Author Interviews, Ovarian Cancer / 04.11.2024
Mt. Sinai Study of Ovarian Cancer Finds Factor That Promotes Immunotherapy Resistance
MedicalResearch.com Interview with:
[caption id="attachment_64543" align="alignleft" width="114"] Dr .Baccarini[/caption]
Alessia Baccarini, PhD
Assistant Professor
Dept. of Immunology and Immunotherapy (DII)
Icahn School of Medicine at Mount Sinai
New York, New York 10029
MedicalResearch.com: What is the background for this study?
Response: Ovarian cancer is characterized by a complex tumor microenvironment (TME) that significantly contributes to resistance against immunotherapy, particularly immune checkpoint blockade (ICB) therapies like anti-PD-1. Understanding the extracellular signals—such as cytokines and chemokines—that ovarian cancer cells utilize to create an immunosuppressive TME is critical for improving treatment outcomes.
Our research focuses on elucidating how these signaling factors contribute to tumor growth and immune evasion. We utilized a novel genomic functional approach called Perturb-map to study intratumoral heterogeneity (ITH) in ovarian cancer within a mouse model, allowing us to investigate the communication between ovarian cancer cells and immune cells.
Dr .Baccarini[/caption]
Alessia Baccarini, PhD
Assistant Professor
Dept. of Immunology and Immunotherapy (DII)
Icahn School of Medicine at Mount Sinai
New York, New York 10029
MedicalResearch.com: What is the background for this study?
Response: Ovarian cancer is characterized by a complex tumor microenvironment (TME) that significantly contributes to resistance against immunotherapy, particularly immune checkpoint blockade (ICB) therapies like anti-PD-1. Understanding the extracellular signals—such as cytokines and chemokines—that ovarian cancer cells utilize to create an immunosuppressive TME is critical for improving treatment outcomes.
Our research focuses on elucidating how these signaling factors contribute to tumor growth and immune evasion. We utilized a novel genomic functional approach called Perturb-map to study intratumoral heterogeneity (ITH) in ovarian cancer within a mouse model, allowing us to investigate the communication between ovarian cancer cells and immune cells.
 Dr .Baccarini[/caption]
Alessia Baccarini, PhD
Assistant Professor
Dept. of Immunology and Immunotherapy (DII)
Icahn School of Medicine at Mount Sinai
New York, New York 10029
MedicalResearch.com: What is the background for this study?
Response: Ovarian cancer is characterized by a complex tumor microenvironment (TME) that significantly contributes to resistance against immunotherapy, particularly immune checkpoint blockade (ICB) therapies like anti-PD-1. Understanding the extracellular signals—such as cytokines and chemokines—that ovarian cancer cells utilize to create an immunosuppressive TME is critical for improving treatment outcomes.
Our research focuses on elucidating how these signaling factors contribute to tumor growth and immune evasion. We utilized a novel genomic functional approach called Perturb-map to study intratumoral heterogeneity (ITH) in ovarian cancer within a mouse model, allowing us to investigate the communication between ovarian cancer cells and immune cells.
Dr .Baccarini[/caption]
Alessia Baccarini, PhD
Assistant Professor
Dept. of Immunology and Immunotherapy (DII)
Icahn School of Medicine at Mount Sinai
New York, New York 10029
MedicalResearch.com: What is the background for this study?
Response: Ovarian cancer is characterized by a complex tumor microenvironment (TME) that significantly contributes to resistance against immunotherapy, particularly immune checkpoint blockade (ICB) therapies like anti-PD-1. Understanding the extracellular signals—such as cytokines and chemokines—that ovarian cancer cells utilize to create an immunosuppressive TME is critical for improving treatment outcomes.
Our research focuses on elucidating how these signaling factors contribute to tumor growth and immune evasion. We utilized a novel genomic functional approach called Perturb-map to study intratumoral heterogeneity (ITH) in ovarian cancer within a mouse model, allowing us to investigate the communication between ovarian cancer cells and immune cells.

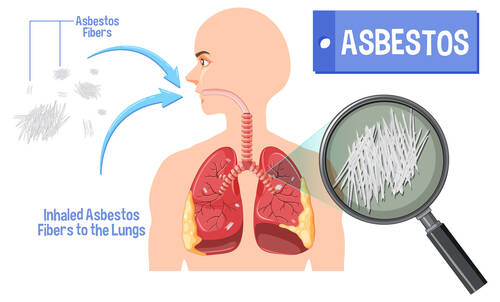








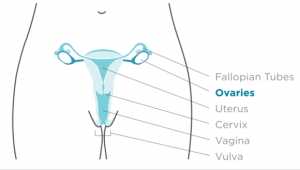
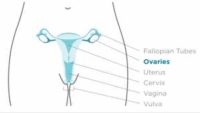










 Dr. Sibaji Sarkar[/caption]
Sibaji Sarkar Ph.D
Instructor of medicine
Boston University School of Medicine
Boston
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Sarkar: Although breast and ovarian cancers have different clinical presentations, there are certain molecular events that are conserved between the two types of cancers. For example, mutation in a few genes, such as BRCA1, BRCA2, is an indicator of possible development of both breast and ovarian cancers. ARHI, a pro-apoptotic imprinted gene is epigenetically silenced in both breast and ovarian cancers. A similar pattern was observed in microRNA as well. There are also several genes which are differentially expressed in these two types of cancers but few of these striking resemblances led us to investigate whether they have a common origin. In this paper, we compared genetic and epigenetic events in both breast and ovarian cancers and we hypothesize that they may have similar origin (mechanism of formation of cancer progenitor cells), which should be regulated by epigenetic mechanism.
Dr. Sibaji Sarkar[/caption]
Sibaji Sarkar Ph.D
Instructor of medicine
Boston University School of Medicine
Boston
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Sarkar: Although breast and ovarian cancers have different clinical presentations, there are certain molecular events that are conserved between the two types of cancers. For example, mutation in a few genes, such as BRCA1, BRCA2, is an indicator of possible development of both breast and ovarian cancers. ARHI, a pro-apoptotic imprinted gene is epigenetically silenced in both breast and ovarian cancers. A similar pattern was observed in microRNA as well. There are also several genes which are differentially expressed in these two types of cancers but few of these striking resemblances led us to investigate whether they have a common origin. In this paper, we compared genetic and epigenetic events in both breast and ovarian cancers and we hypothesize that they may have similar origin (mechanism of formation of cancer progenitor cells), which should be regulated by epigenetic mechanism.

