Author Interviews, Breast Cancer, Cancer Research, Genetic Research, Ovarian Cancer / 22.07.2020
Population Based BRCA-Testing Can Detect More Breast and Ovarian Cancers, and Be Cost-Effective
MedicalResearch.com Interview with:
[caption id="attachment_54909" align="alignleft" width="200"] Prof. Manchanda[/caption]
Prof Ranjit Manchanda MD, MRCOG, PhD
Professor of Gynaecological Oncology & Consultant Gynaecological Oncologist
NHS Innovation Accelerator (NIA) Fellow
Integrated Academic Training Programme Director
London Specialty School of Obstetrics & Gynaecology, Health Education England
Specialty Research Lead for Gynaecological Cancer, NIHR, North Thames Clinical Research Network
Cancer Research UK, Barts Centre | Queen Mary University of London
Centre for Cancer Prevention, Wolfson Institute of Preventive Medicine | Charterhouse Square | London
Department of Gynaecological Oncology | Barts Health NHS Trust, Royal London Hospital
MedicalResearch.com: What is the background for this study?
Response: Around 10–20% of ovarian cancers and 6% breast cancers overall are caused by inheritable BRCA1/BRCA2 mutations. Women carrying BRCA1/BRCA2 mutations have a 17–44% risk of ovarian cancer and 69–72% risk of breast cancer until age 80 years. Most of these cancers can be prevented in unaffected BRCA1/BRCA2 women carriers. Women can opt for a range of options including screening, preventive, and reproductive choices to minimise their risk.
The current approach uses established clinical-criteria/family-history (FH) based a priori BRCA probability thresholds to identify high-risk individuals eligible for BRCA testing. However, this requires individuals and health practitioners to recognise and act on a significant FH. BRCA carriers, who are unaware of their FH, unappreciative of its risk/significance, not proactive in seeking advice, or lack a strong FH (small families/paternal inheritance/chance) get excluded. Over 50% BRCA carriers do not fulfil clinical criteria and are missed.
Despite >25 years of BRCA testing and effective mechanisms for prevention, current guidelines and access to testing pathways remain complex and associated with a massive under-utilisation of genetic testing. Only 20% of eligible women have accessed/undergone genetic testing and our earlier analysis showed that 97% of BRCA carriers in the population remain unidentified. Current detection rates are inadequate to identify all BRCA carriers and even doubling detection rates will not work.
Why should we wait for decades for people to develop cancer before identifying BRCA carriers and unaffected at-risk family members to offer prevention?. This highlights substantial missed opportunities for early detection and prevention. A new population testing approach can change this. Jewish population studies show this is feasible, acceptable, has high satisfaction (91–95%), significantly reduces anxiety, doesn’t harm psychological well-being or quality of life, and is extremely cost-effective. However, this has not been evaluated in the general population and in particular across different countries or health systems. The potential applicability and scope for this approach transcends continents and countries. Additionally, for interventions to be sustainable, they need to be cost-effective and affordable.
We have undertaken a cost-effectiveness analysis of population based BRCA testing compared with current standard clinical testing of women designated as high risk, across high income countries (UK/USA/Netherlands), upper-middle income countries (China/Brazil), and low-middle income country (India).
Prof. Manchanda[/caption]
Prof Ranjit Manchanda MD, MRCOG, PhD
Professor of Gynaecological Oncology & Consultant Gynaecological Oncologist
NHS Innovation Accelerator (NIA) Fellow
Integrated Academic Training Programme Director
London Specialty School of Obstetrics & Gynaecology, Health Education England
Specialty Research Lead for Gynaecological Cancer, NIHR, North Thames Clinical Research Network
Cancer Research UK, Barts Centre | Queen Mary University of London
Centre for Cancer Prevention, Wolfson Institute of Preventive Medicine | Charterhouse Square | London
Department of Gynaecological Oncology | Barts Health NHS Trust, Royal London Hospital
MedicalResearch.com: What is the background for this study?
Response: Around 10–20% of ovarian cancers and 6% breast cancers overall are caused by inheritable BRCA1/BRCA2 mutations. Women carrying BRCA1/BRCA2 mutations have a 17–44% risk of ovarian cancer and 69–72% risk of breast cancer until age 80 years. Most of these cancers can be prevented in unaffected BRCA1/BRCA2 women carriers. Women can opt for a range of options including screening, preventive, and reproductive choices to minimise their risk.
The current approach uses established clinical-criteria/family-history (FH) based a priori BRCA probability thresholds to identify high-risk individuals eligible for BRCA testing. However, this requires individuals and health practitioners to recognise and act on a significant FH. BRCA carriers, who are unaware of their FH, unappreciative of its risk/significance, not proactive in seeking advice, or lack a strong FH (small families/paternal inheritance/chance) get excluded. Over 50% BRCA carriers do not fulfil clinical criteria and are missed.
