AACR, Author Interviews, Biomarkers, Breast Cancer, Cancer Research / 11.04.2024
AACR24: MUC4 is a Biomarker of Metastasis in Triple Negative Breast Cancer and Can Help Improve Treatment Results
[caption id="attachment_61560" align="alignleft" width="125"] Dr. Tesi[/caption]
MedicalResearch.com Interview with:
RJ Tesi M.D.
CEO and Founder of INmune Bio
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Tesi[/caption]
MedicalResearch.com Interview with:
RJ Tesi M.D.
CEO and Founder of INmune Bio
MedicalResearch.com: What is the background for this study? What are the main findings?
 Dr. Tesi[/caption]
MedicalResearch.com Interview with:
RJ Tesi M.D.
CEO and Founder of INmune Bio
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Tesi[/caption]
MedicalResearch.com Interview with:
RJ Tesi M.D.
CEO and Founder of INmune Bio
MedicalResearch.com: What is the background for this study? What are the main findings?
- MUC4 expression by high-risk breast cancer (HER2+ or TNBC) is a biomarker that predicts resistance to therapy and an increased risk a metastasis. MUC4 expression can be determined at time of biopsy and therapeutic decisions should be adjusted to optimize the chance of response to first line therapy.
- Soluble TNF causes the up regulation of immune checkpoint proteins of cells of the TME. This includes CD47 and SIRPa on tumor based macrophages and CTLA4, PD1, LAG3 and TIGIT on T cells in the TME. INB03 is a pan immune checkpoint modulator. Treatment with INB03 downregulates all immune checkpoint proteins on the cells. Downmodulation of all immune checkpoint proteins improves response to immunotherapy.
- In TNBC, MUC4 expression predicts both resistance to anti-PD1 therapy and increased risk of distant metastasis. Treatment with INB03 decreases expression of proteins associated with tumor metastasis, decreases the number of metastasis and improves response to anti-PD1 therapy. Early use of INB03 may prevent distal disease and improve tumor control.









 According to the AACR Cancer Progress Report, cancer survivors have significantly improved from 50 years ago. It constituted only 1.4 percent of the US population earlier, but they have increased considerably. The number of cancer survivors is estimated to grow to
According to the AACR Cancer Progress Report, cancer survivors have significantly improved from 50 years ago. It constituted only 1.4 percent of the US population earlier, but they have increased considerably. The number of cancer survivors is estimated to grow to 

 Prof. Rong Xu[/caption]
Rong Xu, PhD
Prof. Rong Xu[/caption]
Rong Xu, PhD

 Lisa-Marie Smale, PharmD
Lisa-Marie Smale, PharmD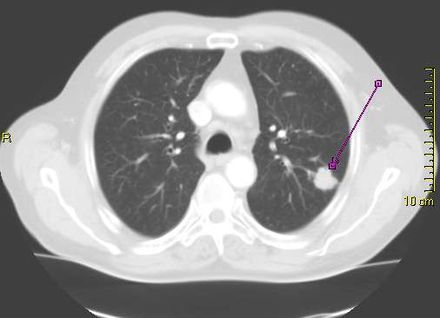
 Dr. Han[/caption]
Summer S Han, PhD
Associate Professor
Dr. Han[/caption]
Summer S Han, PhD
Associate Professor
 Dr. Choi[/caption]
Dr. Eunji Choi PhD
Instructor, Neurosurgery
Department: Adult Neurosurgery
Stanford University School of Medicine
MedicalResearch.com: What is the background for this study?
Dr. Choi[/caption]
Dr. Eunji Choi PhD
Instructor, Neurosurgery
Department: Adult Neurosurgery
Stanford University School of Medicine
MedicalResearch.com: What is the background for this study?


 Dr. Stark[/caption]
Dr Mitchell Stark
Dr. Stark[/caption]
Dr Mitchell Stark
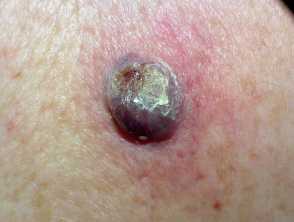
 One example of a nodular melanoma without pigment.
One example of a nodular melanoma without pigment. 




