MedicalResearch.com Interview with:
[caption id="attachment_45754" align="alignleft" width="130"]

Dr. Radcliffe[/caption]
Nathan Radcliffe, MD
Senior Faculty, Ophthalmology Glaucoma and Cataract surgeon
Mount Sinai Health System
MedicalResearch.com: What is the background for this study?
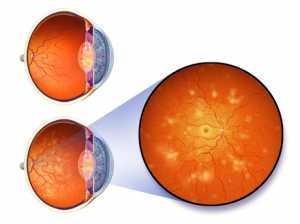
Response: Glaucoma is a leading cause of blindness and the mainstay of therapy is to lower the intraocular pressure (IOP) with topical eye drops.
Up to 40% of patients may require more than one eye drop to control the disease, and yet taking more than one eye drop bottle can result in higher costs, more eye irritation, worse therapeutic compliance, and possibly worse outcomes, be sure to consult your
eye surgeon before increasing any eye treatment to ensure it won't do any further damage to the eye. Compounded therapies (not FDA approved, but made at the physician’s request by a compounding pharmacy) can be created to contain multiple glaucoma therapies in one bottle.
We sought to determine if a compounded solution containing three or four drops in one bottle could control glaucoma as well as three or four separate bottles (standard of care) in patients requiring three or four eye drop bottles to control glaucoma.
We performed a multi-center, randomized, observer-masked, parallel-group study comparing a compounded therapy containing latanoprost 0.05%, dorzolamide hydrochloride 2%, timolol maleate 0.5%, brimonidine tartrate 0.2% with 0.01% BAK to standard three or four bottle regaimins. We measured IOP and corneal staining (a sign of preservative toxicity), as well as other safety measures at week one, month one, two and three.
 Source[/caption]
Source[/caption]
 Source[/caption]
Source[/caption]




 Prof. FAN Zhiyong PhD
University of California, Irvine
HKUST School of Engineering
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: According to the report of The World Health Organization, there are over 252 million people suffering from visual impairment globally and 15 million of them are difficult to cure by conventional medical methods. However, today, even the best bionic eyes have only 200 clinical trials, less than 1 ppm of all the patients, mainly due to their poor performance and high cost. The huge gap in supply and demand triggers the study of bionic eyes with performance comparable to human eyes. One important reason for their poor performance is the mismatch in shape between the flat bionic eyes and concave sclera. To protect the soft tissue in eyes from being damaged by the bionic surface, the implanted bionic eyes have to be small. This has limited the sensing area and further the electrodes number, and finally yielded poor image sensing characters with low resolution and narrow field-of-view.
In this work, we are trying to achieve high performance image sensing by biomimeticing human eyes. The high-density NWs are well aligned and embedded in a hemispherical template to serve as retina. The conformal attachment of bionic eyes with sclera enables the large sensing area and wide visual angle. In addition, each individual high-density nanowires can potentially work as an individual pixel. By addressing these challenges, our device design has huge potential to improve the image sensing performance of bionic eyes.
Prof. FAN Zhiyong PhD
University of California, Irvine
HKUST School of Engineering
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: According to the report of The World Health Organization, there are over 252 million people suffering from visual impairment globally and 15 million of them are difficult to cure by conventional medical methods. However, today, even the best bionic eyes have only 200 clinical trials, less than 1 ppm of all the patients, mainly due to their poor performance and high cost. The huge gap in supply and demand triggers the study of bionic eyes with performance comparable to human eyes. One important reason for their poor performance is the mismatch in shape between the flat bionic eyes and concave sclera. To protect the soft tissue in eyes from being damaged by the bionic surface, the implanted bionic eyes have to be small. This has limited the sensing area and further the electrodes number, and finally yielded poor image sensing characters with low resolution and narrow field-of-view.
In this work, we are trying to achieve high performance image sensing by biomimeticing human eyes. The high-density NWs are well aligned and embedded in a hemispherical template to serve as retina. The conformal attachment of bionic eyes with sclera enables the large sensing area and wide visual angle. In addition, each individual high-density nanowires can potentially work as an individual pixel. By addressing these challenges, our device design has huge potential to improve the image sensing performance of bionic eyes.