Author Interviews, Dental Research, JAMA, Pediatrics, USPSTF / 10.12.2021
USPSTF: Importance of Preventing Dental Caries in Young Children
MedicalResearch.com Interview with:
[caption id="attachment_58505" align="alignleft" width="150"] Dr. Cabana[/caption]
Michael Cabana, M.D., M.A., M.P.H
Professor of Pediatrics
Albert Einstein College of Medicine.
Physician-in-chief , Children's Hospital at Montefiore
Chair of the Department of Pediatrics
Albert Einstein College of Medicine
Member, U.S. Preventive Services Task Force
MedicalResearch.com: What is the background for this study and recommendation statement?
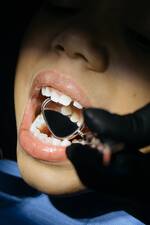
Response: Dental caries, also known as cavities or tooth decay, is the most common chronic disease in children in the United States and can develop in any child whose teeth have come in. Many young children under five years old do not visit a dentist, so the Task Force reviewed the latest evidence on how primary care clinicians can help prevent tooth decay in young children.
The Task Force’s research led to two important findings: all young children whose teeth have come in should have fluoride varnish applied by their clinician, and all children six months and older whose water supply doesn’t contain enough fluoride should receive fluoride supplements. Both approaches can help prevent cavities in kids.
The Task Force also determined that there is not enough evidence to recommend for or against screening for tooth decay in the primary care setting for children under five. This is consistent with the Task Force’s 2014 recommendation on dental caries.
Dr. Cabana[/caption]
Michael Cabana, M.D., M.A., M.P.H
Professor of Pediatrics
Albert Einstein College of Medicine.
Physician-in-chief , Children's Hospital at Montefiore
Chair of the Department of Pediatrics
Albert Einstein College of Medicine
Member, U.S. Preventive Services Task Force
MedicalResearch.com: What is the background for this study and recommendation statement?
Response: Dental caries, also known as cavities or tooth decay, is the most common chronic disease in children in the United States and can develop in any child whose teeth have come in. Many young children under five years old do not visit a dentist, so the Task Force reviewed the latest evidence on how primary care clinicians can help prevent tooth decay in young children.
The Task Force’s research led to two important findings: all young children whose teeth have come in should have fluoride varnish applied by their clinician, and all children six months and older whose water supply doesn’t contain enough fluoride should receive fluoride supplements. Both approaches can help prevent cavities in kids.
The Task Force also determined that there is not enough evidence to recommend for or against screening for tooth decay in the primary care setting for children under five. This is consistent with the Task Force’s 2014 recommendation on dental caries.
 Dr. Cabana[/caption]
Michael Cabana, M.D., M.A., M.P.H
Professor of Pediatrics
Albert Einstein College of Medicine.
Physician-in-chief , Children's Hospital at Montefiore
Chair of the Department of Pediatrics
Albert Einstein College of Medicine
Member, U.S. Preventive Services Task Force
MedicalResearch.com: What is the background for this study and recommendation statement?
Response: Dental caries, also known as cavities or tooth decay, is the most common chronic disease in children in the United States and can develop in any child whose teeth have come in. Many young children under five years old do not visit a dentist, so the Task Force reviewed the latest evidence on how primary care clinicians can help prevent tooth decay in young children.
The Task Force’s research led to two important findings: all young children whose teeth have come in should have fluoride varnish applied by their clinician, and all children six months and older whose water supply doesn’t contain enough fluoride should receive fluoride supplements. Both approaches can help prevent cavities in kids.
The Task Force also determined that there is not enough evidence to recommend for or against screening for tooth decay in the primary care setting for children under five. This is consistent with the Task Force’s 2014 recommendation on dental caries.
Dr. Cabana[/caption]
Michael Cabana, M.D., M.A., M.P.H
Professor of Pediatrics
Albert Einstein College of Medicine.
Physician-in-chief , Children's Hospital at Montefiore
Chair of the Department of Pediatrics
Albert Einstein College of Medicine
Member, U.S. Preventive Services Task Force
MedicalResearch.com: What is the background for this study and recommendation statement?
Response: Dental caries, also known as cavities or tooth decay, is the most common chronic disease in children in the United States and can develop in any child whose teeth have come in. Many young children under five years old do not visit a dentist, so the Task Force reviewed the latest evidence on how primary care clinicians can help prevent tooth decay in young children.
The Task Force’s research led to two important findings: all young children whose teeth have come in should have fluoride varnish applied by their clinician, and all children six months and older whose water supply doesn’t contain enough fluoride should receive fluoride supplements. Both approaches can help prevent cavities in kids.
