Author Interviews, Prostate Cancer / 05.04.2023
Prostate Cancer: 15 Year Follow Up Compares Treatment with Surgery, Radiation and Active Surveillance
MedicalResearch.com Interview with:
[caption id="attachment_60281" align="alignleft" width="125"] Prof. Hamdy[/caption]
Freddie C. Hamdy FRCS, FMedSci
Nuffield Professor of Surgery, University of Oxford
[caption id="attachment_60283" align="alignleft" width="100"]
Prof. Hamdy[/caption]
Freddie C. Hamdy FRCS, FMedSci
Nuffield Professor of Surgery, University of Oxford
[caption id="attachment_60283" align="alignleft" width="100"] Prof. Donovan[/caption]
Jenny L. Donovan PhD, FMedSci
Professor of Social Medicine, University of Bristol
MedicalResearch.com: What is the background for this study?
Response: Prostate cancer is a common malignancy in men. Prostate cancer diagnosis is made largely through opportunistic screening with a PSA (Prostate Specific Antigen) blood test, followed by prostate biopsies. The ProtecT study, funded by the National Institute for Health and Care Research in the UK, is the largest randomised trial of treatment in screen-detected localised prostate cancer. The study began by testing 82,429 men between the ages of 50 and 69 years, across nine UK centres with a PSA blood test, followed by biopsies of the prostate if the PSA level was elevated. 2,664 men with clinically localised prostate cancer were found. From these, 1,643 (62%) agreed to be randomised to Surgery (radical prostatectomy to remove the prostate gland), Radiotherapy (external beam with a period of hormone treatment beforehand), or Active Monitoring (where men received regular checks and further investigations, with change to radical treatment as necessary). The men were carefully followed up for an average of 15 years. In parallel, the side-effects of treatments and quality of life of these men was investigated using patient-reported outcomes included in an annual study questionnaire completed for at least 12 years.
Prof. Donovan[/caption]
Jenny L. Donovan PhD, FMedSci
Professor of Social Medicine, University of Bristol
MedicalResearch.com: What is the background for this study?
Response: Prostate cancer is a common malignancy in men. Prostate cancer diagnosis is made largely through opportunistic screening with a PSA (Prostate Specific Antigen) blood test, followed by prostate biopsies. The ProtecT study, funded by the National Institute for Health and Care Research in the UK, is the largest randomised trial of treatment in screen-detected localised prostate cancer. The study began by testing 82,429 men between the ages of 50 and 69 years, across nine UK centres with a PSA blood test, followed by biopsies of the prostate if the PSA level was elevated. 2,664 men with clinically localised prostate cancer were found. From these, 1,643 (62%) agreed to be randomised to Surgery (radical prostatectomy to remove the prostate gland), Radiotherapy (external beam with a period of hormone treatment beforehand), or Active Monitoring (where men received regular checks and further investigations, with change to radical treatment as necessary). The men were carefully followed up for an average of 15 years. In parallel, the side-effects of treatments and quality of life of these men was investigated using patient-reported outcomes included in an annual study questionnaire completed for at least 12 years.
 Prof. Hamdy[/caption]
Freddie C. Hamdy FRCS, FMedSci
Nuffield Professor of Surgery, University of Oxford
[caption id="attachment_60283" align="alignleft" width="100"]
Prof. Hamdy[/caption]
Freddie C. Hamdy FRCS, FMedSci
Nuffield Professor of Surgery, University of Oxford
[caption id="attachment_60283" align="alignleft" width="100"] Prof. Donovan[/caption]
Jenny L. Donovan PhD, FMedSci
Professor of Social Medicine, University of Bristol
MedicalResearch.com: What is the background for this study?
Response: Prostate cancer is a common malignancy in men. Prostate cancer diagnosis is made largely through opportunistic screening with a PSA (Prostate Specific Antigen) blood test, followed by prostate biopsies. The ProtecT study, funded by the National Institute for Health and Care Research in the UK, is the largest randomised trial of treatment in screen-detected localised prostate cancer. The study began by testing 82,429 men between the ages of 50 and 69 years, across nine UK centres with a PSA blood test, followed by biopsies of the prostate if the PSA level was elevated. 2,664 men with clinically localised prostate cancer were found. From these, 1,643 (62%) agreed to be randomised to Surgery (radical prostatectomy to remove the prostate gland), Radiotherapy (external beam with a period of hormone treatment beforehand), or Active Monitoring (where men received regular checks and further investigations, with change to radical treatment as necessary). The men were carefully followed up for an average of 15 years. In parallel, the side-effects of treatments and quality of life of these men was investigated using patient-reported outcomes included in an annual study questionnaire completed for at least 12 years.
Prof. Donovan[/caption]
Jenny L. Donovan PhD, FMedSci
Professor of Social Medicine, University of Bristol
MedicalResearch.com: What is the background for this study?
Response: Prostate cancer is a common malignancy in men. Prostate cancer diagnosis is made largely through opportunistic screening with a PSA (Prostate Specific Antigen) blood test, followed by prostate biopsies. The ProtecT study, funded by the National Institute for Health and Care Research in the UK, is the largest randomised trial of treatment in screen-detected localised prostate cancer. The study began by testing 82,429 men between the ages of 50 and 69 years, across nine UK centres with a PSA blood test, followed by biopsies of the prostate if the PSA level was elevated. 2,664 men with clinically localised prostate cancer were found. From these, 1,643 (62%) agreed to be randomised to Surgery (radical prostatectomy to remove the prostate gland), Radiotherapy (external beam with a period of hormone treatment beforehand), or Active Monitoring (where men received regular checks and further investigations, with change to radical treatment as necessary). The men were carefully followed up for an average of 15 years. In parallel, the side-effects of treatments and quality of life of these men was investigated using patient-reported outcomes included in an annual study questionnaire completed for at least 12 years.


