Author Interviews, Macular Degeneration, Ophthalmology, PNAS / 27.10.2021
Fluoxetine – Prozac – May Be Beneficial for Dry Macular Degeneration
MedicalResearch.com Interview with:
[caption id="attachment_58321" align="alignleft" width="200"] Dr. Gelfand[/caption]
Bradley D. Gelfand PhD
Center for Advanced Vision Science
Department of Ophthalmology
Department of Biomedical Engineering
University of Virginia School of Medicine
Charlottesville, VA 22908
MedicalResearch.com: What is the background for this study? Would you briefly describe dry AMD?
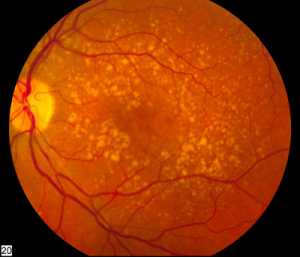
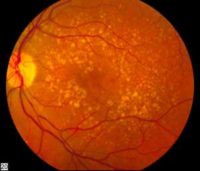
Response: Dry age-related macular degeneration (AMD) is a form of AMD that affects about 11 million people in the United States, and many millions more worldwide. Dry AMD is a disease affecting the macula, the central part of our retina that is responsible for fine visual acuity tasks - things like reading, driving, and recognizing faces. Dry AMD typically develops in people in their 6th, 7th, and 8th decades of life and begins with small changes to the retina that are unlikely to affect vision at first. As the disease progresses, it can develop into more advanced stages ("wet" AMD and geographic atrophy), which can cause blindness. Unfortunately, there is no approved treatment that can prevent dry AMD or its progression to advanced blinding stages.
Dr. Gelfand[/caption]
Bradley D. Gelfand PhD
Center for Advanced Vision Science
Department of Ophthalmology
Department of Biomedical Engineering
University of Virginia School of Medicine
Charlottesville, VA 22908
MedicalResearch.com: What is the background for this study? Would you briefly describe dry AMD?
Response: Dry age-related macular degeneration (AMD) is a form of AMD that affects about 11 million people in the United States, and many millions more worldwide. Dry AMD is a disease affecting the macula, the central part of our retina that is responsible for fine visual acuity tasks - things like reading, driving, and recognizing faces. Dry AMD typically develops in people in their 6th, 7th, and 8th decades of life and begins with small changes to the retina that are unlikely to affect vision at first. As the disease progresses, it can develop into more advanced stages ("wet" AMD and geographic atrophy), which can cause blindness. Unfortunately, there is no approved treatment that can prevent dry AMD or its progression to advanced blinding stages.
 Dr. Gelfand[/caption]
Bradley D. Gelfand PhD
Center for Advanced Vision Science
Department of Ophthalmology
Department of Biomedical Engineering
University of Virginia School of Medicine
Charlottesville, VA 22908
MedicalResearch.com: What is the background for this study? Would you briefly describe dry AMD?
Response: Dry age-related macular degeneration (AMD) is a form of AMD that affects about 11 million people in the United States, and many millions more worldwide. Dry AMD is a disease affecting the macula, the central part of our retina that is responsible for fine visual acuity tasks - things like reading, driving, and recognizing faces. Dry AMD typically develops in people in their 6th, 7th, and 8th decades of life and begins with small changes to the retina that are unlikely to affect vision at first. As the disease progresses, it can develop into more advanced stages ("wet" AMD and geographic atrophy), which can cause blindness. Unfortunately, there is no approved treatment that can prevent dry AMD or its progression to advanced blinding stages.
Dr. Gelfand[/caption]
Bradley D. Gelfand PhD
Center for Advanced Vision Science
Department of Ophthalmology
Department of Biomedical Engineering
University of Virginia School of Medicine
Charlottesville, VA 22908
MedicalResearch.com: What is the background for this study? Would you briefly describe dry AMD?
Response: Dry age-related macular degeneration (AMD) is a form of AMD that affects about 11 million people in the United States, and many millions more worldwide. Dry AMD is a disease affecting the macula, the central part of our retina that is responsible for fine visual acuity tasks - things like reading, driving, and recognizing faces. Dry AMD typically develops in people in their 6th, 7th, and 8th decades of life and begins with small changes to the retina that are unlikely to affect vision at first. As the disease progresses, it can develop into more advanced stages ("wet" AMD and geographic atrophy), which can cause blindness. Unfortunately, there is no approved treatment that can prevent dry AMD or its progression to advanced blinding stages.






 Adam Glassman[/caption]
MedicalResearch.com Interview with:
Adam Glassman, M.S.
Director, DRCRnet Coordinating Center
Jaeb Center for Health Research
Tampa, FL 33647
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Diabetic macular edema (DME) is the most common cause of vision loss in patients with diabetes, impairing the vision of approximately 750,000 people in the United States. The most common treatment involves the injection into the eye of one of 3 drugs that inhibit vascular endothelial growth factor (VEGF). The Diabetic Retinopathy Clinical Research Network, funded by the National Institutes of Health, conducted a randomized clinical trial on the comparative effectiveness of the 3 anti-VEGF drugs EYLEA®, Avastin®, or Lucentis® for eyes with decreased vision from diabetic macular edema. There are substantial cost differences between the three drugs. In the United States, EYLEA® costs approximately $1850, repackaged (compounded) Avastin® $60, and Lucentis® $1170 per injection. In eyes with relatively good starting vision, there were no differences in vision outcomes; all three groups, on average, had improved vision. In eyes with starting vision of 20/50 or worse, EYLEA® had better vision outcomes at 1 year than either Avastin® or Lucentis®, and better vision outcomes at 2 years than Avastin®. However, given that, on average, eyes will receive 9 to 10 injections within the first year of treatment and 5 injections in the second year, neither EYLEA® nor Lucentis® would be considered cost effective by standard benchmarks compared with Avastin® unless their prices decrease substantially.
