[caption id="attachment_73889" align="aligncenter" width="500"] Photo by Gustavo Fring[/caption]
Historically, medical and dental care have been treated as separate disciplines in the minds of many patients. Most people associate dental visits primarily with cavity prevention, fresh breath, and achieving a bright, confident smile. However, modern clinical research paints a much broader and more complex picture of why oral hygiene is so critical. The human mouth is a literal gateway to the entire body, and neglecting its care can lead to a cascade of medical issues that extend far beyond tooth decay. Establishing a consistent routine with a reliable local dental spot for preventative screenings and professional cleans is actually one of the most effective ways to protect yourself against long-term, chronic systemic inflammation.
The mechanism behind this whole-body impact comes down to the immune system's biological response to bacterial overgrowth. When plaque is allowed to accumulate and harden into tartar, it creates a highly protected environment where harmful bacteria thrive along and beneath the gumline. This bacterial invasion triggers an immediate immune response, causing localised inflammation known as gingivitis. If left untreated by a professional, this early-stage condition progresses into periodontitis. Periodontitis is a severe infection that breaks down the soft tissue and bone supporting the teeth, creating pockets where even more bacteria can rapidly multiply.
Photo by Gustavo Fring[/caption]
Historically, medical and dental care have been treated as separate disciplines in the minds of many patients. Most people associate dental visits primarily with cavity prevention, fresh breath, and achieving a bright, confident smile. However, modern clinical research paints a much broader and more complex picture of why oral hygiene is so critical. The human mouth is a literal gateway to the entire body, and neglecting its care can lead to a cascade of medical issues that extend far beyond tooth decay. Establishing a consistent routine with a reliable local dental spot for preventative screenings and professional cleans is actually one of the most effective ways to protect yourself against long-term, chronic systemic inflammation.
The mechanism behind this whole-body impact comes down to the immune system's biological response to bacterial overgrowth. When plaque is allowed to accumulate and harden into tartar, it creates a highly protected environment where harmful bacteria thrive along and beneath the gumline. This bacterial invasion triggers an immediate immune response, causing localised inflammation known as gingivitis. If left untreated by a professional, this early-stage condition progresses into periodontitis. Periodontitis is a severe infection that breaks down the soft tissue and bone supporting the teeth, creating pockets where even more bacteria can rapidly multiply.
 Photo by Gustavo Fring[/caption]
Historically, medical and dental care have been treated as separate disciplines in the minds of many patients. Most people associate dental visits primarily with cavity prevention, fresh breath, and achieving a bright, confident smile. However, modern clinical research paints a much broader and more complex picture of why oral hygiene is so critical. The human mouth is a literal gateway to the entire body, and neglecting its care can lead to a cascade of medical issues that extend far beyond tooth decay. Establishing a consistent routine with a reliable local dental spot for preventative screenings and professional cleans is actually one of the most effective ways to protect yourself against long-term, chronic systemic inflammation.
The mechanism behind this whole-body impact comes down to the immune system's biological response to bacterial overgrowth. When plaque is allowed to accumulate and harden into tartar, it creates a highly protected environment where harmful bacteria thrive along and beneath the gumline. This bacterial invasion triggers an immediate immune response, causing localised inflammation known as gingivitis. If left untreated by a professional, this early-stage condition progresses into periodontitis. Periodontitis is a severe infection that breaks down the soft tissue and bone supporting the teeth, creating pockets where even more bacteria can rapidly multiply.
Photo by Gustavo Fring[/caption]
Historically, medical and dental care have been treated as separate disciplines in the minds of many patients. Most people associate dental visits primarily with cavity prevention, fresh breath, and achieving a bright, confident smile. However, modern clinical research paints a much broader and more complex picture of why oral hygiene is so critical. The human mouth is a literal gateway to the entire body, and neglecting its care can lead to a cascade of medical issues that extend far beyond tooth decay. Establishing a consistent routine with a reliable local dental spot for preventative screenings and professional cleans is actually one of the most effective ways to protect yourself against long-term, chronic systemic inflammation.
The mechanism behind this whole-body impact comes down to the immune system's biological response to bacterial overgrowth. When plaque is allowed to accumulate and harden into tartar, it creates a highly protected environment where harmful bacteria thrive along and beneath the gumline. This bacterial invasion triggers an immediate immune response, causing localised inflammation known as gingivitis. If left untreated by a professional, this early-stage condition progresses into periodontitis. Periodontitis is a severe infection that breaks down the soft tissue and bone supporting the teeth, creating pockets where even more bacteria can rapidly multiply.

