Alzheimer's - Dementia, Author Interviews, Cost of Health Care, Medicare, UCLA / 15.10.2024
Wasted Alzheimer’s Drug Can Cost Medicare Hundreds of Millions Annually
MedicalResearch.com Interview with:
[caption id="attachment_64014" align="alignleft" width="150"] Frank F Zhou[/caption]
Frank F. Zhou | he/him
MD Candidate, Class of 2025
David Geffen School of Medicine at UCLA
MedicalResearch.com: What is the background for this study? What is Lecanemab used for? How is it given to patients?
Response: Lecanemab is a new infusion therapy for Alzheimer's disease. Its dosing is based on each patient's body weight (10 mg/kg every two weeks), but the drug is only available in 500 mg and 200 mg single-use vials, meaning that any leftover drug in vials must be thrown away. Given that lecanemab is expected to cost Medicare billions of dollars each year, we hypothesized that discarded drug could result in significant wasteful spending.
Frank F Zhou[/caption]
Frank F. Zhou | he/him
MD Candidate, Class of 2025
David Geffen School of Medicine at UCLA
MedicalResearch.com: What is the background for this study? What is Lecanemab used for? How is it given to patients?
Response: Lecanemab is a new infusion therapy for Alzheimer's disease. Its dosing is based on each patient's body weight (10 mg/kg every two weeks), but the drug is only available in 500 mg and 200 mg single-use vials, meaning that any leftover drug in vials must be thrown away. Given that lecanemab is expected to cost Medicare billions of dollars each year, we hypothesized that discarded drug could result in significant wasteful spending.
 Frank F Zhou[/caption]
Frank F. Zhou | he/him
MD Candidate, Class of 2025
David Geffen School of Medicine at UCLA
MedicalResearch.com: What is the background for this study? What is Lecanemab used for? How is it given to patients?
Response: Lecanemab is a new infusion therapy for Alzheimer's disease. Its dosing is based on each patient's body weight (10 mg/kg every two weeks), but the drug is only available in 500 mg and 200 mg single-use vials, meaning that any leftover drug in vials must be thrown away. Given that lecanemab is expected to cost Medicare billions of dollars each year, we hypothesized that discarded drug could result in significant wasteful spending.
Frank F Zhou[/caption]
Frank F. Zhou | he/him
MD Candidate, Class of 2025
David Geffen School of Medicine at UCLA
MedicalResearch.com: What is the background for this study? What is Lecanemab used for? How is it given to patients?
Response: Lecanemab is a new infusion therapy for Alzheimer's disease. Its dosing is based on each patient's body weight (10 mg/kg every two weeks), but the drug is only available in 500 mg and 200 mg single-use vials, meaning that any leftover drug in vials must be thrown away. Given that lecanemab is expected to cost Medicare billions of dollars each year, we hypothesized that discarded drug could result in significant wasteful spending.



 Dr. Callaghan[/caption]
Bridget Callaghan Ph.D.
Assistant Professor of Psychology
UCLA
Dr. Callahan studies interactions between mental and physical health across development.
MedicalResearch.com: What is the background for this study?
Response: A growing body of evidence links the gut microbiome to brain and immune functioning, and changes to that community of microorganisms is likely among the ways that hardship affects children’s socioemotional development.
Limited evidence in humans has demonstrated the adversities experienced prenatally and during early life influence the composition of the gut microbiome, but no studies had examined whether stress experienced in a mother's own childhood could influence the microbiome of the next generation of children.
Dr. Callaghan[/caption]
Bridget Callaghan Ph.D.
Assistant Professor of Psychology
UCLA
Dr. Callahan studies interactions between mental and physical health across development.
MedicalResearch.com: What is the background for this study?
Response: A growing body of evidence links the gut microbiome to brain and immune functioning, and changes to that community of microorganisms is likely among the ways that hardship affects children’s socioemotional development.
Limited evidence in humans has demonstrated the adversities experienced prenatally and during early life influence the composition of the gut microbiome, but no studies had examined whether stress experienced in a mother's own childhood could influence the microbiome of the next generation of children.

