Author Interviews, JAMA, OBGYNE, Pediatrics, USPSTF / 09.08.2023
USPSTF: Anyone Who Could Become Pregnant Should Take Folic Acid To Prevent Spinal Cord Defects
MedicalResearch.com Interview with:x
[caption id="attachment_60752" align="alignleft" width="133"] Dr. Nicholson[/caption]
Wanda K. Nicholson, M.D., M.P.H., M.B.A.
Senior Associate Dean for Diversity, Equity, and Inclusion
Professor of Prevention and Community Health
Milken Institute School of Public Health
George Washington University
Vice chair of the U.S. Preventive Services Task Force
MedicalResearch.com: What is the background for this study?
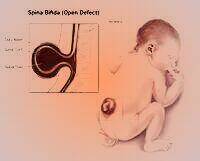
Response: Neural tube defects are when a baby’s spinal cord or brain don’t develop properly during pregnancy, which can cause serious complications including disability and death. The good news is that taking folic acid supplements before and during early pregnancy is proven to help prevent this from happening.
Dr. Nicholson[/caption]
Wanda K. Nicholson, M.D., M.P.H., M.B.A.
Senior Associate Dean for Diversity, Equity, and Inclusion
Professor of Prevention and Community Health
Milken Institute School of Public Health
George Washington University
Vice chair of the U.S. Preventive Services Task Force
MedicalResearch.com: What is the background for this study?
Response: Neural tube defects are when a baby’s spinal cord or brain don’t develop properly during pregnancy, which can cause serious complications including disability and death. The good news is that taking folic acid supplements before and during early pregnancy is proven to help prevent this from happening.
 Dr. Nicholson[/caption]
Wanda K. Nicholson, M.D., M.P.H., M.B.A.
Senior Associate Dean for Diversity, Equity, and Inclusion
Professor of Prevention and Community Health
Milken Institute School of Public Health
George Washington University
Vice chair of the U.S. Preventive Services Task Force
MedicalResearch.com: What is the background for this study?
Response: Neural tube defects are when a baby’s spinal cord or brain don’t develop properly during pregnancy, which can cause serious complications including disability and death. The good news is that taking folic acid supplements before and during early pregnancy is proven to help prevent this from happening.
Dr. Nicholson[/caption]
Wanda K. Nicholson, M.D., M.P.H., M.B.A.
Senior Associate Dean for Diversity, Equity, and Inclusion
Professor of Prevention and Community Health
Milken Institute School of Public Health
George Washington University
Vice chair of the U.S. Preventive Services Task Force
MedicalResearch.com: What is the background for this study?
Response: Neural tube defects are when a baby’s spinal cord or brain don’t develop properly during pregnancy, which can cause serious complications including disability and death. The good news is that taking folic acid supplements before and during early pregnancy is proven to help prevent this from happening.




 Leighton Ku, PhD, MPH
Professor, Dept. of Health Policy and Management
Director, Center for Health Policy Research
Milken Institute School of Public Health
George Washington University
Washington, DC 20052
MedicalResearch.com: What is the background for this study?
Response: In this study, we examined how requirements that low-income adults work in order to keep their food assistance benefits (SNAP, formerly called food stamps) affects the number of people receiving benefits. Briefly, we found, based on analyses of data from 2,410 counties from 2013 to 2017, that soon after work requirements are introduced, more than a third of affected participants lose their food assistance. This meant that about 600,000 poor adults lost food assistance very quickly.
This is important for two reasons:
(1) Work requirements create greater hardship, including food insecurity and increased risk of health problems, when poor people lose their nutrition benefits.
(2) The Trump Administration is trying to broaden this policy, expanding it further in SNAP, but also applying work requirements to Medicaid (for health insurance) and public housing benefits. This is a massive effort at social experimentation that will cause tremendous harm.
And the sad part is that we already know, from other research, that these work requirement programs do not actually help people get jobs, keep them or to become more self-sufficient. This is because the work requirements do not address the real needs of low-income unemployed people, to learn how to get better job skills or to have supports, such as child care, transportation or health insurance, that let them keep working.
Leighton Ku, PhD, MPH
Professor, Dept. of Health Policy and Management
Director, Center for Health Policy Research
Milken Institute School of Public Health
George Washington University
Washington, DC 20052
MedicalResearch.com: What is the background for this study?
Response: In this study, we examined how requirements that low-income adults work in order to keep their food assistance benefits (SNAP, formerly called food stamps) affects the number of people receiving benefits. Briefly, we found, based on analyses of data from 2,410 counties from 2013 to 2017, that soon after work requirements are introduced, more than a third of affected participants lose their food assistance. This meant that about 600,000 poor adults lost food assistance very quickly.
This is important for two reasons:
(1) Work requirements create greater hardship, including food insecurity and increased risk of health problems, when poor people lose their nutrition benefits.
(2) The Trump Administration is trying to broaden this policy, expanding it further in SNAP, but also applying work requirements to Medicaid (for health insurance) and public housing benefits. This is a massive effort at social experimentation that will cause tremendous harm.
And the sad part is that we already know, from other research, that these work requirement programs do not actually help people get jobs, keep them or to become more self-sufficient. This is because the work requirements do not address the real needs of low-income unemployed people, to learn how to get better job skills or to have supports, such as child care, transportation or health insurance, that let them keep working.