MedicalResearch.com Interview with:
[caption id="attachment_46583" align="alignleft" width="148"]

Dr. Romine[/caption]
Dr. Kristine A. Romine MD
CEO and Founder of Camelback Dermatology & Skin Surgery
Phoenix, AZ
MedicalResearch.com: Would you give a brief overview of the different types of skin cancer?
Response: There are multiple types of skin cancer, including: melanoma, basal cell carcinoma, squamous cell carcinoma, and actinic keratosis. Known as the deadliest form of skin cancer, melanoma develops when irreparable DNA damage results in malignant transformation of melanocytes. This type of skin cancer is most commonly caused by intense UV exposure from the sun or tanning beds, which activate mutations that lead skin cells to rapidly multiply and form malignant tumors. Melanoma can range in color from dark brown to black and are rarely red or even skin colored. They are usually irregular and asymmetrical. In 2018, there were an estimated
91,270 new cases of melanoma (American Cancer Society, 2018).
Basal cell carcinoma (BCC) is the most common type of skin cancer and cancer diagnosed. BCCs arise in the skins’ outermost layers. BCCs resemble open sores, red or pink plaques, pearly nodules with telangiectasia, or scars. It is estimated that 4.3 million BCCs are diagnosed in the U.S. every year (Skin Cancer Foundation, 2018).
Squamous cell carcinoma (SCC), the second most common type of skin cancer, arises from the squamous cells in the skin that have been exposed to UV over long periods of time. SCCs appear as scaly red or pink macules, papules, or plaques. They can be crusted and appear eroded and can commonly arise within a solar keratosis. More than 1 million cases of SCC are diagnosed in the U.S. every year (Skin Cancer Foundation, 2018).
Lastly, actinic keratoses (AKs) are the most common pre-cancerous skin growth that can develop into a SCC if left untreated. Similar to all other types, AKs are caused by exposure to UV light and, in rare cases, high exposures to x-rays. AKs can appear on sun-exposed areas, including the face, scalp, ears, shoulders, and legs. They resemble pink, scaly rough patches on the skin.
 Anne Cust[/caption]
Anne Cust | PhD, Professor of Cancer Epidemiology
The University of Sydney
Faculty of Medicine and Health
Sydney School of Public Health
MedicalResearch.com: What is the background for this study? Are the screeners specially trained, use full body photographs, dermoscopy etc?
Response: The Melanoma High-risk Clinic Study was developed to optimise the early detection of new melanomas in people at high risk of developing melanoma. A previous single-centre study observed fewer excisions and healthcare costs, thinner melanomas and better quality of life when surveillance of high risk patients was conducted in a melanoma dermatology clinic with a structured surveillance protocol involving 6-monthly full body examinations aided by total body photography (TBP) and sequential digital dermoscopy imaging (SDDI). The initial pilot study was performed in a single tertiary referral specialist centre using trained dermatologists who routinely used the diagnostic interventions.
Our objective was to examine longer-term sustainability and expansion of the program to multiple practices including a primary care skin cancer clinic setting. The hypothesis was that the outcomes would be similar if using the same protocol and diagnostic tools. The participating doctors were trained to follow the protocol, which included instruction on how to respond and interpret changing lesions, but not in use of dermoscopy or skin examinations, which were routinely and consistently used in all clinics prior to the study commencing. There were 593 participants assessed as very high risk of melanoma who participated in the Melanoma High-risk Clinic Study from 2012-2018. Nearly all of the participants had had a previous melanoma and had additional melanoma risk factors. 57% were male and the median age at study entry was 58 years.
Anne Cust[/caption]
Anne Cust | PhD, Professor of Cancer Epidemiology
The University of Sydney
Faculty of Medicine and Health
Sydney School of Public Health
MedicalResearch.com: What is the background for this study? Are the screeners specially trained, use full body photographs, dermoscopy etc?
Response: The Melanoma High-risk Clinic Study was developed to optimise the early detection of new melanomas in people at high risk of developing melanoma. A previous single-centre study observed fewer excisions and healthcare costs, thinner melanomas and better quality of life when surveillance of high risk patients was conducted in a melanoma dermatology clinic with a structured surveillance protocol involving 6-monthly full body examinations aided by total body photography (TBP) and sequential digital dermoscopy imaging (SDDI). The initial pilot study was performed in a single tertiary referral specialist centre using trained dermatologists who routinely used the diagnostic interventions.
Our objective was to examine longer-term sustainability and expansion of the program to multiple practices including a primary care skin cancer clinic setting. The hypothesis was that the outcomes would be similar if using the same protocol and diagnostic tools. The participating doctors were trained to follow the protocol, which included instruction on how to respond and interpret changing lesions, but not in use of dermoscopy or skin examinations, which were routinely and consistently used in all clinics prior to the study commencing. There were 593 participants assessed as very high risk of melanoma who participated in the Melanoma High-risk Clinic Study from 2012-2018. Nearly all of the participants had had a previous melanoma and had additional melanoma risk factors. 57% were male and the median age at study entry was 58 years.