Despite >25 years of BRCA testing and effective mechanisms for prevention, current guidelines and access to testing pathways remain complex and associated with a massive under-utilisation of genetic testing. Only 20% of eligible women have accessed/undergone genetic testing and our earlier analysis showed that 97% of BRCA carriers in the population remain unidentified. Current detection rates are inadequate to identify all BRCA carriers and even doubling detection rates will not work.
Why should we wait for decades for people to develop cancer before identifying BRCA carriers and unaffected at-risk family members to offer prevention?. This highlights substantial missed opportunities for early detection and prevention. A new population testing approach can change this. Jewish population studies show this is feasible, acceptable, has high satisfaction (91–95%), significantly reduces anxiety, doesn’t harm psychological well-being or quality of life, and is extremely cost-effective. However, this has not been evaluated in the general population and in particular across different countries or health systems. The potential applicability and scope for this approach transcends continents and countries. Additionally, for interventions to be sustainable, they need to be cost-effective and affordable.
We have undertaken a cost-effectiveness analysis of population based BRCA testing compared with current standard clinical testing of women designated as high risk, across high income countries (UK/USA/Netherlands), upper-middle income countries (China/Brazil), and low-middle income country (India).
 Prof. Manchanda[/caption]
Prof Ranjit Manchanda MD, MRCOG, PhD
Professor of Gynaecological Oncology & Consultant Gynaecological Oncologist
NHS Innovation Accelerator (NIA) Fellow
Integrated Academic Training Programme Director
London Specialty School of Obstetrics & Gynaecology, Health Education England
Specialty Research Lead for Gynaecological Cancer, NIHR, North Thames Clinical Research Network
Cancer Research UK, Barts Centre | Queen Mary University of London
Centre for Cancer Prevention, Wolfson Institute of Preventive Medicine | Charterhouse Square | London
Department of Gynaecological Oncology | Barts Health NHS Trust, Royal London Hospital
MedicalResearch.com: What is the background for this study?
Response: Around 10–20% of ovarian cancers and 6% breast cancers overall are caused by inheritable BRCA1/BRCA2 mutations. Women carrying BRCA1/BRCA2 mutations have a 17–44% risk of ovarian cancer and 69–72% risk of breast cancer until age 80 years. Most of these cancers can be prevented in unaffected BRCA1/BRCA2 women carriers. Women can opt for a range of options including screening, preventive, and reproductive choices to minimise their risk.
The current approach uses established clinical-criteria/family-history (FH) based a priori BRCA probability thresholds to identify high-risk individuals eligible for BRCA testing. However, this requires individuals and health practitioners to recognise and act on a significant FH. BRCA carriers, who are unaware of their FH, unappreciative of its risk/significance, not proactive in seeking advice, or lack a strong FH (small families/paternal inheritance/chance) get excluded. Over 50% BRCA carriers do not fulfil clinical criteria and are missed.
Despite >25 years of BRCA testing and effective mechanisms for prevention, current guidelines and access to testing pathways remain complex and associated with a massive under-utilisation of genetic testing. Only 20% of eligible women have accessed/undergone genetic testing and our earlier analysis showed that 97% of BRCA carriers in the population remain unidentified. Current detection rates are inadequate to identify all BRCA carriers and even doubling detection rates will not work.
Why should we wait for decades for people to develop cancer before identifying BRCA carriers and unaffected at-risk family members to offer prevention?. This highlights substantial missed opportunities for early detection and prevention. A new population testing approach can change this. Jewish population studies show this is feasible, acceptable, has high satisfaction (91–95%), significantly reduces anxiety, doesn’t harm psychological well-being or quality of life, and is extremely cost-effective. However, this has not been evaluated in the general population and in particular across different countries or health systems. The potential applicability and scope for this approach transcends continents and countries. Additionally, for interventions to be sustainable, they need to be cost-effective and affordable.
We have undertaken a cost-effectiveness analysis of population based BRCA testing compared with current standard clinical testing of women designated as high risk, across high income countries (UK/USA/Netherlands), upper-middle income countries (China/Brazil), and low-middle income country (India).
Prof. Manchanda[/caption]
Prof Ranjit Manchanda MD, MRCOG, PhD
Professor of Gynaecological Oncology & Consultant Gynaecological Oncologist
NHS Innovation Accelerator (NIA) Fellow
Integrated Academic Training Programme Director
London Specialty School of Obstetrics & Gynaecology, Health Education England
Specialty Research Lead for Gynaecological Cancer, NIHR, North Thames Clinical Research Network
Cancer Research UK, Barts Centre | Queen Mary University of London
Centre for Cancer Prevention, Wolfson Institute of Preventive Medicine | Charterhouse Square | London
Department of Gynaecological Oncology | Barts Health NHS Trust, Royal London Hospital
MedicalResearch.com: What is the background for this study?