 Dr. Levin[/caption]
Trevor Levin Ph.D.
Founder and CEO of Convergent Genomics that produces the Uroamp assay
San Francisco, CA
MedicalResearch.com: What is the background for this study?
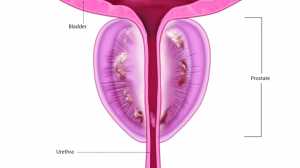
Response: Bladder cancer is one of the most expensive and challenging to diagnose and treat. Therefore, identifying cost-effective urine bladder cancer biomarkers to complement or replace the gold-standard invasive and costly cystoscopy for the early detection and monitoring of this highly recurrent disease is crucial. At the international Agency for research on Cancer (IARC-WHO), we have developed a simple urine-based assay TERT promoter mutations, the most common mutations in bladder cancer, and showed that the urine biomarker could detect bladder cancer patients at diagnosis but many years prior to clinical diagnosis. However, in this study, we wanted to see whether a more comprehensive genomic profiling of urine samples collected years prior to clinical diagnosis of bladder cancer could identify even more patients before they develop any symptoms.
The study was based on the UroAmp test, a general urine test that identifies mutations in 60 genes, developed by the Oregon Health Science University spin out company, Convergent Genomics. Drawing on previous research to identify genetic mutations linked to bladder cancer, the research team narrowed the new test down to focus on mutations within just ten genes.
Working with colleagues from the Tehran University of Medical Sciences in Iran, they trialled the potential new test using samples from the Golestan Cohort Study, which has tracked the health of more than 50,000 participants over ten years, all of whom provided urine samples at recruitment. Forty people within the study developed bladder cancer during that decade, and the team were able to test urine samples from twenty-nine of them, along with samples from 98 other similar participants as controls.
Dr. Levin[/caption]
Trevor Levin Ph.D.
Founder and CEO of Convergent Genomics that produces the Uroamp assay
San Francisco, CA
MedicalResearch.com: What is the background for this study?
Response: Bladder cancer is one of the most expensive and challenging to diagnose and treat. Therefore, identifying cost-effective urine bladder cancer biomarkers to complement or replace the gold-standard invasive and costly cystoscopy for the early detection and monitoring of this highly recurrent disease is crucial. At the international Agency for research on Cancer (IARC-WHO), we have developed a simple urine-based assay TERT promoter mutations, the most common mutations in bladder cancer, and showed that the urine biomarker could detect bladder cancer patients at diagnosis but many years prior to clinical diagnosis. However, in this study, we wanted to see whether a more comprehensive genomic profiling of urine samples collected years prior to clinical diagnosis of bladder cancer could identify even more patients before they develop any symptoms.
The study was based on the UroAmp test, a general urine test that identifies mutations in 60 genes, developed by the Oregon Health Science University spin out company, Convergent Genomics. Drawing on previous research to identify genetic mutations linked to bladder cancer, the research team narrowed the new test down to focus on mutations within just ten genes.
Working with colleagues from the Tehran University of Medical Sciences in Iran, they trialled the potential new test using samples from the Golestan Cohort Study, which has tracked the health of more than 50,000 participants over ten years, all of whom provided urine samples at recruitment. Forty people within the study developed bladder cancer during that decade, and the team were able to test urine samples from twenty-nine of them, along with samples from 98 other similar participants as controls.
 Dr. Koh[/caption]
Andrew Y. Koh, M.D.
Associate Professor, Pediatrics and Microbiology
Dr. Koh[/caption]
Andrew Y. Koh, M.D.
Associate Professor, Pediatrics and Microbiology