 Dr. Dirk Timmerman[/caption]
Dr. Dirk Timmerman[/caption]
 Dr. Hamanishi[/caption]
MedicalResearch.com Interview with:
Junzo Hamanishi M.D., Ph.D.
Department of Gynecology and Obstetrics,
Kyoto University Graduate School of Medicine
Assistant Professor
Kyoto Japan
Medical Research: What is the background for this study?
Dr. Hamanishi: More than 70% of patients with advanced ovarian cancer who achieve remission ultimately relapse and there are few effective treatments for these patients. Because the development of new treatment strategies for these patients is urgently required, we have focused on and studied the potential of cancer cells to escape from host immunity with PD-1/PD-L1 immunosuppressive signal in the tumor microenvironment to find new treatment strategies to overcome this phenomenon,
Dr. Hamanishi[/caption]
MedicalResearch.com Interview with:
Junzo Hamanishi M.D., Ph.D.
Department of Gynecology and Obstetrics,
Kyoto University Graduate School of Medicine
Assistant Professor
Kyoto Japan
Medical Research: What is the background for this study?
Dr. Hamanishi: More than 70% of patients with advanced ovarian cancer who achieve remission ultimately relapse and there are few effective treatments for these patients. Because the development of new treatment strategies for these patients is urgently required, we have focused on and studied the potential of cancer cells to escape from host immunity with PD-1/PD-L1 immunosuppressive signal in the tumor microenvironment to find new treatment strategies to overcome this phenomenon,  Dr. McDonald[/caption]
MedicalResearch.com Interview with:
Professor John McDonald PhD
Director of its Integrated Cancer Research Center
School of Biology at the Georgia Institute of Technology
Medical Research: What is the background for this study? What are the main findings?
Response: Ovarian cancer is a deadly disease because it cannot be diagnosed at early stages when it can be most effectively effectively treated.
It has long been recognized that there is a great need for an accurate diagnostic test for early stage ovarian cancer.
Until now, efforts to develop a highly accurate way to detect early stage ovarian cancer have been unsuccessful.
We have used a novel approach that integrates advanced methods in analytical chemistry with advanced machine learning algorithms to identify 16 metabolites that collectively can detect ovarian cancer with extremely high accuracy (100% in the samples tested in our study)
Dr. McDonald[/caption]
MedicalResearch.com Interview with:
Professor John McDonald PhD
Director of its Integrated Cancer Research Center
School of Biology at the Georgia Institute of Technology
Medical Research: What is the background for this study? What are the main findings?
Response: Ovarian cancer is a deadly disease because it cannot be diagnosed at early stages when it can be most effectively effectively treated.
It has long been recognized that there is a great need for an accurate diagnostic test for early stage ovarian cancer.
Until now, efforts to develop a highly accurate way to detect early stage ovarian cancer have been unsuccessful.
We have used a novel approach that integrates advanced methods in analytical chemistry with advanced machine learning algorithms to identify 16 metabolites that collectively can detect ovarian cancer with extremely high accuracy (100% in the samples tested in our study)
 Dr. Qin[/caption]
MedicalResearch.com Interview with:
Bo (Bonnie) Qin, PhD
Postdoctoral associate at Rutgers Cancer Institute of New Jersey
Medical Research: What is the background for this study? What are the main findings?
Response: Ovarian cancer is among the top five causes of cancer death among women in the US. Compared to white women, African-American women tend to have a worse 5-year survival rate of ovarian cancer. It highlights a critical need for identifying preventive factors in African Americans, particularly through dietary modification, which is relatively low cost and low risk compared to medical treatments.
We found that adherence to an overall healthy dietary pattern i.e. Alternate Healthy Eating Index (AHEI)-2010 may reduce ovarian cancer risk in African-American women, and particularly among postmenopausal women. Adherence to the current Dietary Guidelines for Americans i.e. Healthy Eating Index-2010, were also strongly associated with reduced risk of ovarian cancer among postmenopausal African-American women.
Dr. Qin[/caption]
MedicalResearch.com Interview with:
Bo (Bonnie) Qin, PhD
Postdoctoral associate at Rutgers Cancer Institute of New Jersey
Medical Research: What is the background for this study? What are the main findings?
Response: Ovarian cancer is among the top five causes of cancer death among women in the US. Compared to white women, African-American women tend to have a worse 5-year survival rate of ovarian cancer. It highlights a critical need for identifying preventive factors in African Americans, particularly through dietary modification, which is relatively low cost and low risk compared to medical treatments.
We found that adherence to an overall healthy dietary pattern i.e. Alternate Healthy Eating Index (AHEI)-2010 may reduce ovarian cancer risk in African-American women, and particularly among postmenopausal women. Adherence to the current Dietary Guidelines for Americans i.e. Healthy Eating Index-2010, were also strongly associated with reduced risk of ovarian cancer among postmenopausal African-American women.