![Most people only think about their teeth when something hurts. A twinge. A sensitivity. A filling that feels loose. Then the appointment gets booked, the problem gets fixed, and life moves on until the next issue surfaces. It is understandable. But it misses something important. Your mouth is connected to your heart, your lungs, your blood sugar, and your immune response. Researchers have been mapping these connections for decades, yet most people never hear about them in a routine check-up. [ IMAGE 1 ] Your Mouth and Your Heart Have More in Common Than You Think The mouth hosts hundreds of bacterial species. In a healthy mouth, they coexist without causing problems. When the balance shifts, certain bacteria become destructive. They inflame gum tissue — and that inflammation does not stay put. Studies in cardiovascular medicine have found consistent associations between gum disease and elevated heart disease risk. The mechanism is surprisingly direct: bacteria from infected gum tissue enter the bloodstream and travel to arterial walls. Researchers have actually found oral bacteria inside arterial plaque samples. That shifted the conversation from statistical association to something far more specific. Diabetes adds another layer. People with poorly controlled blood sugar tend to have more severe gum disease, and untreated gum disease appears to make blood sugar harder to regulate in return. It runs both ways. Respiratory health is also gaining attention — bacteria from the mouth have been linked to pneumonia and lung infections, particularly in older adults. In pregnancy, gum disease has been associated with preterm birth and low birth weight, and some health systems now recommend dental check-ups as standard prenatal care. The Inflammation Factor Nobody Talks About Short-term inflammation is useful — it is the body defending itself. Chronic, low-grade inflammation that drags on for months or years is something else entirely. It sits at the root of heart disease, type 2 diabetes, certain cancers, autoimmune disorders, and cognitive decline. Advanced gum disease is a chronic inflammatory condition. The gums become a persistent source of immune activation, and the chemicals produced — called cytokines — circulate through the body. This is why dental health is no longer just about avoiding cavities. It is about managing one genuine contributor to body-wide inflammation. Gum disease is largely preventable and responds well to treatment. Adults who have drifted away from regular dental care often find that re-establishing it is one of the more impactful decisions they can make. Finding a dentist in Brighton or a trusted local practice and booking that overdue check-up is a reasonable first step. Not perfection — just professional oversight back in the picture. [ IMAGE 2 ] Why Childhood Sets the Trajectory There is a concept in medicine called the critical window — a period in development when habits and exposures have an outsized effect on long-term outcomes. For oral health, that window opens early. Children who see a dentist regularly from a young age get more than clean teeth. They get comfortable with the environment, and that matters more than most parents realise. Dental anxiety stops many adults from seeking timely care, and a significant amount of that anxiety traces back to early experiences. Primary teeth matter too. They hold space in the jaw for permanent teeth, support speech development, and allow children to eat without pain. When lost too early through decay or infection, they disrupt everything that follows. There is a social dimension as well. Children with visible decay or dental pain often hold back — they avoid smiling, eat less comfortably at school, and stay quiet in class. Getting children into a supportive, child-focused environment early makes a genuine difference. Families who want that specialist approach will find that a dedicated kids dentist Newtown or a similarly focused local practice offers both clinical expertise and the patient manner that makes dental visits manageable rather than dreaded. [ IMAGE 3 ] What Good Daily Habits Actually Look Like Brushing twice a day with fluoride toothpaste is still the foundation. Two minutes is the clinical recommendation — most people do around 45 seconds. A simple phone timer changes this more than any gadget will. Electric toothbrushes consistently outperform manual ones in the research, particularly along the gumline. Flossing clears the contact points between teeth that bristles cannot reach — precisely where decay and gum disease most often begin. If flossing feels awkward, interdental brushes are easier and equally effective. Diet matters more than most people expect, but perhaps not in the way they think. Frequency of sugar exposure is more damaging than total intake. Each sugary encounter triggers an acid attack on enamel lasting around 20 minutes — sipping a soft drink across three hours is far harder on teeth than something sweet eaten once with a meal. Stress is the overlooked factor. It contributes to grinding and clenching during sleep, which wears enamel and can fracture teeth over time. A dentist can spot the signs early and recommend a night guard before real damage accumulates. Why Waiting Almost Always Costs More A small cavity caught early takes minutes to treat. Left alone, it reaches the