Addiction, Author Interviews, COVID -19 Coronavirus, Opiods / 07.02.2023
Geisinger Study: Methadone ‘take-homes’ during COVID-19 Resulted in Greater Methadone Overdoses
MedicalResearch.com Interview with:
[caption id="attachment_56276" align="alignleft" width="200"] Dr. Piper[/caption]
Brian Piper, PhD MS
Assistant Professor of Neuroscience
Center for Pharmacy Innovation & Outcomes
Geisinger School of Graduate Education
MedicalResearch.com: What is the background for this study?
Response: Methadone is an evidence-based treatment of opioid use disorder (OUD) and pain. However, this Schedule II opioid can also cause respiratory depression, which can result in lethality. The need for supervised administration is a long-standing source of frustration in the U.S. for many opioid use disorder (OUD) methadone patients. However, there was an accommodation in early 2020 thanks to the COVID-19 pandemic. This involved extending the take-home supply to up to 28-days for stable patients and 14 days for less stable patients. Prior research found that the implementation of supervised administration in England greatly reduced methadone overdoses [1]. The primary objective of this study [2] from Geisinger Commonwealth School of Medicine was to determine if the relaxation of the take-home rules resulted in more methadone overdoses.
Dr. Piper[/caption]
Brian Piper, PhD MS
Assistant Professor of Neuroscience
Center for Pharmacy Innovation & Outcomes
Geisinger School of Graduate Education
MedicalResearch.com: What is the background for this study?
Response: Methadone is an evidence-based treatment of opioid use disorder (OUD) and pain. However, this Schedule II opioid can also cause respiratory depression, which can result in lethality. The need for supervised administration is a long-standing source of frustration in the U.S. for many opioid use disorder (OUD) methadone patients. However, there was an accommodation in early 2020 thanks to the COVID-19 pandemic. This involved extending the take-home supply to up to 28-days for stable patients and 14 days for less stable patients. Prior research found that the implementation of supervised administration in England greatly reduced methadone overdoses [1]. The primary objective of this study [2] from Geisinger Commonwealth School of Medicine was to determine if the relaxation of the take-home rules resulted in more methadone overdoses.
 Dr. Piper[/caption]
Brian Piper, PhD MS
Assistant Professor of Neuroscience
Center for Pharmacy Innovation & Outcomes
Geisinger School of Graduate Education
MedicalResearch.com: What is the background for this study?
Response: Methadone is an evidence-based treatment of opioid use disorder (OUD) and pain. However, this Schedule II opioid can also cause respiratory depression, which can result in lethality. The need for supervised administration is a long-standing source of frustration in the U.S. for many opioid use disorder (OUD) methadone patients. However, there was an accommodation in early 2020 thanks to the COVID-19 pandemic. This involved extending the take-home supply to up to 28-days for stable patients and 14 days for less stable patients. Prior research found that the implementation of supervised administration in England greatly reduced methadone overdoses [1]. The primary objective of this study [2] from Geisinger Commonwealth School of Medicine was to determine if the relaxation of the take-home rules resulted in more methadone overdoses.
Dr. Piper[/caption]
Brian Piper, PhD MS
Assistant Professor of Neuroscience
Center for Pharmacy Innovation & Outcomes
Geisinger School of Graduate Education
MedicalResearch.com: What is the background for this study?
Response: Methadone is an evidence-based treatment of opioid use disorder (OUD) and pain. However, this Schedule II opioid can also cause respiratory depression, which can result in lethality. The need for supervised administration is a long-standing source of frustration in the U.S. for many opioid use disorder (OUD) methadone patients. However, there was an accommodation in early 2020 thanks to the COVID-19 pandemic. This involved extending the take-home supply to up to 28-days for stable patients and 14 days for less stable patients. Prior research found that the implementation of supervised administration in England greatly reduced methadone overdoses [1]. The primary objective of this study [2] from Geisinger Commonwealth School of Medicine was to determine if the relaxation of the take-home rules resulted in more methadone overdoses.
