Author Interviews, Dermatology, Nutrition / 05.11.2025
Can Diet or Lifestyle Changes Help Control Psoriasis?
[caption id="attachment_71293" align="aligncenter" width="267"] Photo by Karola G[/caption]
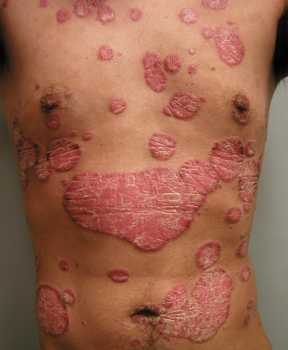
Can certain foods actually reduce the thick, scaly patches that characterize psoriasis? While medications remain the primary treatment, certain dietary changes and lifestyle modifications may help reduce flare frequency and severity by modulating inflammation pathways and immune system responses.
If you’re managing chronic psoriasis, visiting a psoriasis clinic Singapore may help you understand how diet and lifestyle changes complement medical treatments. The chronic inflammatory nature of psoriasis means daily choices may influence symptom patterns. Foods consumed, stress levels, and lifestyle habits can all affect the inflammatory cascade that drives skin cell overproduction.
Photo by Karola G[/caption]
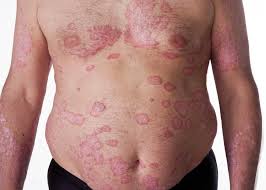
Can certain foods actually reduce the thick, scaly patches that characterize psoriasis? While medications remain the primary treatment, certain dietary changes and lifestyle modifications may help reduce flare frequency and severity by modulating inflammation pathways and immune system responses.
If you’re managing chronic psoriasis, visiting a psoriasis clinic Singapore may help you understand how diet and lifestyle changes complement medical treatments. The chronic inflammatory nature of psoriasis means daily choices may influence symptom patterns. Foods consumed, stress levels, and lifestyle habits can all affect the inflammatory cascade that drives skin cell overproduction.
 Photo by Karola G[/caption]
Can certain foods actually reduce the thick, scaly patches that characterize psoriasis? While medications remain the primary treatment, certain dietary changes and lifestyle modifications may help reduce flare frequency and severity by modulating inflammation pathways and immune system responses.
If you’re managing chronic psoriasis, visiting a psoriasis clinic Singapore may help you understand how diet and lifestyle changes complement medical treatments. The chronic inflammatory nature of psoriasis means daily choices may influence symptom patterns. Foods consumed, stress levels, and lifestyle habits can all affect the inflammatory cascade that drives skin cell overproduction.
Photo by Karola G[/caption]
Can certain foods actually reduce the thick, scaly patches that characterize psoriasis? While medications remain the primary treatment, certain dietary changes and lifestyle modifications may help reduce flare frequency and severity by modulating inflammation pathways and immune system responses.
If you’re managing chronic psoriasis, visiting a psoriasis clinic Singapore may help you understand how diet and lifestyle changes complement medical treatments. The chronic inflammatory nature of psoriasis means daily choices may influence symptom patterns. Foods consumed, stress levels, and lifestyle habits can all affect the inflammatory cascade that drives skin cell overproduction.




