The Pharmacokinetics of the Last Mile: Protecting Cannabinoid Integrity in 2026
 Source: IMAGE[/caption]
Source: IMAGE[/caption]
Editor's note: Cannabis and THCA/Hemp CBD products should have an active ingredient list on the container and a Certificate of Analysis (COA). Discuss your use of THC, cannabis, or CBD products with your health care provider. Dosing of cannabis products is variable, especially since they are not FDA regulated. Cannabis/CBD may interfere with other medications and should not be used in individuals with certain health conditions, including liver issues. CBD skin care products can be absorbed through the skin and have similar effects. Do not use cannabis products including edibles, drinks, and CBD if you are pregnant, nursing, or may become pregnant. Do not use cannabis products if driving or operating difficult or dangerous machinery. Children should not be exposed to cannabis or CBD products.
Whereas logistics professionals are more likely to concentrate on the purity and quality of laboratory extractions or cultivations, one of the most unstable parts of the medicinal substance's route to delivery — the final mile — may sometimes be neglected. The cannabis industry has turned to a pharmaceutical approach in 2026 to safeguard the chemical composition of the plant from exposure to damaging effects before reaching the end user.







 Dr. Kruger[/caption]
MedicalResearch.com: What is the background for this study?
[caption id="attachment_72137" align="alignleft" width="100"]
Dr. Kruger[/caption]
MedicalResearch.com: What is the background for this study?
[caption id="attachment_72137" align="alignleft" width="100"] Dr. Felicione[/caption]
Response: Alcohol consumption, especially heavy alcohol consumption, is associated with many health risks and nearly 200 different health conditions and diseases. Reducing alcohol consumption reduces the risks and harms from alcohol. Previous research has demonstrated that people have reduced their alcohol consumption when they have access to cannabis. Cannabis beverages have emerged in States where cannabis is legal for adult or medical use.
Dr. Felicione[/caption]
Response: Alcohol consumption, especially heavy alcohol consumption, is associated with many health risks and nearly 200 different health conditions and diseases. Reducing alcohol consumption reduces the risks and harms from alcohol. Previous research has demonstrated that people have reduced their alcohol consumption when they have access to cannabis. Cannabis beverages have emerged in States where cannabis is legal for adult or medical use.





 Dr. Dunbar[/caption]
MedicalResearch.com Interview with:
Michael S. Dunbar, PhD
Dr. Dunbar[/caption]
MedicalResearch.com Interview with:
Michael S. Dunbar, PhD














 Elena Stains[/caption]
Elena Stains
Medical Student
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: In 2019 to 2020, 2.5% of Americans reported using cannabis for medical needs, compared to 1.2% in 2013-2014, representing a 12.9% annual increase1. Forty states and the District of Columbia have legislation for some form of medical cannabis (MC) in 2024. Because MC is not federally legalized, each state creates its own legislation on the conditions that qualify a person for MC, without any standardized process to determine what qualifying conditions (QC) are proven to be aided by MC. Thus, the QCs chosen by states vary widely. Common QCs include cancer, dementia, and PTSD.
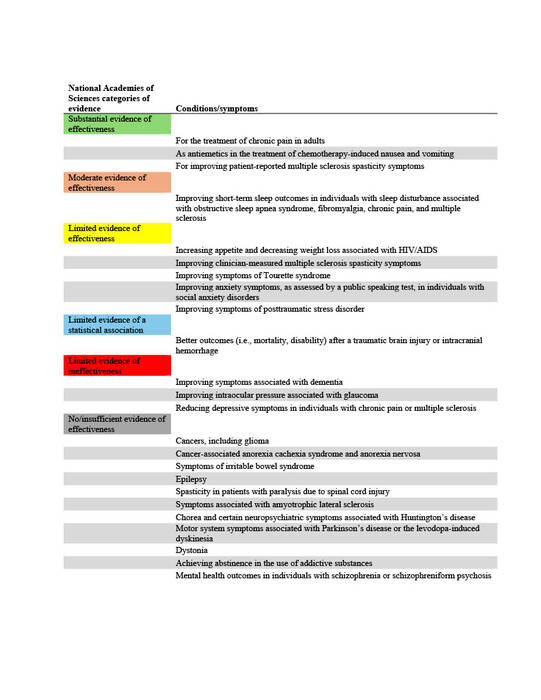
The National Academies of Sciences, Engineering, and Medicine (NAS) published a report in 2017 on the evidence for the therapeutic effects of cannabis and cannabinoids for over twenty conditions2. This report reviews the evidence of effectiveness of medical cannabis for the most common QCs chosen by states. The researchers at Geisinger Commonwealth School of Medicine aimed to compare the evidence found by the NAS report with the QCs of 38 states (including the District of Columbia) in both 2017 and 2024. QCs were categorized based on NAS-established level of evidence: limited, moderate, or substantial/conclusive evidence of effectiveness, limited evidence of ineffectiveness, or no/insufficient evidence to support or refute effectiveness (Table 1).
Elena Stains[/caption]
Elena Stains
Medical Student
Department of Medical Education
Geisinger Commonwealth School of Medicine
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: In 2019 to 2020, 2.5% of Americans reported using cannabis for medical needs, compared to 1.2% in 2013-2014, representing a 12.9% annual increase1. Forty states and the District of Columbia have legislation for some form of medical cannabis (MC) in 2024. Because MC is not federally legalized, each state creates its own legislation on the conditions that qualify a person for MC, without any standardized process to determine what qualifying conditions (QC) are proven to be aided by MC. Thus, the QCs chosen by states vary widely. Common QCs include cancer, dementia, and PTSD.
The National Academies of Sciences, Engineering, and Medicine (NAS) published a report in 2017 on the evidence for the therapeutic effects of cannabis and cannabinoids for over twenty conditions2. This report reviews the evidence of effectiveness of medical cannabis for the most common QCs chosen by states. The researchers at Geisinger Commonwealth School of Medicine aimed to compare the evidence found by the NAS report with the QCs of 38 states (including the District of Columbia) in both 2017 and 2024. QCs were categorized based on NAS-established level of evidence: limited, moderate, or substantial/conclusive evidence of effectiveness, limited evidence of ineffectiveness, or no/insufficient evidence to support or refute effectiveness (Table 1).