Response: Around 10–20% of ovarian cancers and 6% breast cancers overall are caused by inheritable BRCA1/BRCA2 mutations. Women carrying BRCA1/BRCA2 mutations have a 17–44% risk of ovarian cancer and 69–72% risk of breast cancer until age 80 years. Most of these cancers can be prevented in unaffected BRCA1/BRCA2 women carriers. Women can opt for a range of options including screening, preventive, and reproductive choices to minimise their risk.
The current approach uses established clinical-criteria/family-history (FH) based a priori BRCA probability thresholds to identify high-risk individuals eligible for BRCA testing. However, this requires individuals and health practitioners to recognise and act on a significant FH. BRCA carriers, who are unaware of their FH, unappreciative of its risk/significance, not proactive in seeking advice, or lack a strong FH (small families/paternal inheritance/chance) get excluded. Over 50% BRCA carriers do not fulfil clinical criteria and are missed.
Despite >25 years of BRCA testing and effective mechanisms for prevention, current guidelines and access to testing pathways remain complex and associated with a massive under-utilisation of genetic testing. Only 20% of eligible women have accessed/undergone genetic testing and our earlier analysis showed that 97% of BRCA carriers in the population remain unidentified. Current detection rates are inadequate to identify all BRCA carriers and even doubling detection rates will not work.
Why should we wait for decades for people to develop cancer before identifying BRCA carriers and unaffected at-risk family members to offer prevention?. This highlights substantial missed opportunities for early detection and prevention. A new population testing approach can change this. Jewish population studies show this is feasible, acceptable, has high satisfaction (91–95%), significantly reduces anxiety, doesn’t harm psychological well-being or quality of life, and is extremely cost-effective. However, this has not been evaluated in the general population and in particular across different countries or health systems. The potential applicability and scope for this approach transcends continents and countries. Additionally, for interventions to be sustainable, they need to be cost-effective and affordable.
We have undertaken a cost-effectiveness analysis of population based BRCA testing compared with current standard clinical testing of women designated as high risk, across high income countries (UK/USA/Netherlands), upper-middle income countries (China/Brazil), and low-middle income country (India).




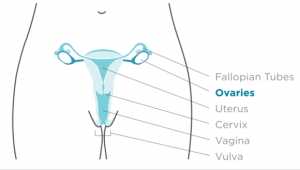
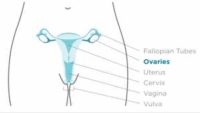




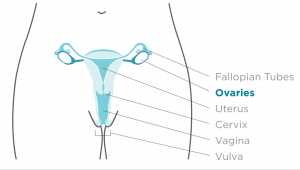






 Dr. Sibaji Sarkar[/caption]
Sibaji Sarkar Ph.D
Instructor of medicine
Boston University School of Medicine
Boston
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Sarkar: Although breast and ovarian cancers have different clinical presentations, there are certain molecular events that are conserved between the two types of cancers. For example, mutation in a few genes, such as BRCA1, BRCA2, is an indicator of possible development of both breast and ovarian cancers. ARHI, a pro-apoptotic imprinted gene is epigenetically silenced in both breast and ovarian cancers. A similar pattern was observed in microRNA as well. There are also several genes which are differentially expressed in these two types of cancers but few of these striking resemblances led us to investigate whether they have a common origin. In this paper, we compared genetic and epigenetic events in both breast and ovarian cancers and we hypothesize that they may have similar origin (mechanism of formation of cancer progenitor cells), which should be regulated by epigenetic mechanism.
Dr. Sibaji Sarkar[/caption]
Sibaji Sarkar Ph.D
Instructor of medicine
Boston University School of Medicine
Boston
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Sarkar: Although breast and ovarian cancers have different clinical presentations, there are certain molecular events that are conserved between the two types of cancers. For example, mutation in a few genes, such as BRCA1, BRCA2, is an indicator of possible development of both breast and ovarian cancers. ARHI, a pro-apoptotic imprinted gene is epigenetically silenced in both breast and ovarian cancers. A similar pattern was observed in microRNA as well. There are also several genes which are differentially expressed in these two types of cancers but few of these striking resemblances led us to investigate whether they have a common origin. In this paper, we compared genetic and epigenetic events in both breast and ovarian cancers and we hypothesize that they may have similar origin (mechanism of formation of cancer progenitor cells), which should be regulated by epigenetic mechanism.

 Dr. Dirk Timmerman[/caption]
Dr. Dirk Timmerman[/caption]
 Dr. Hamanishi[/caption]
MedicalResearch.com Interview with:
Junzo Hamanishi M.D., Ph.D.