nerve — meaning root canal treatment. Left longer still, the tooth may not be salvageable at all. Then comes the extraction, the bone loss, the shifting of adjacent teeth, and eventually the conversation about implants or bridges. At every stage the cost increases. The treatment that costs least and causes least discomfort is always the earliest one. Gum disease follows the same pattern. Early-stage gingivitis reverses with a professional clean and better home care. Advanced periodontitis involves bone loss that cannot be restored, only managed. The only thing separating those two outcomes is usually how long treatment was delayed. Rethinking What Dental Care Is Actually For Your mouth is not separate from your health — it is part of it. Treating dental care as optional, or as something to deal with only when things go wrong, ignores what the evidence has been building toward for years. For adults, that means a regular check-up rhythm with a practice you trust. For parents, it means introducing dental visits early, keeping them calm and low-key, and not letting your own anxieties pass to your children. The research keeps deepening and the connections between oral health and the rest of the body keep getting clearer. Taking care of your mouth is, increasingly, one of the more straightforward things you can do for your overall health. Disclaimer: The information on MedicalResearch.com is provided for educational purposes only, and is in no way intended to diagnose, cure, or treat any medical or other condition. Some links are sponsored. Products, services and providers are not warranted or endorsed by MedicalResearch.com or Eminent Domains Inc. Always seek the advice of your physician or other qualified health and ask your doctor any questions you may have regarding a medical condition. In addition to all other limitations and disclaimers in this agreement, service provider and its third party providers disclaim any liability or loss in connection with the content provided on this website.](https://medicalresearch.com/wp-content/uploads/2026/05/oral-health-and-bodys-well-being.png)









 Dr. Sinha[/caption]
Pranay Sinha, MD
Research Fellow
Section of Infectious Diseases
Boston University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: In the early days of the COVID-19 pandemic there were no evidence-based treatments for severely ill patients infected with this virus. We formed an interdisciplinary group of physicians from departments of adult and pediatric infectious diseases, rheumatology, and pulmonary/critical care as well as clinical pharmacy specialists. Given some promising data from China, we instituted treatment with off-label IL-6 receptor inhibitors (tocilizumab and sarilumab). The rationale was to mitigate the exuberant immune response observed in some patients infected with SARS-CoV-2 (also called cytokine storm or cytokine release syndrome).
Quite quickly, we started noticing that giving the drug to our sickest patients wasn’t eliciting dramatic improvement. We reasoned that by the time patients were severely ill and requiring ventilators, the damage to their lungs from the cytokine storm had already taken place. It was like closing the barn door after the horse had already bolted.
Dr. Sinha[/caption]
Pranay Sinha, MD
Research Fellow
Section of Infectious Diseases
Boston University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: In the early days of the COVID-19 pandemic there were no evidence-based treatments for severely ill patients infected with this virus. We formed an interdisciplinary group of physicians from departments of adult and pediatric infectious diseases, rheumatology, and pulmonary/critical care as well as clinical pharmacy specialists. Given some promising data from China, we instituted treatment with off-label IL-6 receptor inhibitors (tocilizumab and sarilumab). The rationale was to mitigate the exuberant immune response observed in some patients infected with SARS-CoV-2 (also called cytokine storm or cytokine release syndrome).
Quite quickly, we started noticing that giving the drug to our sickest patients wasn’t eliciting dramatic improvement. We reasoned that by the time patients were severely ill and requiring ventilators, the damage to their lungs from the cytokine storm had already taken place. It was like closing the barn door after the horse had already bolted.


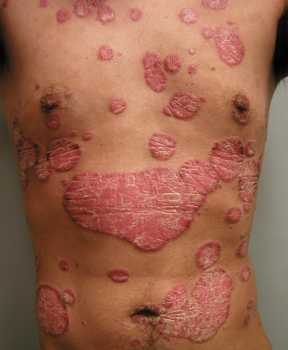
 Dr. Bollag[/caption]
Wendy Bollag, PhD, FAHA
Professor of Physiology
VA Research Career Scientist
Augusta University, Georgia
MedicalResearch.com: What is the background for this study?
Response: We have previously shown that the lipid (fat) phosphatidylglycerol (PG) is able to inhibit rapidly growing keratinocytes (skin cells) and promote their maturation. We also found that PG can suppress skin inflammation.
Since the common skin disease psoriasis is characterized by inflammation and excessive growth and abnormal maturation of skin cells, we believed that PG might be useful as a treatment. However, the mechanism of its anti-inflammatory effect was unknown. PG in the lung has been found to inhibit inflammation induced by microbes or their components, which work by activating the innate immune system via binding to proteins called toll-like receptors (TLRs); however, psoriasis is not considered to be an infectious disease.
