Allergies, Author Interviews, Immunotherapy, Pediatrics / 18.04.2019
Oral Peanut Immunotherapy Evaluated for Preschool Children
MedicalResearch.com Interview with:
[caption id="attachment_48695" align="alignleft" width="162"] Dr. Soller[/caption]
Lianne Soller, PhD
Allergy Research Manager
University of British Columbia
Vancouver, BC, Canada
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In 2017, a clinical trial of 37 subjects demonstrated that preschool peanut oral immunotherapy was safe, with predominantly mild symptoms reported and only one moderate reaction requiring epinephrine. Our study aimed to examine whether these findings would be applicable in a real-world setting (i.e., outside of research).
We found that peanut oral immunotherapy is safe in the vast majority of preschoolers, with only 0.4% of patients experiencing a severe reaction, and only 12 out of ~40,000 peanut doses needed epinephrine (0.03%).
Dr. Soller[/caption]
Lianne Soller, PhD
Allergy Research Manager
University of British Columbia
Vancouver, BC, Canada
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In 2017, a clinical trial of 37 subjects demonstrated that preschool peanut oral immunotherapy was safe, with predominantly mild symptoms reported and only one moderate reaction requiring epinephrine. Our study aimed to examine whether these findings would be applicable in a real-world setting (i.e., outside of research).
We found that peanut oral immunotherapy is safe in the vast majority of preschoolers, with only 0.4% of patients experiencing a severe reaction, and only 12 out of ~40,000 peanut doses needed epinephrine (0.03%).
 Dr. Soller[/caption]
Lianne Soller, PhD
Allergy Research Manager
University of British Columbia
Vancouver, BC, Canada
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In 2017, a clinical trial of 37 subjects demonstrated that preschool peanut oral immunotherapy was safe, with predominantly mild symptoms reported and only one moderate reaction requiring epinephrine. Our study aimed to examine whether these findings would be applicable in a real-world setting (i.e., outside of research).
We found that peanut oral immunotherapy is safe in the vast majority of preschoolers, with only 0.4% of patients experiencing a severe reaction, and only 12 out of ~40,000 peanut doses needed epinephrine (0.03%).
Dr. Soller[/caption]
Lianne Soller, PhD
Allergy Research Manager
University of British Columbia
Vancouver, BC, Canada
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: In 2017, a clinical trial of 37 subjects demonstrated that preschool peanut oral immunotherapy was safe, with predominantly mild symptoms reported and only one moderate reaction requiring epinephrine. Our study aimed to examine whether these findings would be applicable in a real-world setting (i.e., outside of research).
We found that peanut oral immunotherapy is safe in the vast majority of preschoolers, with only 0.4% of patients experiencing a severe reaction, and only 12 out of ~40,000 peanut doses needed epinephrine (0.03%). 




 Dr. Robinson[/caption]
Anne Robinson, Pharm D
Executive Scientific Director
AbbVie
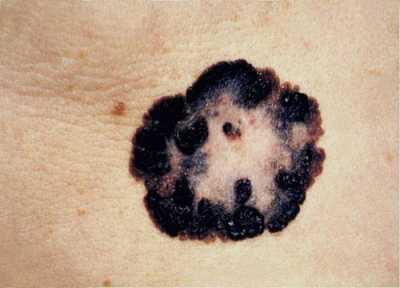
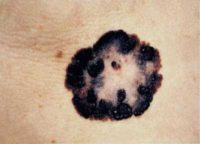
MedicalResearch.com: What is the background for the risankizumab data presented at the American Academy of Dermatology 2019 Annual Meeting?
Response: Abstracts presented by AbbVie at the American Academy of Dermatology (AAD) 2019 Annual Meeting highlight additional data from the Phase 3 clinical trial program evaluating the safety and efficacy of risankizumab, an investigational interleukin-23 (IL-23) inhibitor. The registrational program for risankizumab evaluated more than 2,000 adult patients with moderate to severe plaque psoriasis across four pivotal studies.
Dr. Robinson[/caption]
Anne Robinson, Pharm D
Executive Scientific Director
AbbVie
MedicalResearch.com: What is the background for the risankizumab data presented at the American Academy of Dermatology 2019 Annual Meeting?
Response: Abstracts presented by AbbVie at the American Academy of Dermatology (AAD) 2019 Annual Meeting highlight additional data from the Phase 3 clinical trial program evaluating the safety and efficacy of risankizumab, an investigational interleukin-23 (IL-23) inhibitor. The registrational program for risankizumab evaluated more than 2,000 adult patients with moderate to severe plaque psoriasis across four pivotal studies.