Department of Gynecology and Obstetrics,
Kyoto University Graduate School of Medicine
Assistant Professor
Kyoto Japan
Medical Research: What is the background for this study?
Dr. Hamanishi: More than 70% of patients with advanced ovarian cancer who achieve remission ultimately relapse and there are few effective treatments for these patients. Because the development of new treatment strategies for these patients is urgently required, we have focused on and studied the potential of cancer cells to escape from host immunity with PD-1/PD-L1 immunosuppressive signal in the tumor microenvironment to find new treatment strategies to overcome this phenomenon,
Dr. Hamanishi[/caption]
MedicalResearch.com Interview with:
Junzo Hamanishi M.D., Ph.D.
Department of Gynecology and Obstetrics,
Kyoto University Graduate School of Medicine
Assistant Professor
Kyoto Japan
Medical Research: What is the background for this study?
Dr. Hamanishi: More than 70% of patients with advanced ovarian cancer who achieve remission ultimately relapse and there are few effective treatments for these patients. Because the development of new treatment strategies for these patients is urgently required, we have focused on and studied the potential of cancer cells to escape from host immunity with PD-1/PD-L1 immunosuppressive signal in the tumor microenvironment to find new treatment strategies to overcome this phenomenon,  Dr. McDonald[/caption]
MedicalResearch.com Interview with:
Professor John McDonald PhD
Director of its Integrated Cancer Research Center
School of Biology at the Georgia Institute of Technology
Medical Research: What is the background for this study? What are the main findings?
Response: Ovarian cancer is a deadly disease because it cannot be diagnosed at early stages when it can be most effectively effectively treated.
It has long been recognized that there is a great need for an accurate diagnostic test for early stage ovarian cancer.
Until now, efforts to develop a highly accurate way to detect early stage ovarian cancer have been unsuccessful.
We have used a novel approach that integrates advanced methods in analytical chemistry with advanced machine learning algorithms to identify 16 metabolites that collectively can detect ovarian cancer with extremely high accuracy (100% in the samples tested in our study)
Dr. McDonald[/caption]
MedicalResearch.com Interview with:
Professor John McDonald PhD
Director of its Integrated Cancer Research Center
School of Biology at the Georgia Institute of Technology
Medical Research: What is the background for this study? What are the main findings?
Response: Ovarian cancer is a deadly disease because it cannot be diagnosed at early stages when it can be most effectively effectively treated.
It has long been recognized that there is a great need for an accurate diagnostic test for early stage ovarian cancer.
Until now, efforts to develop a highly accurate way to detect early stage ovarian cancer have been unsuccessful.
We have used a novel approach that integrates advanced methods in analytical chemistry with advanced machine learning algorithms to identify 16 metabolites that collectively can detect ovarian cancer with extremely high accuracy (100% in the samples tested in our study)
 Dr. Qin[/caption]
MedicalResearch.com Interview with:
Bo (Bonnie) Qin, PhD
Postdoctoral associate at Rutgers Cancer Institute of New Jersey
Medical Research: What is the background for this study? What are the main findings?
Response: Ovarian cancer is among the top five causes of cancer death among women in the US. Compared to white women, African-American women tend to have a worse 5-year survival rate of ovarian cancer. It highlights a critical need for identifying preventive factors in African Americans, particularly through dietary modification, which is relatively low cost and low risk compared to medical treatments.
We found that adherence to an overall healthy dietary pattern i.e. Alternate Healthy Eating Index (AHEI)-2010 may reduce ovarian cancer risk in African-American women, and particularly among postmenopausal women. Adherence to the current Dietary Guidelines for Americans i.e. Healthy Eating Index-2010, were also strongly associated with reduced risk of ovarian cancer among postmenopausal African-American women.
Dr. Qin[/caption]
MedicalResearch.com Interview with:
Bo (Bonnie) Qin, PhD
Postdoctoral associate at Rutgers Cancer Institute of New Jersey
Medical Research: What is the background for this study? What are the main findings?
Response: Ovarian cancer is among the top five causes of cancer death among women in the US. Compared to white women, African-American women tend to have a worse 5-year survival rate of ovarian cancer. It highlights a critical need for identifying preventive factors in African Americans, particularly through dietary modification, which is relatively low cost and low risk compared to medical treatments.
We found that adherence to an overall healthy dietary pattern i.e. Alternate Healthy Eating Index (AHEI)-2010 may reduce ovarian cancer risk in African-American women, and particularly among postmenopausal women. Adherence to the current Dietary Guidelines for Americans i.e. Healthy Eating Index-2010, were also strongly associated with reduced risk of ovarian cancer among postmenopausal African-American women.





