MedicalResearch.com Interview with:
[caption id="attachment_39686" align="alignleft" width="247"]
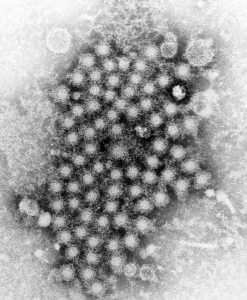
In the United States, hepatitis A, hepatitis B and hepatitis C are the most common types, but also included are hepatitis D and E.
CDC/ E.H. Cook, Jr.[/caption]
Stefan Zeuzem, M.D.
Professor of Medicine
Chief Internal Medicine
Goethe University Hospital
Frankfurt, Germany
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Chronic hepatitis C virus (HCV) infection is a major global public health problem with more than 71 million people infected worldwide, and can result in significant morbidity and mortality, including liver cirrhosis, hepatocellular carcinoma, and death.
1
This publication describes the efficacy and safety results from two Phase 3 clinical trials, ENDURANCE-1 and ENDURANCE-3, in patients with chronic HCV genotypes (GT) 1 or 3 infection who were treated with an all-oral, once-daily combination regimen of direct-acting antiviral agents (DAA) glecaprevir (GLE) at 300 mg and pibrentasvir (PIB) at 120 mg.
The findings from ENDURANCE-1 trial show that the GLE/PIB combination regimen (G/P) given for 8 weeks to HCV GT1 chronically infected non-cirrhotic treatment-naïve or treatment-experienced (with sofosbuvir and/or interferon with ribavirin) patients was safe and well-tolerated, achieved high efficacy with a sustained virologic response at post-treatment week 12 (SVR12) rate >99% and was non-inferior to 12-week treatment with G/P.
The trial also included subjects who were co-infected with human immunodeficiency virus (HIV), and all of these subjects achieved SVR12 while maintaining HIV suppression throughout the study. ENDURANCE-3 trial results show that the G/P regimen given for 8 weeks to HCV GT3 chronically infected non-cirrhotic treatment-naïve patients was safe and well-tolerated, achieved high efficacy in this historically difficult to cure GT with an SVR12 rate >94%, and was non-inferior to 12-week treatment with G/P, which in turn was non-inferior to the treatment with 12-week DAA regimen of sofosbivir and daclatasvir.












 Dr. Darius Lakdawalla[/caption]
Darius Lakdawalla PhD
Quintiles Chair in Pharmaceutical Development and Regulatory Innovation
School of Pharmacy
Professor in the Sol Price School of Public Policy
University of Southern California
MedicalResearch.com: What is the background for this study?
Dr. Lakdawalla: New treatments for hepatitis-C are highly effective but also involve high upfront costs. Because they effectively cure the disease, all the costs of treatments are paid over a short period of time – about three months – but the benefits accrue for the rest of a patient’s life. This creates problems for the private health insurance system, where patients switch insurers. The insurer that pays the bill for the treatment might not be around to enjoy the benefits of averting liver damage, liver transplants, and other costly complications associated with hepatitis-C.
Dr. Darius Lakdawalla[/caption]
Darius Lakdawalla PhD
Quintiles Chair in Pharmaceutical Development and Regulatory Innovation
School of Pharmacy
Professor in the Sol Price School of Public Policy
University of Southern California
MedicalResearch.com: What is the background for this study?
Dr. Lakdawalla: New treatments for hepatitis-C are highly effective but also involve high upfront costs. Because they effectively cure the disease, all the costs of treatments are paid over a short period of time – about three months – but the benefits accrue for the rest of a patient’s life. This creates problems for the private health insurance system, where patients switch insurers. The insurer that pays the bill for the treatment might not be around to enjoy the benefits of averting liver damage, liver transplants, and other costly complications associated with hepatitis-C.

 MedicalResearch.com Interview with:
Brittany Kmush, ScM
Doctoral Candidate
Global Disease Epidemiology and Control
Department of International Health
Johns Hopkins Bloomberg School of Public Health
Baltimore, MD
Medical Research: What is the background for this study? What are the main findings?
Response: Hepatitis E virus (HEV) is a global pathogen responsible for approximately 20 million infections every year in developing countries. In the general population, HEV causes acute, self-limiting hepatitis with only a 1-2% case fatality rate. However, in pregnant women, Hepatitis E virus infection can be very severe, resulting in fulminant hepatic failure and death, with a case fatality rate around 30%. Despite this important burden, Hepatitis E virus remains an under-recognized and under-reported pathogen. The early years of HEV research were plagued by poor quality commercial assays, highly variable in sensitivity and specificity. As a result, there is still no diagnostic assay approved for commercial use in the United States. However, over the past two decades, several new, highly sensitive and specific assays have been developed.
In this study, we re-tested banked sera from a population-based sero-survey of over 1000 participants from rural Bangladesh in order to investigate the comparability of a high-performing first generation test to recently developed, commercially available assay. In the early 2000s, the Walter Reed Army Institute of Research (WRAIR, Bethesda, MD) developed an in-house enzyme immune-assay (EIA) to diagnose Hepatitis E virus infections by detecting anti-HEV total immunoglobulin (Ig) in serum. More recently, Wantai Diagnostics (Beijing, China) developed a commercially available EIA for detecting anti-HEV IgG.
The WRAIR assay estimated the overall population seroprevalence as 26.6% while the Wantai assay produced significantly higher estimated seroprevalence, 46.7%. There was a 77% agreement between the two tests. Overall, the Wantai assay found a much higher seroprevalence of anti-HEV antibodies compared to the WRAIR assay, using the same serum. Additionally, the majority of the differences between the two tests are from people initially classified by WRAIR as anti-HEV negative that Wantai classified as anti-HEV positive.
MedicalResearch.com Interview with:
Brittany Kmush, ScM
Doctoral Candidate
Global Disease Epidemiology and Control
Department of International Health
Johns Hopkins Bloomberg School of Public Health
Baltimore, MD
Medical Research: What is the background for this study? What are the main findings?
Response: Hepatitis E virus (HEV) is a global pathogen responsible for approximately 20 million infections every year in developing countries. In the general population, HEV causes acute, self-limiting hepatitis with only a 1-2% case fatality rate. However, in pregnant women, Hepatitis E virus infection can be very severe, resulting in fulminant hepatic failure and death, with a case fatality rate around 30%. Despite this important burden, Hepatitis E virus remains an under-recognized and under-reported pathogen. The early years of HEV research were plagued by poor quality commercial assays, highly variable in sensitivity and specificity. As a result, there is still no diagnostic assay approved for commercial use in the United States. However, over the past two decades, several new, highly sensitive and specific assays have been developed.
In this study, we re-tested banked sera from a population-based sero-survey of over 1000 participants from rural Bangladesh in order to investigate the comparability of a high-performing first generation test to recently developed, commercially available assay. In the early 2000s, the Walter Reed Army Institute of Research (WRAIR, Bethesda, MD) developed an in-house enzyme immune-assay (EIA) to diagnose Hepatitis E virus infections by detecting anti-HEV total immunoglobulin (Ig) in serum. More recently, Wantai Diagnostics (Beijing, China) developed a commercially available EIA for detecting anti-HEV IgG.
The WRAIR assay estimated the overall population seroprevalence as 26.6% while the Wantai assay produced significantly higher estimated seroprevalence, 46.7%. There was a 77% agreement between the two tests. Overall, the Wantai assay found a much higher seroprevalence of anti-HEV antibodies compared to the WRAIR assay, using the same serum. Additionally, the majority of the differences between the two tests are from people initially classified by WRAIR as anti-HEV negative that Wantai classified as anti-HEV positive.