 Julia Cave Arbanas[/caption]
Julia Cave Arbanas
Project Manager and
Julia Cave Arbanas[/caption]
Julia Cave Arbanas
Project Manager and
 Dr. D'Orsogna[/caption]
Maria-Rita D'Orsogna Ph.D.
Professor, Mathematics
California State University, Northridge
Adjunct Associate Professor
Department of Computational Medicine at UCLA
MedicalResearch.com: What is the background for this study?
Response: Drug overdose deaths have been increasing in the USA for the past two decades. A ‘third wave’ of overdose fatalities started in 2013, with a shift from prescription opioids towards synthetic ones, in particular illicit fentanyl.
To examine trends in drug overdose deaths by gender, race and geography in the United States during the period 2013-2020, we used an epidemiological database provided by the Centers for Disease Control and Prevention, extracting rates by race and gender in all 50 states plus the District of Columbia. We considered the impact of four main drug categories psychostimulants with addiction potential such as methamphetamines; heroin; prescription opioids and synthetic opioids such as fentanyl and its derivatives.
Dr. D'Orsogna[/caption]
Maria-Rita D'Orsogna Ph.D.
Professor, Mathematics
California State University, Northridge
Adjunct Associate Professor
Department of Computational Medicine at UCLA
MedicalResearch.com: What is the background for this study?
Response: Drug overdose deaths have been increasing in the USA for the past two decades. A ‘third wave’ of overdose fatalities started in 2013, with a shift from prescription opioids towards synthetic ones, in particular illicit fentanyl.
To examine trends in drug overdose deaths by gender, race and geography in the United States during the period 2013-2020, we used an epidemiological database provided by the Centers for Disease Control and Prevention, extracting rates by race and gender in all 50 states plus the District of Columbia. We considered the impact of four main drug categories psychostimulants with addiction potential such as methamphetamines; heroin; prescription opioids and synthetic opioids such as fentanyl and its derivatives.

 Dr. Wong[/caption]
Mitchell Wong, MD PhD
Professor of Medicine
Executive Vice Chair for Research Training
Department of Medicine
Executive Co-Director, Specialty Training and Advanced Research (STAR) Program
Director, UCLA CTSI KL2 Program
UCLA Division of General Internal Medicine and Health Services Research Los Angeles, CA 90024
MedicalResearch.com: What is the background for this study?
Response: It is estimated that social factors like poverty, education, and housing have a large impact on health. Yet, there are few interventions that exist to directly address those issues. Schools are a promising solution since society already invests heavily in education and schools are an everyday part of most children’s lives.
Dr. Wong[/caption]
Mitchell Wong, MD PhD
Professor of Medicine
Executive Vice Chair for Research Training
Department of Medicine
Executive Co-Director, Specialty Training and Advanced Research (STAR) Program
Director, UCLA CTSI KL2 Program
UCLA Division of General Internal Medicine and Health Services Research Los Angeles, CA 90024
MedicalResearch.com: What is the background for this study?
Response: It is estimated that social factors like poverty, education, and housing have a large impact on health. Yet, there are few interventions that exist to directly address those issues. Schools are a promising solution since society already invests heavily in education and schools are an everyday part of most children’s lives.
 Dr. Dan P. Ly[/caption]
Division of General Internal Medicine and Health Services Research
David Geffen School of Medicine at UCLA
Los Angeles, CA
MedicalResearch.com: What is the background for this study?
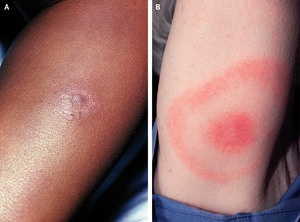
Response: Lyme disease presents first on the skin with the classic “bull’s-eye” rash. But such rashes in Black patients aren’t well-represented in medical textbooks. This may lead to physicians not recognizing such rashes in Black patients.
As a result, Black patients are more likely to present with later complications of Lyme disease when first diagnosed such as neurologic complications.
Dr. Dan P. Ly[/caption]
Division of General Internal Medicine and Health Services Research
David Geffen School of Medicine at UCLA
Los Angeles, CA
MedicalResearch.com: What is the background for this study?
Response: Lyme disease presents first on the skin with the classic “bull’s-eye” rash. But such rashes in Black patients aren’t well-represented in medical textbooks. This may lead to physicians not recognizing such rashes in Black patients.
As a result, Black patients are more likely to present with later complications of Lyme disease when first diagnosed such as neurologic complications. 