 Dr. Brooks[/caption]
Dr. Kelly Brooks PhD
Research Officer
QIMR Berghofer Medical Research Institute
MedicalResearch.com: What is the background for this study?
[caption id="attachment_54258" align="alignleft" width="200"]
Dr. Brooks[/caption]
Dr. Kelly Brooks PhD
Research Officer
QIMR Berghofer Medical Research Institute
MedicalResearch.com: What is the background for this study?
[caption id="attachment_54258" align="alignleft" width="200"]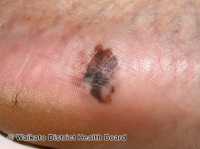









 Dr. Helen Marsden PhD
Skin Analytics Limited
London, United Kingdom
MedicalResearch.com: What is the background for this study?
Response: In this technology age, with the explosion of interest and applications using Artificial Intelligence, it is easy to accept the output of a technology-based test - such as a smartphone app designed to identify skin cancer - without thinking too much about it. In reality, technology is only as good as the way it has been developed, tested and validated. In particular, AI algorithms are prone to a lack of “generalisation” - i.e. their performance drops when presented with data it has not seen before. In the medical field, and particularly in areas where AI is being developed to direct a patient’s diagnosis or care, this is particularly problematic. Inappropriate diagnosis or advice to patients can lead to false reassurance, heightened concern and pressure on NHS services, or worse. It is concerning, therefore, that there are a large number of smartphone apps available that provide an assessment of skin lesions, including some that provide an estimate of the probability of malignancy, that have not been assessed for diagnostic accuracy.
Skin Analytics has developed an AI-based algorithm, named: Deep Ensemble for Recognition of Malignancy (DERM), for use as a decision support tool for healthcare providers. DERM determines the likelihood of skin cancer from dermoscopic images of skin lesions. It was developed using deep learning techniques that identify and assess features of these lesions which are associated with melanoma, using over 7,000 archived dermoscopic images. Using these images, it was shown to identify melanoma with similar accuracy to specialist physicians. However, to prove the algorithm could be used in a real life clinical setting, Skin Analytics set out to conduct a clinical validation study.
Dr. Helen Marsden PhD
Skin Analytics Limited
London, United Kingdom
MedicalResearch.com: What is the background for this study?
Response: In this technology age, with the explosion of interest and applications using Artificial Intelligence, it is easy to accept the output of a technology-based test - such as a smartphone app designed to identify skin cancer - without thinking too much about it. In reality, technology is only as good as the way it has been developed, tested and validated. In particular, AI algorithms are prone to a lack of “generalisation” - i.e. their performance drops when presented with data it has not seen before. In the medical field, and particularly in areas where AI is being developed to direct a patient’s diagnosis or care, this is particularly problematic. Inappropriate diagnosis or advice to patients can lead to false reassurance, heightened concern and pressure on NHS services, or worse. It is concerning, therefore, that there are a large number of smartphone apps available that provide an assessment of skin lesions, including some that provide an estimate of the probability of malignancy, that have not been assessed for diagnostic accuracy.
Skin Analytics has developed an AI-based algorithm, named: Deep Ensemble for Recognition of Malignancy (DERM), for use as a decision support tool for healthcare providers. DERM determines the likelihood of skin cancer from dermoscopic images of skin lesions. It was developed using deep learning techniques that identify and assess features of these lesions which are associated with melanoma, using over 7,000 archived dermoscopic images. Using these images, it was shown to identify melanoma with similar accuracy to specialist physicians. However, to prove the algorithm could be used in a real life clinical setting, Skin Analytics set out to conduct a clinical validation study.
 Dr. Qing Chen[/caption]
Qing Chen, M.D., Ph.D.
Assistant Professor, Immunology, Microenvironment & Metastasis Program
Scientific Director, Imaging Facility
The Wistar Institute
MedicalResearch.com: What is the background for this study?
Response: We are focusing on how a specific type of brain cells, astrocytes, helps the cancer cells from melanoma and breast cancer to form metastatic lesions.
Dr. Qing Chen[/caption]
Qing Chen, M.D., Ph.D.
Assistant Professor, Immunology, Microenvironment & Metastasis Program
Scientific Director, Imaging Facility
The Wistar Institute
MedicalResearch.com: What is the background for this study?
Response: We are focusing on how a specific type of brain cells, astrocytes, helps the cancer cells from melanoma and breast cancer to form metastatic lesions.