The Task Force also determined that there is not enough evidence to recommend for or against screening for tooth decay in the primary care setting for children under five. This is consistent with the Task Force’s 2014 recommendation on dental caries.


 Dr. Frost[/caption]
Holly Frost, MD
Assistant Professor Pediatrics
University of Colorado Anschutz School of Medicine
[caption id="attachment_58185" align="alignleft" width="150"]
Dr. Frost[/caption]
Holly Frost, MD
Assistant Professor Pediatrics
University of Colorado Anschutz School of Medicine
[caption id="attachment_58185" align="alignleft" width="150"]




 Dr. Gary Smith[/caption]
Gary A. Smith, MD, DrPH
Director, Center for Injury Research and Policy
Nationwide Children’s Hospital
Columbus, OH
MedicalResearch.com: What is the background for this study?
Response: Furniture and TV tip-overs are an important source of injury, especially for children younger than 6 years old. Our study found that an estimated 560,200 children younger than 18 years old were treated in U.S. emergency departments for furniture or TV tip-over injuries from 1990 through 2019. In 2019, there were 11,521 injured children, which is an average of one child every 46 minutes.
Dr. Gary Smith[/caption]
Gary A. Smith, MD, DrPH
Director, Center for Injury Research and Policy
Nationwide Children’s Hospital
Columbus, OH
MedicalResearch.com: What is the background for this study?
Response: Furniture and TV tip-overs are an important source of injury, especially for children younger than 6 years old. Our study found that an estimated 560,200 children younger than 18 years old were treated in U.S. emergency departments for furniture or TV tip-over injuries from 1990 through 2019. In 2019, there were 11,521 injured children, which is an average of one child every 46 minutes.

 Dr. Forbes[/caption]
Lisa Forbes, Ph.D, LPC, NCC
Clinical Assistant Professor
Counseling Program
University of Colorado Denver
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: The most common mode of learning in tertiary education is lecture-based learning despite the knowledge that more active, engaged, and flexible approaches to teaching may better support the learning process. This study aimed to understand graduate students’ experiences with a playful pedagogy as an alternative approach to learning.
Dr. Forbes[/caption]
Lisa Forbes, Ph.D, LPC, NCC
Clinical Assistant Professor
Counseling Program
University of Colorado Denver
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: The most common mode of learning in tertiary education is lecture-based learning despite the knowledge that more active, engaged, and flexible approaches to teaching may better support the learning process. This study aimed to understand graduate students’ experiences with a playful pedagogy as an alternative approach to learning.
 Dr. Hoberman[/caption]
Alejandro Hoberman, M.D.
Vice Chair of Clinical Research, Division Director, General Academic Pediatrics, and Professor of Pediatrics and Clinical and Translational Science
Jack L. Paradise, MD Endowed Professor of Pediatric Research, UPMC Children's Hospital of PittsburghPresident, UPMC Children's Community Pediatrics
MedicalResearch.com: What is the background for this study?
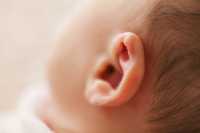
Response: Acute otitis media (AOM) is the most frequently diagnosed illness in children in the United States for which antibiotics are prescribed. Recurrent AOM is the principal indication for tympanostomy-tube placement, the most frequently performed operation in children after the newborn period. Supporting the performance of tympanostomy-tube placement for recurrent acute otitis media has been the commonplace observation, after surgery, of acute otitis media–free periods of varying duration. Counterbalancing this view have been the cost of tympanostomy-tube placement; risks and possible late sequelae of anesthesia in young children; the possible occurrence of refractory tube otorrhea, tube blockage, premature extrusion, or dislocation of the tube into the middle-ear cavity; various structural tympanic membrane sequelae; and the possible development of mild conductive hearing loss. Tempering support for surgery is the progressive reduction in the incidence of acute otitis media that usually accompanies a child’s increasing age.
Previous trials of tympanostomy-tube placement for recurrent acute otitis media, all conducted before the introduction of pneumococcal conjugate vaccine, have given mixed results and were limited, variously, by small sample size, uncertain validity of diagnoses of acute otitis media determining trial eligibility, short periods of follow-up, and substantial attrition of participants. Official recommendations regarding tympanostomy-tube placement for children with recurrent acute otitis media differ — an otolaryngologic guideline recommends the procedure for children with recurrent acute otitis media, provided that middle-ear effusion is present in at least one ear; a contemporaneous pediatric guideline discusses tympanostomy-tube placement as an “option [that] clinicians may offer.”