 SooYoung VanDeMar[/caption]
SooYoung VanDeMark, MBS
Geisinger Commonwealth School of Medicine
Scranton, Pennsylvania
MedicalResearch.com: What is the background for this study?
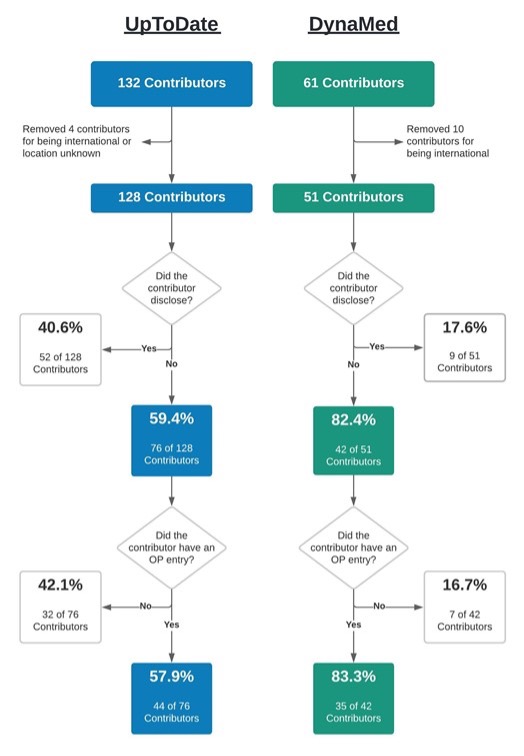
Response: Health care providers utilize subscription-based, point-of-care databases such as DynaMed and UpToDate to provide clinical care guidance and remain current on the latest evidence-based findings. Both of these websites maintain this content through a cadre of physician contributors who write and edit articles for these sites. These physician contributors are required to self-report any conflicts of interest (COI) as outlined by the respective policies on each website. However, prior COI research into similarly self-regulated areas, such as medical and pharmacology textbooks, and clinical practice guidelines, has found both appreciable potential COI and inconsistencies between self-reported and industry mandated disclosures (1-3).
This study (4) explored the accuracy of physician contributors to DynaMed and UpToDate by comparing their self-reported disclosure status with the financial remunerations they received from the healthcare industry (e.g., pharmaceutical companies) as reported to the U.S. Centers for Medicare and Medicaid Services’ Open Payments database. Physician contributors who reported “nothing to disclose” on their respective article topic but had an entry on Open Payments for having received money from industry, were classified as discordant and, thus, as having the potential for a COI. Additionally, total remuneration, gender, and payment category were investigated more in depth for each database.
SooYoung VanDeMar[/caption]
SooYoung VanDeMark, MBS
Geisinger Commonwealth School of Medicine
Scranton, Pennsylvania
MedicalResearch.com: What is the background for this study?
Response: Health care providers utilize subscription-based, point-of-care databases such as DynaMed and UpToDate to provide clinical care guidance and remain current on the latest evidence-based findings. Both of these websites maintain this content through a cadre of physician contributors who write and edit articles for these sites. These physician contributors are required to self-report any conflicts of interest (COI) as outlined by the respective policies on each website. However, prior COI research into similarly self-regulated areas, such as medical and pharmacology textbooks, and clinical practice guidelines, has found both appreciable potential COI and inconsistencies between self-reported and industry mandated disclosures (1-3).
This study (4) explored the accuracy of physician contributors to DynaMed and UpToDate by comparing their self-reported disclosure status with the financial remunerations they received from the healthcare industry (e.g., pharmaceutical companies) as reported to the U.S. Centers for Medicare and Medicaid Services’ Open Payments database. Physician contributors who reported “nothing to disclose” on their respective article topic but had an entry on Open Payments for having received money from industry, were classified as discordant and, thus, as having the potential for a COI. Additionally, total remuneration, gender, and payment category were investigated more in depth for each database.