 Prof. Satchi-Fainaro[/caption]
Prof. Ronit Satchi-Fainaro, PhD
Head, Cancer Research and Nanomedicine Laboratory
Department of Physiology and Pharmacology,
Sackler Faculty of Medicine,
Tel Aviv University,
Tel-Aviv 69978, Israel
[caption id="attachment_50670" align="alignleft" width="116"]
Prof. Satchi-Fainaro[/caption]
Prof. Ronit Satchi-Fainaro, PhD
Head, Cancer Research and Nanomedicine Laboratory
Department of Physiology and Pharmacology,
Sackler Faculty of Medicine,
Tel Aviv University,
Tel-Aviv 69978, Israel
[caption id="attachment_50670" align="alignleft" width="116"] Prof. Florindo[/caption]
Prof. Helena Florindo, PhD
Head, BioNanoSciences – iMed.ULisboa
Faculty of Pharmacy,
University of Lisbon
Lisbon, Portugal
MedicalResearch.com: What are the main findings?
Response: The war against cancer in general, and melanoma in particular, has advanced over the years through a variety of treatment modalities, such as surgery, chemotherapy, radiation therapy and immunotherapy. The immune checkpoint inhibitors brought a breakthrough solution for advanced melanoma patients, but only a low percentage of those respond to this therapy, developing resistance and being affected by severe side effects. Despite the success of several vaccines against viral diseases, this success has not been materialized yet against cancer.
This study led by my lab at Tel Aviv University, and Helena Florindo’s lab at the University of Lisbon, describes the development of an effective nano-vaccine against melanoma, that also sensitizes the immune system to immunotherapies.
This nano-vaccine prevented melanoma, and also led to remarkable tumor inhibition and prolonged survival in mice already affected by this disease.
Prof. Florindo[/caption]
Prof. Helena Florindo, PhD
Head, BioNanoSciences – iMed.ULisboa
Faculty of Pharmacy,
University of Lisbon
Lisbon, Portugal
MedicalResearch.com: What are the main findings?
Response: The war against cancer in general, and melanoma in particular, has advanced over the years through a variety of treatment modalities, such as surgery, chemotherapy, radiation therapy and immunotherapy. The immune checkpoint inhibitors brought a breakthrough solution for advanced melanoma patients, but only a low percentage of those respond to this therapy, developing resistance and being affected by severe side effects. Despite the success of several vaccines against viral diseases, this success has not been materialized yet against cancer.
This study led by my lab at Tel Aviv University, and Helena Florindo’s lab at the University of Lisbon, describes the development of an effective nano-vaccine against melanoma, that also sensitizes the immune system to immunotherapies.
This nano-vaccine prevented melanoma, and also led to remarkable tumor inhibition and prolonged survival in mice already affected by this disease. 




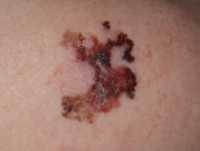
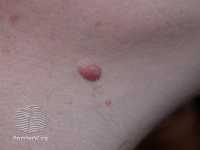
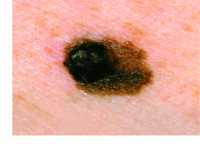
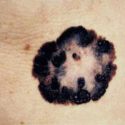 Example of one type of melanoma[/caption]
Dr. Catherine M. Olsen
Associate Professor
Cancer Control Group
QIMR Berghofer Medical Research Institute
MedicalResearch.com: What is the background for this study?
Response: Melanoma incidence and mortality rates are increasing globally. Public health campaigns aiming to reduce sun exposure and use of sunbed have been implemented in many parts of the world, but there is significant variability in terms of the history and reach of these campaigns across countries. We examined melanoma incidence rates in eight different countries with different patterns of sun exposure and varying approaches to melanoma control.
Example of one type of melanoma[/caption]
Dr. Catherine M. Olsen
Associate Professor
Cancer Control Group
QIMR Berghofer Medical Research Institute
MedicalResearch.com: What is the background for this study?
Response: Melanoma incidence and mortality rates are increasing globally. Public health campaigns aiming to reduce sun exposure and use of sunbed have been implemented in many parts of the world, but there is significant variability in terms of the history and reach of these campaigns across countries. We examined melanoma incidence rates in eight different countries with different patterns of sun exposure and varying approaches to melanoma control.