Author Interviews, COVID -19 Coronavirus, Race/Ethnic Diversity, Vaccine Studies / 10.02.2023
Factors Influencing COVID-19 Vaccine Hesitancy in Eastern Pennsylvania
MedicalResearch.com Interview with:
[caption id="attachment_60007" align="alignleft" width="125"] Kenya Colvin[/caption]
Kenya Colvin, MBS
Department of Medical Education
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: Vaccine hesitancy is a major driver of COVID-19 vaccination disparities between minority and non-Hispanic White communities. Our goal was to understand what factors influenced vaccine hesitancy among individuals in Eastern Pennsylvania to identify more effective ways to promote vaccine uptake within minority communities.
Kenya Colvin[/caption]
Kenya Colvin, MBS
Department of Medical Education
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: Vaccine hesitancy is a major driver of COVID-19 vaccination disparities between minority and non-Hispanic White communities. Our goal was to understand what factors influenced vaccine hesitancy among individuals in Eastern Pennsylvania to identify more effective ways to promote vaccine uptake within minority communities.
 Kenya Colvin[/caption]
Kenya Colvin, MBS
Department of Medical Education
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: Vaccine hesitancy is a major driver of COVID-19 vaccination disparities between minority and non-Hispanic White communities. Our goal was to understand what factors influenced vaccine hesitancy among individuals in Eastern Pennsylvania to identify more effective ways to promote vaccine uptake within minority communities.
Kenya Colvin[/caption]
Kenya Colvin, MBS
Department of Medical Education
Scranton, PA
MedicalResearch.com: What is the background for this study?
Response: Vaccine hesitancy is a major driver of COVID-19 vaccination disparities between minority and non-Hispanic White communities. Our goal was to understand what factors influenced vaccine hesitancy among individuals in Eastern Pennsylvania to identify more effective ways to promote vaccine uptake within minority communities.




 Dr. Cheng-Ying Ho[/caption]
Cheng-Ying Ho, MD, PhD
Associate Professor
Department of Pathology
Johns Hopkins University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: Smell loss is one of the most common symptoms of COVID-19 infection. The mechanism of COVID-19-related smell loss is unclear. Previous studies mainly focused on the effect of the viral infection on the lining of the nasal cavity. We went a step beyond to examine the olfactory bulb, a region that transmits smell-related signals to the brain.
Dr. Cheng-Ying Ho[/caption]
Cheng-Ying Ho, MD, PhD
Associate Professor
Department of Pathology
Johns Hopkins University School of Medicine
MedicalResearch.com: What is the background for this study?
Response: Smell loss is one of the most common symptoms of COVID-19 infection. The mechanism of COVID-19-related smell loss is unclear. Previous studies mainly focused on the effect of the viral infection on the lining of the nasal cavity. We went a step beyond to examine the olfactory bulb, a region that transmits smell-related signals to the brain.




 Dr. Pollitt[/caption]
Krystal Pollitt, PhD, P.Eng.
Assistant Professor of Epidemiology (Environmental Health Sciences)
Assistant Professor in Chemical and Environmental Engineering
Affiliated Faculty, Yale Institute for Global Health
Yale School of Public Health
MedicalResearch.com: What is the background for this study?
Response: People infected with COVID-19 can release SARS-CoV-2 virus in aerosol and droplets when they exhale. This can be from coughing or sneezing but also when they speaker or just breathe. While the larger droplets can settle to the ground quickly (seconds to minutes), smaller aerosol can remain in the air in longer periods (minutes to hours). SARS-CoV-2 can be transmitted by inhaling aerosol or droplets containing infectious virus. The Fresh Air Clip enables detection of droplet and aerosol containing virus.
Dr. Pollitt[/caption]
Krystal Pollitt, PhD, P.Eng.
Assistant Professor of Epidemiology (Environmental Health Sciences)
Assistant Professor in Chemical and Environmental Engineering
Affiliated Faculty, Yale Institute for Global Health
Yale School of Public Health
MedicalResearch.com: What is the background for this study?
Response: People infected with COVID-19 can release SARS-CoV-2 virus in aerosol and droplets when they exhale. This can be from coughing or sneezing but also when they speaker or just breathe. While the larger droplets can settle to the ground quickly (seconds to minutes), smaller aerosol can remain in the air in longer periods (minutes to hours). SARS-CoV-2 can be transmitted by inhaling aerosol or droplets containing infectious virus. The Fresh Air Clip enables detection of droplet and aerosol containing virus.



