanah[/caption]
Luke Cavanah, BS
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: It is well-known that schedule II stimulants, which are those that are highly addictive and include amphetamine, methylphenidate, and lisdexamfetamine, have had increasing use and misuse in the US.
Despite understanding the presence of this phenomenon, the reason for it is poorly understood. The purpose of this study was to see if rising rates of schedule II stimulants are related to the legalization of medical marijuana. We were interested in this because schedule II stimulants are primarily used as the treatment for attention-deficit hyperactivity disorder (ADHD), chronic cannabis use has been demonstrated to cause neurocognitive deficits resembling that of ADHD, and the conditions have been shown to affect similar brain regions.
Luke Cavanah[/caption]
Luke Cavanah, BS
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: It is well-known that schedule II stimulants, which are those that are highly addictive and include amphetamine, methylphenidate, and lisdexamfetamine, have had increasing use and misuse in the US.
Despite understanding the presence of this phenomenon, the reason for it is poorly understood. The purpose of this study was to see if rising rates of schedule II stimulants are related to the legalization of medical marijuana. We were interested in this because schedule II stimulants are primarily used as the treatment for attention-deficit hyperactivity disorder (ADHD), chronic cannabis use has been demonstrated to cause neurocognitive deficits resembling that of ADHD, and the conditions have been shown to affect similar brain regions.



 Edward Liu[/caption]
Edward Liu, BA
Second year medical student
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA
Medicalresearch.com: What is the background for this study?
Response: The United States’ opioid epidemic continues to rise because of increasing opioid use and availability, contributing to prescription opioid misuse, mortality, and rising cost.1 The worsening health and economic impact of opioid use disorder in the US warrants further attention on the adverse effects and distribution pattern of commonly prescribed opioids like oxycodone (OxyContin), fentanyl (Duragesic), and morphine (MS-Contin). Using the Automated Reports and Consolidated Ordering System (ARCOS) database,2 a comprehensive data collection system of pharmacies and hospitals distribution of Schedule II and III controlled substances in the US with the FDA Adverse Event Reporting System (FAERS)3 has never been done before. This approach may provide a more complete picture of the risks of prescription opioids which can include drowsiness, nausea, and potentially fatal respiratory depression.
Edward Liu[/caption]
Edward Liu, BA
Second year medical student
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA
Medicalresearch.com: What is the background for this study?
Response: The United States’ opioid epidemic continues to rise because of increasing opioid use and availability, contributing to prescription opioid misuse, mortality, and rising cost.1 The worsening health and economic impact of opioid use disorder in the US warrants further attention on the adverse effects and distribution pattern of commonly prescribed opioids like oxycodone (OxyContin), fentanyl (Duragesic), and morphine (MS-Contin). Using the Automated Reports and Consolidated Ordering System (ARCOS) database,2 a comprehensive data collection system of pharmacies and hospitals distribution of Schedule II and III controlled substances in the US with the FDA Adverse Event Reporting System (FAERS)3 has never been done before. This approach may provide a more complete picture of the risks of prescription opioids which can include drowsiness, nausea, and potentially fatal respiratory depression. 
 Alexia Aguilar[/caption]
Alexia Aguilar
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: Traditional antidepressants like Zoloft and Lexapro have three major drawbacks.
Alexia Aguilar[/caption]
Alexia Aguilar
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: Traditional antidepressants like Zoloft and Lexapro have three major drawbacks.

 Kenya Colvin[/caption]
Kenya Colvin, MBS
Department of Medical Education
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: Vaccine hesitancy is a major driver of COVID-19 vaccination disparities between minority and non-Hispanic White communities. Our goal was to understand what factors influenced vaccine hesitancy among individuals in Eastern Pennsylvania to identify more effective ways to promote vaccine uptake within minority communities.
Kenya Colvin[/caption]
Kenya Colvin, MBS
Department of Medical Education
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: Vaccine hesitancy is a major driver of COVID-19 vaccination disparities between minority and non-Hispanic White communities. Our goal was to understand what factors influenced vaccine hesitancy among individuals in Eastern Pennsylvania to identify more effective ways to promote vaccine uptake within minority communities.