Given these uncertainties, we undertook the present trial involving children 6 to 35 months of age who had a history of recurrent acute otitis media to determine whether tympanostomy-tube placement, as compared with medical management (comprising episodic antimicrobial treatment, with the option of tympanostomy-tube placement in the event of treatment failure), would result in a greater reduction in the children’s rate of recurrence of acute otitis media during the ensuing 2-year period.
Dr. Hoberman[/caption]
Alejandro Hoberman, M.D.
Vice Chair of Clinical Research, Division Director, General Academic Pediatrics, and Professor of Pediatrics and Clinical and Translational Science
Jack L. Paradise, MD Endowed Professor of Pediatric Research, UPMC Children's Hospital of PittsburghPresident, UPMC Children's Community Pediatrics
MedicalResearch.com: What is the background for this study?
Response: Acute otitis media (AOM) is the most frequently diagnosed illness in children in the United States for which antibiotics are prescribed. Recurrent AOM is the principal indication for tympanostomy-tube placement, the most frequently performed operation in children after the newborn period. Supporting the performance of tympanostomy-tube placement for recurrent acute otitis media has been the commonplace observation, after surgery, of acute otitis media–free periods of varying duration. Counterbalancing this view have been the cost of tympanostomy-tube placement; risks and possible late sequelae of anesthesia in young children; the possible occurrence of refractory tube otorrhea, tube blockage, premature extrusion, or dislocation of the tube into the middle-ear cavity; various structural tympanic membrane sequelae; and the possible development of mild conductive hearing loss. Tempering support for surgery is the progressive reduction in the incidence of acute otitis media that usually accompanies a child’s increasing age.
Previous trials of tympanostomy-tube placement for recurrent acute otitis media, all conducted before the introduction of pneumococcal conjugate vaccine, have given mixed results and were limited, variously, by small sample size, uncertain validity of diagnoses of acute otitis media determining trial eligibility, short periods of follow-up, and substantial attrition of participants. Official recommendations regarding tympanostomy-tube placement for children with recurrent acute otitis media differ — an otolaryngologic guideline recommends the procedure for children with recurrent acute otitis media, provided that middle-ear effusion is present in at least one ear; a contemporaneous pediatric guideline discusses tympanostomy-tube placement as an “option [that] clinicians may offer.”
Given these uncertainties, we undertook the present trial involving children 6 to 35 months of age who had a history of recurrent acute otitis media to determine whether tympanostomy-tube placement, as compared with medical management (comprising episodic antimicrobial treatment, with the option of tympanostomy-tube placement in the event of treatment failure), would result in a greater reduction in the children’s rate of recurrence of acute otitis media during the ensuing 2-year period.

 Dr. Bullard[/caption]
Jared Bullard MD FRCPC
Associate Professor, Departments of Pediatrics & Child Health and Medical Microbiology & Infectious Diseases
Max Rady College of Medicine
Rady Faculty of Health Sciences
Cadham Provincial Laboratory
Children’s Hospital Research Institute of Manitoba
MedicalResearch.com: What is the background for this study?
Response: Children are well known to transmit epidemic/endemic respiratory viruses like influenza. Initial public health policy was based on that children were likely to transmit SARS-CoV-2 effectively within a community and subsequently in-person school and extracurricular activities were suspended.
Initial research did not show a clear association with children driving transmission. The purpose of our study was to take respiratory samples from both children and adults with COVID-19 (all had SARS-CoV-2 detected by RT-PCR) and compare those samples by their ability to grow in cell culture and amount of virus in samples.
We took 175 samples from children (97 younger than 10 years of age and 78 between 11-17 years) and compared them to 130 adult samples from the same communities in Manitoba experiencing outbreaks of COVID-19.
Dr. Bullard[/caption]
Jared Bullard MD FRCPC
Associate Professor, Departments of Pediatrics & Child Health and Medical Microbiology & Infectious Diseases
Max Rady College of Medicine
Rady Faculty of Health Sciences
Cadham Provincial Laboratory
Children’s Hospital Research Institute of Manitoba
MedicalResearch.com: What is the background for this study?
Response: Children are well known to transmit epidemic/endemic respiratory viruses like influenza. Initial public health policy was based on that children were likely to transmit SARS-CoV-2 effectively within a community and subsequently in-person school and extracurricular activities were suspended.
Initial research did not show a clear association with children driving transmission. The purpose of our study was to take respiratory samples from both children and adults with COVID-19 (all had SARS-CoV-2 detected by RT-PCR) and compare those samples by their ability to grow in cell culture and amount of virus in samples.
We took 175 samples from children (97 younger than 10 years of age and 78 between 11-17 years) and compared them to 130 adult samples from the same communities in Manitoba experiencing outbreaks of COVID-19. 