Cancer Research / 01.03.2021
Liver Metastases Treatment
 One of the main dangers of cancer is metastasizing. This process can affect any organ in the human body. The most frequent causes of liver metastases are tumors of the gastrointestinal tract, mammary glands, lungs, and pancreas.
One of the modern methods of liver metastases treatment today is chemoembolization procedure. Its use allows doctors to fight cancer liver metastases with minimal harm to the patient. Statistics shows that this method is 30% more effective than traditional treatment of metastases with systemic chemotherapy.
Symptoms
As a rule, secondary liver cancer has no symptoms for a long time. This makes it difficult to diagnose this type of oncology. However, with regular medical check-ups, you can avoid this. To know when you should see a doctor, you need to know the symptoms of liver metastases that are most commonly seen:
One of the main dangers of cancer is metastasizing. This process can affect any organ in the human body. The most frequent causes of liver metastases are tumors of the gastrointestinal tract, mammary glands, lungs, and pancreas.
One of the modern methods of liver metastases treatment today is chemoembolization procedure. Its use allows doctors to fight cancer liver metastases with minimal harm to the patient. Statistics shows that this method is 30% more effective than traditional treatment of metastases with systemic chemotherapy.
Symptoms
As a rule, secondary liver cancer has no symptoms for a long time. This makes it difficult to diagnose this type of oncology. However, with regular medical check-ups, you can avoid this. To know when you should see a doctor, you need to know the symptoms of liver metastases that are most commonly seen:
- Abdominal bloating
- Nausea and vomiting
- Constipation or diarrhea
- Decreased appetite
- Severe weight loss
- Persistent low-grade increase in body temperature
- General weakness and fatigue


 Catharina Svanborg M.D., Ph.D.
Professor at Lund University Department of Laboratory Medicine,
Division of Microbiology, Immunology and Glycobiology
Founder/Chairman of the Board at HAMLET Pharma
MedicalResearch.com: What is the background for this study?
Like many unexpected scientific developments, this finding was serendipitous. In our search for the molecular basis of host susceptibility to infection, we discovered that infection directly affects MYC levels.
Gene expression analysis revealed that MYC itself was inhibited and that genes regulated by MYC were affected in children with acute kidney infection. Rapid reductions in MYC levels was further confirmed by infecting human kidney cells with the pathogenic E. coli bacteria isolated from patients with acute pyelonephritis, allowing us to formulate the hypothesis that bacteria regulate host MYC levels during acute infection and to investigate the mechanism leading to this inhibition. This work was conducted by the
Catharina Svanborg M.D., Ph.D.
Professor at Lund University Department of Laboratory Medicine,
Division of Microbiology, Immunology and Glycobiology
Founder/Chairman of the Board at HAMLET Pharma
MedicalResearch.com: What is the background for this study?
Like many unexpected scientific developments, this finding was serendipitous. In our search for the molecular basis of host susceptibility to infection, we discovered that infection directly affects MYC levels.
Gene expression analysis revealed that MYC itself was inhibited and that genes regulated by MYC were affected in children with acute kidney infection. Rapid reductions in MYC levels was further confirmed by infecting human kidney cells with the pathogenic E. coli bacteria isolated from patients with acute pyelonephritis, allowing us to formulate the hypothesis that bacteria regulate host MYC levels during acute infection and to investigate the mechanism leading to this inhibition. This work was conducted by the 




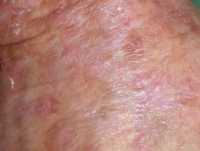
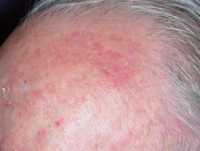 One example of actinic keratoses on scalp
One example of actinic keratoses on scalp













 Response: LENVIMA (lenvatinib), discovered and developed by Eisai, is an orally available multiple receptor tyrosine kinase inhibitor that inhibits the kinase activities of vascular endothelial growth factor (VEGF) receptors VEGFR1 (FLT1), VEGFR2 (KDR), and VEGFR3 (FLT4). LENVIMA inhibits other kinases that have been implicated in pathogenic angiogenesis, tumor growth, and cancer progression in addition to their normal cellular functions, including fibroblast growth factor (FGF) receptors FGFR1-4, the platelet derived growth factor receptor alpha (PDGFRα), KIT, and RET.
LENVIMA is approved in combination with everolimus for the treatment of patients with advanced renal cell carcinoma (RCC) following one prior anti-angiogenic therapy. The approved starting dose for LENVIMA is 18 mg daily. The objective of Study 218, a randomized, open-label, Phase 2 trial, was to assess whether the lower starting dose of LENVIMA (14 mg daily) in combination with everolimus (5 mg daily) would provide similar efficacy with an improved safety profile compared to the FDA-approved starting dose of LENVIMA (18 mg daily) plus everolimus (5 mg daily) in patients with advanced renal cell carcinoma (RCC) following prior treatment with an antiangiogenic therapy.
In the US, LENVIMA is also indicated for:
Response: LENVIMA (lenvatinib), discovered and developed by Eisai, is an orally available multiple receptor tyrosine kinase inhibitor that inhibits the kinase activities of vascular endothelial growth factor (VEGF) receptors VEGFR1 (FLT1), VEGFR2 (KDR), and VEGFR3 (FLT4). LENVIMA inhibits other kinases that have been implicated in pathogenic angiogenesis, tumor growth, and cancer progression in addition to their normal cellular functions, including fibroblast growth factor (FGF) receptors FGFR1-4, the platelet derived growth factor receptor alpha (PDGFRα), KIT, and RET.
LENVIMA is approved in combination with everolimus for the treatment of patients with advanced renal cell carcinoma (RCC) following one prior anti-angiogenic therapy. The approved starting dose for LENVIMA is 18 mg daily. The objective of Study 218, a randomized, open-label, Phase 2 trial, was to assess whether the lower starting dose of LENVIMA (14 mg daily) in combination with everolimus (5 mg daily) would provide similar efficacy with an improved safety profile compared to the FDA-approved starting dose of LENVIMA (18 mg daily) plus everolimus (5 mg daily) in patients with advanced renal cell carcinoma (RCC) following prior treatment with an antiangiogenic therapy.
In the US, LENVIMA is also indicated for: