MedicalResearch.com Interview with:
[caption id="attachment_59510" align="alignleft" width="200"]

Dr. Mak[/caption]
Raymond H. Mak, MD
Radiation Oncology Disease Center Leader for Thoracic Oncology
Director of Patient Safety and QualityDirector of Clinical Innovation
Associate Professor, Harvard Medical School
Cancer - Radiation Oncology, Radiation Oncology
Department of Radiation Oncology
Brigham and Women's Hospital
MedicalResearch.com: What is the background for this study? What is the algorithm detecting?
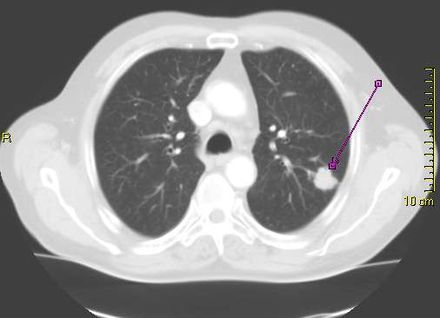
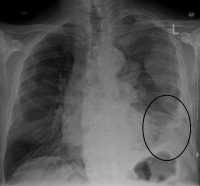
Response: Lung cancer, the most common cancer worldwide is highly lethal, but can be treated and cured in some cases with radiation therapy. Nearly half of lung cancer patients will eventually require some form of radiation therapy, but the planning for a course of radiation therapy currently entails manual, time-consuming, and resource-intensive work by highly trained physicians to segment (target) the cancerous tumors in the lungs and adjacent lymph nodes on three-dimensional images (CT scans). Prior studies have shown substantial variation in how expert clinicians delineate these targets, which can negatively impact outcomes and there is a projected shortage of skilled medical staff to perform these tasks worldwide as cancer rates increase.
To address this critical gap, our team developed deep learning algorithms that can automatically target lung cancer in the lungs and adjacent lymph nodes from CT scans that are used for radiation therapy planning, and can be deployed in seconds.
We trained these artificial intelligence (AI) algorithms using expert-segmented targets from over 700 cases and validated the performance in over 1300 patients in external datasets (including publicly available data from a national trial), benchmarked its performance against expert clinicians, and then further validated the clinical usefulness of the algorithm in human-AI collaboration experiments that measured accuracy, task speed, and end-user satisfaction.



 Dr. Campanella[/caption]
Gabriele Campanella, PhD
Assistant Professor
Windreich Department of Artificial Intelligence and Human Health
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study?
Response: Lung cancer is the most lethal cancer in the US. Lung adenocarcinoma (LUAD) is the most common form of lung cancer with an incidence of over 100k per year in the US. EGFR mutations are common driver mutations in LUAD, and importantly, these mutations can be targeted by TKI therapy, which has high response rates. Because of this, EGFR testing via NGS (Next Generation Sequencing) is considered mandatory by guidelines for any LUAD diagnosis.
In high-resource settings, rapid EGFR testing is done while waiting for confirmation via NGS. This is because NGS takes about 2 weeks on average, while the rapid testing has a median TAT of 2 days. Early treatment decisions could be made based on the rapid test results. Rapid tests have some important drawbacks, most notably, it exhausts tissue. In lung cancer, tissue is scarce in the first place, and up to 25% of cases, after rapid testing there is not enough tissue for NGS. In those circumstances, patients have to be biopsied again, which adds unnecessary risk for the patient. Even worse, in some cases, the NGS is never done. A non-tissue-exhaustive computational biomarker could be used instead of the tissue-based rapid test.
Dr. Campanella[/caption]
Gabriele Campanella, PhD
Assistant Professor
Windreich Department of Artificial Intelligence and Human Health
Icahn School of Medicine at Mount Sinai
MedicalResearch.com: What is the background for this study?
Response: Lung cancer is the most lethal cancer in the US. Lung adenocarcinoma (LUAD) is the most common form of lung cancer with an incidence of over 100k per year in the US. EGFR mutations are common driver mutations in LUAD, and importantly, these mutations can be targeted by TKI therapy, which has high response rates. Because of this, EGFR testing via NGS (Next Generation Sequencing) is considered mandatory by guidelines for any LUAD diagnosis.
In high-resource settings, rapid EGFR testing is done while waiting for confirmation via NGS. This is because NGS takes about 2 weeks on average, while the rapid testing has a median TAT of 2 days. Early treatment decisions could be made based on the rapid test results. Rapid tests have some important drawbacks, most notably, it exhausts tissue. In lung cancer, tissue is scarce in the first place, and up to 25% of cases, after rapid testing there is not enough tissue for NGS. In those circumstances, patients have to be biopsied again, which adds unnecessary risk for the patient. Even worse, in some cases, the NGS is never done. A non-tissue-exhaustive computational biomarker could be used instead of the tissue-based rapid test.


 Dr. Han[/caption]
Summer S Han, PhD
Associate Professor
Dr. Han[/caption]
Summer S Han, PhD
Associate Professor
 Dr. Choi[/caption]
Dr. Eunji Choi PhD
Instructor, Neurosurgery
Department: Adult Neurosurgery
Stanford University School of Medicine
MedicalResearch.com: What is the background for this study?
Dr. Choi[/caption]
Dr. Eunji Choi PhD
Instructor, Neurosurgery
Department: Adult Neurosurgery
Stanford University School of Medicine
MedicalResearch.com: What is the background for this study?

 Dr. Lova Sun[/caption]
Lova L. Sun, MD, MSCE
Medical Oncology
Assistant Professor of Medicine
Hospital of the University of Pennsylvania
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: An common clinical question for patients with metastatic non-small cell lung cancer with long-term response to immunotherapy-based treatment is how long to continue treatment. The major clinical trials stopped immunotherapy at a maximum of 2 years, but in clinical practice many patients and clinicians continue treatment beyond this time point.
We conducted a retrospective study of lung cancer patients across the US with long-term response to immunotherapy, to compare survival between those who stopped treatment at 2 years vs those who continued beyond 2 years. We found that there was no statistically significant difference in survival between the two groups.
Dr. Lova Sun[/caption]
Lova L. Sun, MD, MSCE
Medical Oncology
Assistant Professor of Medicine
Hospital of the University of Pennsylvania
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: An common clinical question for patients with metastatic non-small cell lung cancer with long-term response to immunotherapy-based treatment is how long to continue treatment. The major clinical trials stopped immunotherapy at a maximum of 2 years, but in clinical practice many patients and clinicians continue treatment beyond this time point.
We conducted a retrospective study of lung cancer patients across the US with long-term response to immunotherapy, to compare survival between those who stopped treatment at 2 years vs those who continued beyond 2 years. We found that there was no statistically significant difference in survival between the two groups.

 Dr. Sally Lau[/caption]
Dr. Sally Lau MD
Medical oncologist, NYU Langone’s Perlmutter Cancer Center
Assistant professor of medicine
NYU Grossman School of Medicine
MedicalResearch.com: How big is the problem of
Dr. Sally Lau[/caption]
Dr. Sally Lau MD
Medical oncologist, NYU Langone’s Perlmutter Cancer Center
Assistant professor of medicine
NYU Grossman School of Medicine
MedicalResearch.com: How big is the problem of 


 Dr. Ulhas Nair[/caption]
Nishanth Ulhas Nair, Ph.D.
Affiliation: Staff Scientist at Cancer Data Science Laboratory, Center for Cancer Research
National Cancer Institute (NCI), National Institutes of Health (NIH)
Bethesda, Maryland, USA. Date: April 22, 2021
Dr. Raffit Hassan and Dr. Eytan Ruppin at the National Cancer Institute (NCI) are the senior authors of this study.
MedicalResearch.com: What is the background for this study?
Response: Malignant mesothelioma is an aggressive cancer with limited treatment options and poor prognosis. An in-depth knowledge of genetic, transcriptomic and immunogenic events involved in mesothelioma is critical for successful development of prognostics and therapeutic modalities. In this study we aim to address this by exploring a new large scale patient tumor dataset of 122 mesothelioma patients, called NCI mesothelioma patient data, along with their genomic, transcriptomic, and phenotypic information. Unlike previous large-scale studies which have been focused on malignant pleural mesothelioma patients, our dataset contains an approximately equal representation of malignant pleural and peritoneal mesothelioma patients which allows to identify any differences between them.
Dr. Ulhas Nair[/caption]
Nishanth Ulhas Nair, Ph.D.
Affiliation: Staff Scientist at Cancer Data Science Laboratory, Center for Cancer Research
National Cancer Institute (NCI), National Institutes of Health (NIH)
Bethesda, Maryland, USA. Date: April 22, 2021
Dr. Raffit Hassan and Dr. Eytan Ruppin at the National Cancer Institute (NCI) are the senior authors of this study.
MedicalResearch.com: What is the background for this study?
Response: Malignant mesothelioma is an aggressive cancer with limited treatment options and poor prognosis. An in-depth knowledge of genetic, transcriptomic and immunogenic events involved in mesothelioma is critical for successful development of prognostics and therapeutic modalities. In this study we aim to address this by exploring a new large scale patient tumor dataset of 122 mesothelioma patients, called NCI mesothelioma patient data, along with their genomic, transcriptomic, and phenotypic information. Unlike previous large-scale studies which have been focused on malignant pleural mesothelioma patients, our dataset contains an approximately equal representation of malignant pleural and peritoneal mesothelioma patients which allows to identify any differences between them.