 Dr. Mosley[/caption]
Jonathan Mosley, MD, PhD
Associate Professor
Division of Clinical Pharmacology
Departments of Internal Medicine and Biomedical Informatics
Vanderbilt University Medical Center
MedicalResearch.com: What is the background for this study?
Response: Prostate cancer is an important source of morbidity and mortality among men. Earlier detection of disease is essential to reduce these adverse outcomes. Prostate cancer is heritable, and many single nucleotide polymorphisms (SNPs) associated with disease risk have been identified. Thus, there is considerable interest in using tools such as polygenic risk scores, which measure the burden of genetic risk variants an individual carries, to identify men at elevated risk of disease.
Dr. Mosley[/caption]
Jonathan Mosley, MD, PhD
Associate Professor
Division of Clinical Pharmacology
Departments of Internal Medicine and Biomedical Informatics
Vanderbilt University Medical Center
MedicalResearch.com: What is the background for this study?
Response: Prostate cancer is an important source of morbidity and mortality among men. Earlier detection of disease is essential to reduce these adverse outcomes. Prostate cancer is heritable, and many single nucleotide polymorphisms (SNPs) associated with disease risk have been identified. Thus, there is considerable interest in using tools such as polygenic risk scores, which measure the burden of genetic risk variants an individual carries, to identify men at elevated risk of disease.

 Dr. Tsirigos[/caption]
Aristotelis Tsirigos, Ph.D.
Professor of Medicine and Pathology
Co-director, Precision Medicine
Director, Applied Bioinformatics Laboratories
Dr. Tsirigos[/caption]
Aristotelis Tsirigos, Ph.D.
Professor of Medicine and Pathology
Co-director, Precision Medicine
Director, Applied Bioinformatics Laboratories



 Dr. Sally Lau[/caption]
Dr. Sally Lau MD
Medical oncologist, NYU Langone’s Perlmutter Cancer Center
Assistant professor of medicine
NYU Grossman School of Medicine
MedicalResearch.com: How big is the problem of
Dr. Sally Lau[/caption]
Dr. Sally Lau MD
Medical oncologist, NYU Langone’s Perlmutter Cancer Center
Assistant professor of medicine
NYU Grossman School of Medicine
MedicalResearch.com: How big is the problem of 

 Dr. White[/caption]
Alexandra J. White, PhD, MSPH
Stadtman Investigator
Epidemiology Branch
National Institute of Environmental Health Sciences
National Institutes of Health
Research Triangle Park, NC
MedicalResearch.com: What is the background for this study? What is the chemical primarily used in hair straighteners?
Response: Hair products such as dye and chemical straighteners contain several different chemicals that may act as carcinogens or endocrine disruptors and thus may be important for cancer risk. Straighteners in particular have been found to include chemicals such as phthalates, parabens, cyclosiloxanes and metals and may release formaldehyde when heated. Previous research has suggested that hair dye and chemical straighteners are related to other hormone-sensitive cancers such as breast and ovarian cancer, but no previous study has considered how they are related to uterine cancer risk.
Dr. White[/caption]
Alexandra J. White, PhD, MSPH
Stadtman Investigator
Epidemiology Branch
National Institute of Environmental Health Sciences
National Institutes of Health
Research Triangle Park, NC
MedicalResearch.com: What is the background for this study? What is the chemical primarily used in hair straighteners?
Response: Hair products such as dye and chemical straighteners contain several different chemicals that may act as carcinogens or endocrine disruptors and thus may be important for cancer risk. Straighteners in particular have been found to include chemicals such as phthalates, parabens, cyclosiloxanes and metals and may release formaldehyde when heated. Previous research has suggested that hair dye and chemical straighteners are related to other hormone-sensitive cancers such as breast and ovarian cancer, but no previous study has considered how they are related to uterine cancer risk.







 Dr. Barry[/caption]
Michael J. Barry, M.D
Director of the Informed Medical Decisions Program
Health Decision Sciences Center
Massachusetts General Hospital.
Professor of medicine at Harvard Medical School
Dr. Barry was appointed as Vice Chair of USPSTF in March 2021.
He previously served as a member from January 2017 through December 2020.
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: The Task Force looked at the use of vitamin and mineral supplementation specifically for the prevention of heart disease, stroke, and cancer. We found that there is not enough evidence to recommend for or against taking multivitamin supplements, nor the use of single or paired nutrient supplements, to prevent these conditions.
However, we do know that you should not take vitamin E or beta-carotene for this purpose.
Dr. Barry[/caption]
Michael J. Barry, M.D
Director of the Informed Medical Decisions Program
Health Decision Sciences Center
Massachusetts General Hospital.
Professor of medicine at Harvard Medical School
Dr. Barry was appointed as Vice Chair of USPSTF in March 2021.
He previously served as a member from January 2017 through December 2020.
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: The Task Force looked at the use of vitamin and mineral supplementation specifically for the prevention of heart disease, stroke, and cancer. We found that there is not enough evidence to recommend for or against taking multivitamin supplements, nor the use of single or paired nutrient supplements, to prevent these conditions.
However, we do know that you should not take vitamin E or beta-carotene for this purpose.