 Dr. Lee[/caption]
Brian P. Lee, MD, MAS
Assistant Professor Clinical Medicine
University of Southern Californi
Keck School of Medicine
Los Angeles, California
MedicalResearch.com: What is the background for this study?
Response: The COVID-19 pandemic has been associated with mental health stressors, including anxiety, loneliness, and social instability. We hypothesized the pandemic may have led to increased alcohol and tobacco use as a coping mechanism for these stressors. National retrospective questionnaires had suggested higher reports of substance use, but these are limited by selection and recall biases, in addition to subjective report – we sought to address this knowledge gap by using a nationally-representative longitudinal cohort (Nielsen National Consumer Panel) tracking real-time purchases of households across the US.
Dr. Lee[/caption]
Brian P. Lee, MD, MAS
Assistant Professor Clinical Medicine
University of Southern Californi
Keck School of Medicine
Los Angeles, California
MedicalResearch.com: What is the background for this study?
Response: The COVID-19 pandemic has been associated with mental health stressors, including anxiety, loneliness, and social instability. We hypothesized the pandemic may have led to increased alcohol and tobacco use as a coping mechanism for these stressors. National retrospective questionnaires had suggested higher reports of substance use, but these are limited by selection and recall biases, in addition to subjective report – we sought to address this knowledge gap by using a nationally-representative longitudinal cohort (Nielsen National Consumer Panel) tracking real-time purchases of households across the US. 
 Dr. Zimmerman[/caption]
Frederick Zimmerman, PhD
Professor, Department of Health Policy and Management
Fielding School of Public Health
UCLA
MedicalResearch.com: What is the background for this study?
Response: The science on school transmissions of COVID is becoming clearer all the time in its conclusion that there is little to no transmission in school environments as long as reasonable precautions are taken. Yet one recent study got a lot of attention for claiming that states that allowed their schools to remain open in the early days of the pandemic saw more cases. That study did not control for several important factors that might explain this association, so our study aimed to correct that work.
Dr. Zimmerman[/caption]
Frederick Zimmerman, PhD
Professor, Department of Health Policy and Management
Fielding School of Public Health
UCLA
MedicalResearch.com: What is the background for this study?
Response: The science on school transmissions of COVID is becoming clearer all the time in its conclusion that there is little to no transmission in school environments as long as reasonable precautions are taken. Yet one recent study got a lot of attention for claiming that states that allowed their schools to remain open in the early days of the pandemic saw more cases. That study did not control for several important factors that might explain this association, so our study aimed to correct that work.






 Dr. Childers[/caption]
Chris Childers, MD, PhD
Division of General Surgery
David Geffen School of Medicine at UCLA
Los Angeles, CA 90095
MedicalResearch.com: What is the background for this study?
Response: Patients with end-stage renal disease – poorly functioning kidneys – often have to receive dialysis. This typically requires a patient to visit an outpatient clinic several times a week to have their blood filtered by a machine. Over the past few years, two for-profit companies have increased their control over the outpatient dialysis market – DaVita and Fresenius. Combined they control approximately ¾ of the market. A number of concerns have been raised against these for-profit companies suggesting that the quality of care they deliver may be worse than the care delivered at not-for-profit companies. But, because they control so much of the market and because patients have to receive dialysis so frequently, patients may not have much choice in the clinic they visit.
Medicare covers patients who are 65 years or older and also patients on dialysis regardless of age. Medicare pays a fixed rate for dialysis which they believe is adequate to cover the clinics' costs. However, if a patient also has private insurance, the insurer is required to pay for dialysis instead of Medicare. Whereas Medicare rates are fixed by the federal government, private insurers have to negotiate the price they pay, and may pay much more as a result.
Dr. Childers[/caption]
Chris Childers, MD, PhD
Division of General Surgery
David Geffen School of Medicine at UCLA
Los Angeles, CA 90095
MedicalResearch.com: What is the background for this study?
Response: Patients with end-stage renal disease – poorly functioning kidneys – often have to receive dialysis. This typically requires a patient to visit an outpatient clinic several times a week to have their blood filtered by a machine. Over the past few years, two for-profit companies have increased their control over the outpatient dialysis market – DaVita and Fresenius. Combined they control approximately ¾ of the market. A number of concerns have been raised against these for-profit companies suggesting that the quality of care they deliver may be worse than the care delivered at not-for-profit companies. But, because they control so much of the market and because patients have to receive dialysis so frequently, patients may not have much choice in the clinic they visit.
Medicare covers patients who are 65 years or older and also patients on dialysis regardless of age. Medicare pays a fixed rate for dialysis which they believe is adequate to cover the clinics' costs. However, if a patient also has private insurance, the insurer is required to pay for dialysis instead of Medicare. Whereas Medicare rates are fixed by the federal government, private insurers have to negotiate the price they pay, and may pay much more as a result.









