Author Interviews, Gastrointestinal Disease, JAMA, Microbiome / 16.01.2019
Ulcerative colitis: Fecal Microbiota Transplantation Can Induce Remission
MedicalResearch.com Interview with:
[caption id="attachment_46964" align="alignleft" width="200"] Dr. Costello[/caption]
Samuel P. Costello MBBS
Inflammatory Bowel Disease Service,
Department of Gastroenterology
The Queen Elizabeth Hospital
Australia
MedicalResearch.com: What is the background for this study?
Response: Ulcerative colitis (UC) is an inflammatory bowel disease that has high rates of persistent or relapsing symptoms despite available therapies. Many of these therapies also have the potential for unacceptable side effects including allergy, intolerance, serious infection and malignancy due to long-term immunosuppression. It is for these reasons that new therapies for Ulcerative colitis are required; particularly therapies that target novel pathways and are not immune suppressing.
Dr. Costello[/caption]
Samuel P. Costello MBBS
Inflammatory Bowel Disease Service,
Department of Gastroenterology
The Queen Elizabeth Hospital
Australia
MedicalResearch.com: What is the background for this study?
Response: Ulcerative colitis (UC) is an inflammatory bowel disease that has high rates of persistent or relapsing symptoms despite available therapies. Many of these therapies also have the potential for unacceptable side effects including allergy, intolerance, serious infection and malignancy due to long-term immunosuppression. It is for these reasons that new therapies for Ulcerative colitis are required; particularly therapies that target novel pathways and are not immune suppressing.
 Dr. Costello[/caption]
Samuel P. Costello MBBS
Inflammatory Bowel Disease Service,
Department of Gastroenterology
The Queen Elizabeth Hospital
Australia
MedicalResearch.com: What is the background for this study?
Response: Ulcerative colitis (UC) is an inflammatory bowel disease that has high rates of persistent or relapsing symptoms despite available therapies. Many of these therapies also have the potential for unacceptable side effects including allergy, intolerance, serious infection and malignancy due to long-term immunosuppression. It is for these reasons that new therapies for Ulcerative colitis are required; particularly therapies that target novel pathways and are not immune suppressing.
Dr. Costello[/caption]
Samuel P. Costello MBBS
Inflammatory Bowel Disease Service,
Department of Gastroenterology
The Queen Elizabeth Hospital
Australia
MedicalResearch.com: What is the background for this study?
Response: Ulcerative colitis (UC) is an inflammatory bowel disease that has high rates of persistent or relapsing symptoms despite available therapies. Many of these therapies also have the potential for unacceptable side effects including allergy, intolerance, serious infection and malignancy due to long-term immunosuppression. It is for these reasons that new therapies for Ulcerative colitis are required; particularly therapies that target novel pathways and are not immune suppressing.


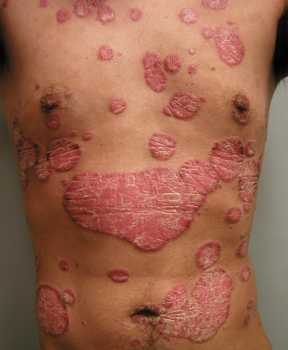
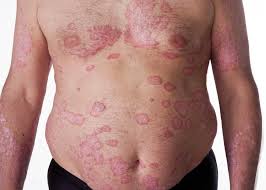 Psoriasis[/caption]
Prof Ching-Chi Chi, MD, MMS, DPhil
Department of Dermatology
Chang Gung Memorial Hospital, Linkou
Guishan Dist, Taoyuan
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Previous studies have shown common genotypes, clinical course, and immunological features shared by psoriasis and inflammatory bowel disease.
However, the relationship between psoriasis and inflammatory bowel disease was largely unclear.
In this study, we found when compared to the general population, psoriatic patients are more likely to have concomitant inflammatory bowel disease.
Psoriasis[/caption]
Prof Ching-Chi Chi, MD, MMS, DPhil
Department of Dermatology
Chang Gung Memorial Hospital, Linkou
Guishan Dist, Taoyuan
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Previous studies have shown common genotypes, clinical course, and immunological features shared by psoriasis and inflammatory bowel disease.
However, the relationship between psoriasis and inflammatory bowel disease was largely unclear.
In this study, we found when compared to the general population, psoriatic patients are more likely to have concomitant inflammatory bowel disease. 










 Dr. William Sandborn[/caption]
William J. Sandborn, MD
Professor of Medicine and Adjunct Professor of Surgery
Chief, Division of Gastroenterology
Director, UCSD IBD Center
University of California San Diego and UC San Diego Health System
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Sandborn: The Phase 3 IM-UNITI study investigated the efficacy and safety of Stelara (ustekinumab) in the treatment of moderate to severe Crohn’s disease as an every 8 or 12 week maintenance therapy.
The study showed a significant proportion of adults with moderate to severe Crohn’s disease who received Stelara maintenance treatment achieved clinical remission.
Dr. William Sandborn[/caption]
William J. Sandborn, MD
Professor of Medicine and Adjunct Professor of Surgery
Chief, Division of Gastroenterology
Director, UCSD IBD Center
University of California San Diego and UC San Diego Health System
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Sandborn: The Phase 3 IM-UNITI study investigated the efficacy and safety of Stelara (ustekinumab) in the treatment of moderate to severe Crohn’s disease as an every 8 or 12 week maintenance therapy.
The study showed a significant proportion of adults with moderate to severe Crohn’s disease who received Stelara maintenance treatment achieved clinical remission.