 Dr. Israel[/caption]
Ariel Israel, M.D., Ph.D.
Director, Leumit Health Services
Tel Aviv, Israel
MedicalResearch.com: What is the background for this study?
Response: As a research institute of Leumit, one of the four state mandated health funds in Israel, we pursue research projects aimed at improving the health of our members, and reducing the burden of disease. For this purpose, we harness the unique resource of the electronic health records of our members, that is available in a central data warehouse for research purposes.
Israel was one of the first countries to roll-out a large-scale vaccination campaign, and to achieve control of the pandemics through vaccination. Nevertheless, since the middle of June '21, we have observed a gradual increase in the rate of COVID-19 infections among our members, even among the vaccinated. This increase was first believed to be due to the emergence of the delta strain, but when we compared vaccinated individuals who suffered from breakthrough infections to other vaccinated individuals, we found that the time that has elapsed since vaccination was significantly longer for individuals who got infected with COVID-19, in each of the age groups.
This prompted us to investigate the issue of a possible waning effect of the vaccine protection with time, that we present in this report, using the test negative study design.
We examined the electronic health records for 80,057 adults (average age 44 years) who received a PCR test at least three weeks after their second injection, and had no evidence of previous covid-19 infection. Of these 80,057 participants, 7,973 (9.6%) had a positive test result. These individuals were then matched to negative controls of the same age and ethnic group who were tested in the same week.
Dr. Israel[/caption]
Ariel Israel, M.D., Ph.D.
Director, Leumit Health Services
Tel Aviv, Israel
MedicalResearch.com: What is the background for this study?
Response: As a research institute of Leumit, one of the four state mandated health funds in Israel, we pursue research projects aimed at improving the health of our members, and reducing the burden of disease. For this purpose, we harness the unique resource of the electronic health records of our members, that is available in a central data warehouse for research purposes.
Israel was one of the first countries to roll-out a large-scale vaccination campaign, and to achieve control of the pandemics through vaccination. Nevertheless, since the middle of June '21, we have observed a gradual increase in the rate of COVID-19 infections among our members, even among the vaccinated. This increase was first believed to be due to the emergence of the delta strain, but when we compared vaccinated individuals who suffered from breakthrough infections to other vaccinated individuals, we found that the time that has elapsed since vaccination was significantly longer for individuals who got infected with COVID-19, in each of the age groups.
This prompted us to investigate the issue of a possible waning effect of the vaccine protection with time, that we present in this report, using the test negative study design.
We examined the electronic health records for 80,057 adults (average age 44 years) who received a PCR test at least three weeks after their second injection, and had no evidence of previous covid-19 infection. Of these 80,057 participants, 7,973 (9.6%) had a positive test result. These individuals were then matched to negative controls of the same age and ethnic group who were tested in the same week.

 Lara van der Schoot[/caption]
Lara van der Schoot
MD, PhD candidate
Department of Dermatology
Radboud University Medical Center
Nijmegen, The Netherlands
MedicalResearch.com: What is the background for this study?
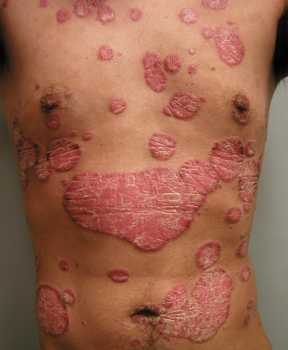
Response: Psoriasis is a chronic, immune mediated skin disease for which effective targeted biological agents have become available the past years. Inherent to their immunomodulatory mechanism of action, biologics might increase infections risk. We know from clinical trial data that respiratory tract infections are among the most common adverse events during biologic treatment, but real-world data is sparse. Regarding the risk of serious infections among biologic users, mostly defined as infections requiring hospitalization, previous studies provided different results and there is limited comparative data for the newer biologics available.
The COVID-19 pandemic turned attention to the risk of infections among biologic users, especially for respiratory tract infections, as they might relate to susceptibility for viral respiratory tract infections such as COVID-19.
In our study, the primary aim was to determine the risk of respiratory tract infections among real-world psoriasis patients treated with biologics, including the newer IL-17 and IL-23 inhibitors. The secondary aim was to assess risk of serious infections in this cohort. Additionally, rates of SARS-CoV-2 infections were assessed.
Lara van der Schoot[/caption]
Lara van der Schoot
MD, PhD candidate
Department of Dermatology
Radboud University Medical Center
Nijmegen, The Netherlands
MedicalResearch.com: What is the background for this study?
Response: Psoriasis is a chronic, immune mediated skin disease for which effective targeted biological agents have become available the past years. Inherent to their immunomodulatory mechanism of action, biologics might increase infections risk. We know from clinical trial data that respiratory tract infections are among the most common adverse events during biologic treatment, but real-world data is sparse. Regarding the risk of serious infections among biologic users, mostly defined as infections requiring hospitalization, previous studies provided different results and there is limited comparative data for the newer biologics available.
The COVID-19 pandemic turned attention to the risk of infections among biologic users, especially for respiratory tract infections, as they might relate to susceptibility for viral respiratory tract infections such as COVID-19.
In our study, the primary aim was to determine the risk of respiratory tract infections among real-world psoriasis patients treated with biologics, including the newer IL-17 and IL-23 inhibitors. The secondary aim was to assess risk of serious infections in this cohort. Additionally, rates of SARS-CoV-2 infections were assessed.