Adam Glassman[/caption]
MedicalResearch.com Interview with:
Adam Glassman, M.S.
Director, DRCRnet Coordinating Center
Jaeb Center for Health Research
Tampa, FL 33647
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Diabetic macular edema (DME) is the most common cause of vision loss in patients with diabetes, impairing the vision of approximately 750,000 people in the United States. The most common treatment involves the injection into the eye of one of 3 drugs that inhibit vascular endothelial growth factor (VEGF). The Diabetic Retinopathy Clinical Research Network, funded by the National Institutes of Health, conducted a randomized clinical trial on the comparative effectiveness of the 3 anti-VEGF drugs EYLEA®, Avastin®, or Lucentis® for eyes with decreased vision from diabetic macular edema. There are substantial cost differences between the three drugs. In the United States, EYLEA® costs approximately $1850, repackaged (compounded) Avastin® $60, and Lucentis® $1170 per injection. In eyes with relatively good starting vision, there were no differences in vision outcomes; all three groups, on average, had improved vision. In eyes with starting vision of 20/50 or worse, EYLEA® had better vision outcomes at 1 year than either Avastin® or Lucentis®, and better vision outcomes at 2 years than Avastin®. However, given that, on average, eyes will receive 9 to 10 injections within the first year of treatment and 5 injections in the second year, neither EYLEA® nor Lucentis® would be considered cost effective by standard benchmarks compared with Avastin® unless their prices decrease substantially.

 Dr. Berg[/caption]
MedicalResearch.com Interview with:
Karina Birgitta Berg MD
Department of Ophthalmology
Oslo University Hospital
Oslo, Norway
Medical Research: What is the background for this study? What are the main findings?
Dr. Berg: Neovascular age-related macular degeneration (nAMD) has been the leading cause of vision loss in the elderly population of Western countries. Inhibition of vascular endothelial growth factor (VEGF) with medications such as bevacizumab and ranibizumab injected into the eye, has dramatically reduced the incidence of social blindness from this disease.
Dr. Berg[/caption]
MedicalResearch.com Interview with:
Karina Birgitta Berg MD
Department of Ophthalmology
Oslo University Hospital
Oslo, Norway
Medical Research: What is the background for this study? What are the main findings?
Dr. Berg: Neovascular age-related macular degeneration (nAMD) has been the leading cause of vision loss in the elderly population of Western countries. Inhibition of vascular endothelial growth factor (VEGF) with medications such as bevacizumab and ranibizumab injected into the eye, has dramatically reduced the incidence of social blindness from this disease.  Dr. McKay[/caption]
MedicalResearch.com Interview with:
Brian S. McKay, Ph.D
Associate Professor
Department of Ophthalmology and Vision Science
University of Arizona
Medical Research Building, Room 212
Tucson, AZ 85724
Medical Research: What is the background for this study?
Dr. McKay: AMD (age-related macular degeneration) is a disease that is race-related. White people get the disease and lose vision to AMD at much higher rate than Blacks or Hispanics.
Thus, while race is complex, pigmentation may protect from the disease. With this starting point, my laboratory went after the pigmentation pathway to determine how pigment may affect photoreceptor (the retinal cells that actually catch the light) survival. The pigmented cells in the back of the eye are the retinal pigment epithelial cells (RPE), the rest of the retina does not pigment, it is clear not brown. We discovered that when the RPE make pigment they turn on molecular pathways to foster photoreceptor survival. Next we discovered the ligand for a receptor on the RPE that was tied to governing photoreceptor survival and pigmentation. That ligand was L-DOPA.
Knowing that L-DOPA is given to many aging individuals (those at risk of AMD), we developed a team to ask whether those taking L-DOPA for movement disorders are protected from AMD.
Dr. McKay[/caption]
MedicalResearch.com Interview with:
Brian S. McKay, Ph.D
Associate Professor
Department of Ophthalmology and Vision Science
University of Arizona
Medical Research Building, Room 212
Tucson, AZ 85724
Medical Research: What is the background for this study?
Dr. McKay: AMD (age-related macular degeneration) is a disease that is race-related. White people get the disease and lose vision to AMD at much higher rate than Blacks or Hispanics.
Thus, while race is complex, pigmentation may protect from the disease. With this starting point, my laboratory went after the pigmentation pathway to determine how pigment may affect photoreceptor (the retinal cells that actually catch the light) survival. The pigmented cells in the back of the eye are the retinal pigment epithelial cells (RPE), the rest of the retina does not pigment, it is clear not brown. We discovered that when the RPE make pigment they turn on molecular pathways to foster photoreceptor survival. Next we discovered the ligand for a receptor on the RPE that was tied to governing photoreceptor survival and pigmentation. That ligand was L-DOPA.
Knowing that L-DOPA is given to many aging individuals (those at risk of AMD), we developed a team to ask whether those taking L-DOPA for movement disorders are protected from AMD.