We hypothesized that PG would also inhibit inflammation induced by anti-microbial peptides that activate TLRs. Anti-microbial peptides, produced normally by the skin to protect against infection, are known to be excessively up-regulated in psoriatic skin.
Dr. Bollag[/caption]
Wendy Bollag, PhD, FAHA
Professor of Physiology
VA Research Career Scientist
Augusta University, Georgia
MedicalResearch.com: What is the background for this study?
Response: We have previously shown that the lipid (fat) phosphatidylglycerol (PG) is able to inhibit rapidly growing keratinocytes (skin cells) and promote their maturation. We also found that PG can suppress skin inflammation.
Since the common skin disease psoriasis is characterized by inflammation and excessive growth and abnormal maturation of skin cells, we believed that PG might be useful as a treatment. However, the mechanism of its anti-inflammatory effect was unknown. PG in the lung has been found to inhibit inflammation induced by microbes or their components, which work by activating the innate immune system via binding to proteins called toll-like receptors (TLRs); however, psoriasis is not considered to be an infectious disease.
We hypothesized that PG would also inhibit inflammation induced by anti-microbial peptides that activate TLRs. Anti-microbial peptides, produced normally by the skin to protect against infection, are known to be excessively up-regulated in psoriatic skin. 






 Dr. Schulert[/caption]
MedicalResearch.com Interview
Grant S Schulert MD, PhD
Clinical Fellow, Division of Rheumatology
Cincinnati Childrens Hospital
Medical Research: What is the background for this study? What are the main findings?
Dr. Schulert: Influenza infection causes millions of illnesses annually, but most of those are relatively mild. In a subset of cases, patients can become critically ill, even if they are relatively young and healthy. Several previous reports had observed in these critically ill patients features of a hyperinflammatory syndrome known as HLH (hemophagocytic lymphohistiocytosis) or MAS (macrophage activation syndrome). This hyperinflammation can be triggered by other infections as well as in a subtype of juvenile arthritis, but there is also a familial form occurring in early childhood with known genetic causes. Our questions with this study were
1) how often are features consistent with HLH/MAS seen in fatal H1N1 influenza infections and
2) do patients with fatal H1N1 infection have genetic mutations associated with HLH/MAS?
Our collaborator Paul Harms, MD, and his team at the Michigan Center for Translational Pathology, University of Michigan Medical School identified 16 cases of fatal H1N1 influenza infection. Based on their clinical features, between 41-88% of these patients could be categorized as having a hyperinflammatory HLH/MAS. We then used processed tissue samples from the patients for whole exome genetic sequencing, which reads the entire genetic code of every gene in a person. Five patients carried mutations in genes which cause HLH, and several others carried mutations in genes linked to MAS. This suggests that there may be genetic risk factors for developing fatal hyperinflammatory syndromes in H1N1 infection.
Dr. Schulert[/caption]
MedicalResearch.com Interview
Grant S Schulert MD, PhD
Clinical Fellow, Division of Rheumatology
Cincinnati Childrens Hospital
Medical Research: What is the background for this study? What are the main findings?
Dr. Schulert: Influenza infection causes millions of illnesses annually, but most of those are relatively mild. In a subset of cases, patients can become critically ill, even if they are relatively young and healthy. Several previous reports had observed in these critically ill patients features of a hyperinflammatory syndrome known as HLH (hemophagocytic lymphohistiocytosis) or MAS (macrophage activation syndrome). This hyperinflammation can be triggered by other infections as well as in a subtype of juvenile arthritis, but there is also a familial form occurring in early childhood with known genetic causes. Our questions with this study were
1) how often are features consistent with HLH/MAS seen in fatal H1N1 influenza infections and
2) do patients with fatal H1N1 infection have genetic mutations associated with HLH/MAS?
Our collaborator Paul Harms, MD, and his team at the Michigan Center for Translational Pathology, University of Michigan Medical School identified 16 cases of fatal H1N1 influenza infection. Based on their clinical features, between 41-88% of these patients could be categorized as having a hyperinflammatory HLH/MAS. We then used processed tissue samples from the patients for whole exome genetic sequencing, which reads the entire genetic code of every gene in a person. Five patients carried mutations in genes which cause HLH, and several others carried mutations in genes linked to MAS. This suggests that there may be genetic risk factors for developing fatal hyperinflammatory syndromes in H1N1 infection.