 Lara van der Schoot[/caption]
Lara van der Schoot
MD, PhD candidate
Department of Dermatology
Radboud University Medical Center
Nijmegen, The Netherlands
MedicalResearch.com: What is the background for this study?
Response: Psoriasis is a chronic, immune mediated skin disease for which effective targeted biological agents have become available the past years. Inherent to their immunomodulatory mechanism of action, biologics might increase infections risk. We know from clinical trial data that respiratory tract infections are among the most common adverse events during biologic treatment, but real-world data is sparse. Regarding the risk of serious infections among biologic users, mostly defined as infections requiring hospitalization, previous studies provided different results and there is limited comparative data for the newer biologics available.
The COVID-19 pandemic turned attention to the risk of infections among biologic users, especially for respiratory tract infections, as they might relate to susceptibility for viral respiratory tract infections such as COVID-19.
In our study, the primary aim was to determine the risk of respiratory tract infections among real-world psoriasis patients treated with biologics, including the newer IL-17 and IL-23 inhibitors. The secondary aim was to assess risk of serious infections in this cohort. Additionally, rates of SARS-CoV-2 infections were assessed.
Lara van der Schoot[/caption]
Lara van der Schoot
MD, PhD candidate
Department of Dermatology
Radboud University Medical Center
Nijmegen, The Netherlands
MedicalResearch.com: What is the background for this study?
Response: Psoriasis is a chronic, immune mediated skin disease for which effective targeted biological agents have become available the past years. Inherent to their immunomodulatory mechanism of action, biologics might increase infections risk. We know from clinical trial data that respiratory tract infections are among the most common adverse events during biologic treatment, but real-world data is sparse. Regarding the risk of serious infections among biologic users, mostly defined as infections requiring hospitalization, previous studies provided different results and there is limited comparative data for the newer biologics available.
The COVID-19 pandemic turned attention to the risk of infections among biologic users, especially for respiratory tract infections, as they might relate to susceptibility for viral respiratory tract infections such as COVID-19.
In our study, the primary aim was to determine the risk of respiratory tract infections among real-world psoriasis patients treated with biologics, including the newer IL-17 and IL-23 inhibitors. The secondary aim was to assess risk of serious infections in this cohort. Additionally, rates of SARS-CoV-2 infections were assessed.

 Prof. Reich[/caption]
Prof. Kristian Reich, MD, PhD
Professor for Translational Research in Inflammatory Skin Diseases
Institute for Health Services Research in Dermatology and Nursing
University Medical Center Hamburg-Eppendorf
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Complete skin clearance is an important treatment goal for patients with psoriasis and is closely associated with treatment satisfaction and improved quality of life. However, it remains an unmet need for many patients.
The interleukin (IL)-17 isoforms IL-17A and IL-17F play central roles in psoriasis pathophysiology and are overexpressed in psoriatic tissues. Existing biologic therapies, such as secukinumab, inhibit IL-17A only. However, increasing evidence indicates that IL-17F contributes independently to the pathobiology of plaque psoriasis, and that blocking both IL-17A and IL-17F may lead to more complete suppression of inflammation and superior clinical outcomes, compared with blocking IL‑17A alone.
Bimekizumab is a humanized monoclonal IgG1 antibody that has been designed to selectively inhibit IL-17F in addition to IL-17A.
Prof. Reich[/caption]
Prof. Kristian Reich, MD, PhD
Professor for Translational Research in Inflammatory Skin Diseases
Institute for Health Services Research in Dermatology and Nursing
University Medical Center Hamburg-Eppendorf
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Complete skin clearance is an important treatment goal for patients with psoriasis and is closely associated with treatment satisfaction and improved quality of life. However, it remains an unmet need for many patients.
The interleukin (IL)-17 isoforms IL-17A and IL-17F play central roles in psoriasis pathophysiology and are overexpressed in psoriatic tissues. Existing biologic therapies, such as secukinumab, inhibit IL-17A only. However, increasing evidence indicates that IL-17F contributes independently to the pathobiology of plaque psoriasis, and that blocking both IL-17A and IL-17F may lead to more complete suppression of inflammation and superior clinical outcomes, compared with blocking IL‑17A alone.
Bimekizumab is a humanized monoclonal IgG1 antibody that has been designed to selectively inhibit IL-17F in addition to IL-17A.

 Dr. Maslin[/caption]
Dr. Douglas Maslin, MPhil, MB BCHir
Dermatologist and Pharmacologist
Addenbrooke's Hospital
Cambridge, UK
MedicalResearch.com: What is the background for this study?
Response: I’d like to answer this question in three parts:
Dr. Maslin[/caption]
Dr. Douglas Maslin, MPhil, MB BCHir
Dermatologist and Pharmacologist
Addenbrooke's Hospital
Cambridge, UK
MedicalResearch.com: What is the background for this study?
Response: I’d like to answer this question in three parts:



 Prof Ching-Chi Chi,[/caption]
Prof Ching-Chi Chi, MD, MMS, DPhil (Oxford)
Department of Dermatology
Chang Gung Memorial Hospital, Linkou
Taiwan
MedicalResearch.com: What is the background for this study?
Response: Psoriasis has been associated various inflammatory comorbidities including diabetes mellitus, coronary artery disease, etc. Moreover, obesity is prevalent among psoriasis patients and has been considered as an independent risk factor for occurrence and worsening of psoriasis by promoting systemic inflammation.
Notably, body weight (BW) gain of psoriasis patients after biologics use has been observed. However, there are inconsistent reports on whether biological therapy relates to BW gain.
Prof Ching-Chi Chi,[/caption]
Prof Ching-Chi Chi, MD, MMS, DPhil (Oxford)
Department of Dermatology
Chang Gung Memorial Hospital, Linkou
Taiwan
MedicalResearch.com: What is the background for this study?
Response: Psoriasis has been associated various inflammatory comorbidities including diabetes mellitus, coronary artery disease, etc. Moreover, obesity is prevalent among psoriasis patients and has been considered as an independent risk factor for occurrence and worsening of psoriasis by promoting systemic inflammation.
Notably, body weight (BW) gain of psoriasis patients after biologics use has been observed. However, there are inconsistent reports on whether biological therapy relates to BW gain. 

 Dr. Robinson[/caption]
Anne Robinson, Pharm D
Executive Scientific Director
AbbVie
MedicalResearch.com: What is the background for the risankizumab data presented at the American Academy of Dermatology 2019 Annual Meeting?
Response: Abstracts presented by AbbVie at the American Academy of Dermatology (AAD) 2019 Annual Meeting highlight additional data from the Phase 3 clinical trial program evaluating the safety and efficacy of risankizumab, an investigational interleukin-23 (IL-23) inhibitor. The registrational program for risankizumab evaluated more than 2,000 adult patients with moderate to severe plaque psoriasis across four pivotal studies.
Dr. Robinson[/caption]
Anne Robinson, Pharm D
Executive Scientific Director
AbbVie
MedicalResearch.com: What is the background for the risankizumab data presented at the American Academy of Dermatology 2019 Annual Meeting?
Response: Abstracts presented by AbbVie at the American Academy of Dermatology (AAD) 2019 Annual Meeting highlight additional data from the Phase 3 clinical trial program evaluating the safety and efficacy of risankizumab, an investigational interleukin-23 (IL-23) inhibitor. The registrational program for risankizumab evaluated more than 2,000 adult patients with moderate to severe plaque psoriasis across four pivotal studies.
 Dr. Bollag[/caption]
Wendy Bollag, PhD, FAHA
Professor of Physiology
VA Research Career Scientist
Augusta University, Georgia
MedicalResearch.com: What is the background for this study?
Response: We have previously shown that the lipid (fat) phosphatidylglycerol (PG) is able to inhibit rapidly growing keratinocytes (skin cells) and promote their maturation. We also found that PG can suppress skin inflammation.
Since the common skin disease psoriasis is characterized by inflammation and excessive growth and abnormal maturation of skin cells, we believed that PG might be useful as a treatment. However, the mechanism of its anti-inflammatory effect was unknown. PG in the lung has been found to inhibit inflammation induced by microbes or their components, which work by activating the innate immune system via binding to proteins called toll-like receptors (TLRs); however, psoriasis is not considered to be an infectious disease.
We hypothesized that PG would also inhibit inflammation induced by anti-microbial peptides that activate TLRs. Anti-microbial peptides, produced normally by the skin to protect against infection, are known to be excessively up-regulated in psoriatic skin.
Dr. Bollag[/caption]
Wendy Bollag, PhD, FAHA
Professor of Physiology
VA Research Career Scientist
Augusta University, Georgia
MedicalResearch.com: What is the background for this study?
Response: We have previously shown that the lipid (fat) phosphatidylglycerol (PG) is able to inhibit rapidly growing keratinocytes (skin cells) and promote their maturation. We also found that PG can suppress skin inflammation.
Since the common skin disease psoriasis is characterized by inflammation and excessive growth and abnormal maturation of skin cells, we believed that PG might be useful as a treatment. However, the mechanism of its anti-inflammatory effect was unknown. PG in the lung has been found to inhibit inflammation induced by microbes or their components, which work by activating the innate immune system via binding to proteins called toll-like receptors (TLRs); however, psoriasis is not considered to be an infectious disease.
We hypothesized that PG would also inhibit inflammation induced by anti-microbial peptides that activate TLRs. Anti-microbial peptides, produced normally by the skin to protect against infection, are known to be excessively up-regulated in psoriatic skin. 