 Dr. Blumenthal[/caption]
Kimberly G. Blumenthal, MD, MSc
Massachusetts General Hospital
The Mongan Institute
Boston, MA 02114
[caption id="attachment_57828" align="alignleft" width="100"]
Dr. Blumenthal[/caption]
Kimberly G. Blumenthal, MD, MSc
Massachusetts General Hospital
The Mongan Institute
Boston, MA 02114
[caption id="attachment_57828" align="alignleft" width="100"] Dr. Krantz[/caption]
Matthew S. Krantz, MD
Division of Allergy, Pulmonary and Critical Care Medicine
Department of Medicine,
Vanderbilt University Medical Center,
Nashville, Tennessee
MedicalResearch.com: What is the background for this study?
Response: During the initial COVID-19 vaccine campaign with healthcare workers in December 2020, there was an unexpected higher than anticipated rate of immediate allergic reactions after Pfizer and Moderna mRNA vaccines. This prompted both patient and provider concerns, particularly in those with underlying allergic histories, on the associated risks for immediate allergic reactions with the mRNA vaccines.
Because of the significantly improved effectiveness of two doses of an mRNA vaccine compared to one dose, it was important to determine if those who experienced immediate allergic reaction symptoms after their first dose could go on to tolerate a second dose safely.
Dr. Krantz[/caption]
Matthew S. Krantz, MD
Division of Allergy, Pulmonary and Critical Care Medicine
Department of Medicine,
Vanderbilt University Medical Center,
Nashville, Tennessee
MedicalResearch.com: What is the background for this study?
Response: During the initial COVID-19 vaccine campaign with healthcare workers in December 2020, there was an unexpected higher than anticipated rate of immediate allergic reactions after Pfizer and Moderna mRNA vaccines. This prompted both patient and provider concerns, particularly in those with underlying allergic histories, on the associated risks for immediate allergic reactions with the mRNA vaccines.
Because of the significantly improved effectiveness of two doses of an mRNA vaccine compared to one dose, it was important to determine if those who experienced immediate allergic reaction symptoms after their first dose could go on to tolerate a second dose safely. 

 Dr. Els Broens[/caption]
Els M. Broens DVM, PhD, Dipl. ECVM, EBVS
European Veteirnary Specialist in Veterinary Microbiology
Associate Professor / Director VMDC
Department Biomolecular Health Sciences (Clinical Infectiology)
Faculty of Veterinary Medicine | Utrecht University
MedicalResearch.com: What is the background for this study?
Response: Several events have demonstrated that SARS-CoV-2 can infect animals, felines and mustelids in particular. In companion animals these are currently considered to be incidents with a negligible risk for public health since the main force of the pandemic is transmission between humans. However, it is urgent to understand the potential risk of animal infections for public health in the later stages of the pandemic when SARS-CoV-2 transmission between humans is greatly reduced and a virus reservoir in animals could become more important.
Incidental cases have shown that COVID-19 positive owners can transmit SARS-CoV-2 to their dog or cat. The close contact between owners and their dogs and cats and the interaction between dogs and cats from different households raises questions about the risk for pets to contract the disease and also about role of these animals in the transmission of SARS-CoV-2.
Dr. Els Broens[/caption]
Els M. Broens DVM, PhD, Dipl. ECVM, EBVS
European Veteirnary Specialist in Veterinary Microbiology
Associate Professor / Director VMDC
Department Biomolecular Health Sciences (Clinical Infectiology)
Faculty of Veterinary Medicine | Utrecht University
MedicalResearch.com: What is the background for this study?
Response: Several events have demonstrated that SARS-CoV-2 can infect animals, felines and mustelids in particular. In companion animals these are currently considered to be incidents with a negligible risk for public health since the main force of the pandemic is transmission between humans. However, it is urgent to understand the potential risk of animal infections for public health in the later stages of the pandemic when SARS-CoV-2 transmission between humans is greatly reduced and a virus reservoir in animals could become more important.
Incidental cases have shown that COVID-19 positive owners can transmit SARS-CoV-2 to their dog or cat. The close contact between owners and their dogs and cats and the interaction between dogs and cats from different households raises questions about the risk for pets to contract the disease and also about role of these animals in the transmission of SARS-CoV-2.