 Dr. Thomas[/caption]
Dr. Daniel Thomas MD PhD FRACP FRCPA
Program Leader, Blood Cancer
Precision Medicine Theme at the South Australia Health Medical Research Institute
Clinical Hematologist, Royal Adelaide Hospital
Associate Professor, Adelaide Medical School, The University of Adelaide
MedicalResearch.com: What is the background for this study? Would you briefly describe the condition of CMML?
Response: Chronic myelomonocytic leukemia (CMML) is a rare, but increasingly frequent, clonal stem cell disorder that results in hyperproliferation of inflammatory monocytes, a form of white blood cells. It features both myelodysplasia and myeloproliferation. CMML is most often found in older adults and leads to anemia, decreased quality of life, and an increased risk of acute myeloid leukemia (AML).
Granulocyte-macrophage colony-stimulating factor (GM-CSF) is a cytokine that stimulates production, growth, differentiation, activation, and function of myeloid cells (monocytes, neutrophils, and eosinophils). In the presence of RAS-pathway mutations, a greater sensitivity to GM-CSF contributes to the hyperproliferation of myelocytes in myelodysplastic leukemias such as CMML, juvenile myelomonocytic leukemia (JMML), and acute myeloid leukemia (AML). In CMML, greater sensitivity to GM-CSF stimulates excessive monocytic precursor proliferation.
The PREACH-M Trial, which stands for PREcision Approach to CHronic Myelomonocytic Leukemia, assesses the efficacy of lenzilumab in addition to azacitidine in treatment-naïve CMML participants with RAS-pathway mutations (KRAS, NRAS, CBL) and separately high dose ascorbate in participants with TET2 mutations who do not have RAS-pathway mutations. The study is currently underway and actively enrolling. It is being conducted and funded by the South Australian Health and Medical Research Institute (SAHMRI).
Dr. Thomas[/caption]
Dr. Daniel Thomas MD PhD FRACP FRCPA
Program Leader, Blood Cancer
Precision Medicine Theme at the South Australia Health Medical Research Institute
Clinical Hematologist, Royal Adelaide Hospital
Associate Professor, Adelaide Medical School, The University of Adelaide
MedicalResearch.com: What is the background for this study? Would you briefly describe the condition of CMML?
Response: Chronic myelomonocytic leukemia (CMML) is a rare, but increasingly frequent, clonal stem cell disorder that results in hyperproliferation of inflammatory monocytes, a form of white blood cells. It features both myelodysplasia and myeloproliferation. CMML is most often found in older adults and leads to anemia, decreased quality of life, and an increased risk of acute myeloid leukemia (AML).
Granulocyte-macrophage colony-stimulating factor (GM-CSF) is a cytokine that stimulates production, growth, differentiation, activation, and function of myeloid cells (monocytes, neutrophils, and eosinophils). In the presence of RAS-pathway mutations, a greater sensitivity to GM-CSF contributes to the hyperproliferation of myelocytes in myelodysplastic leukemias such as CMML, juvenile myelomonocytic leukemia (JMML), and acute myeloid leukemia (AML). In CMML, greater sensitivity to GM-CSF stimulates excessive monocytic precursor proliferation.
The PREACH-M Trial, which stands for PREcision Approach to CHronic Myelomonocytic Leukemia, assesses the efficacy of lenzilumab in addition to azacitidine in treatment-naïve CMML participants with RAS-pathway mutations (KRAS, NRAS, CBL) and separately high dose ascorbate in participants with TET2 mutations who do not have RAS-pathway mutations. The study is currently underway and actively enrolling. It is being conducted and funded by the South Australian Health and Medical Research Institute (SAHMRI). 
 Dr. Kamath[/caption]
Dr. Suneel Kamath MD
Gastrointestinal Oncologist
Cleveland Clinic
Senior Author on this research
MedicalResearch.com: What is the background for this study?
Response: Colorectal cancer rates in young people under age 50 are skyrocketing and have been for the last 3-4 decades. We really don’t understand why because most cases (probably around 70%) are not genetic or hereditary, just random, unfortunate events. We suspect that it is some exposure(s) like excess consumption of red meat, processed foods, sugar-sweetened beverages, excess antibiotic use altering the microbiome, rising incidence of obesity or some other factors. We really don’t know why yet.
Our study used a technology called metabolomics, the study of breakdown products and production building blocks for our bodies, to look for differences in colorectal cancer in young people versus people that are older that developed colorectal cancer. Because metabolomics measures how each individual interacts with the exposures in our environment like diet, air quality, etc., it is a way to bridge the gap between our nature (determined by genetics) and nurture (determined by our exposures).
Dr. Kamath[/caption]
Dr. Suneel Kamath MD
Gastrointestinal Oncologist
Cleveland Clinic
Senior Author on this research
MedicalResearch.com: What is the background for this study?
Response: Colorectal cancer rates in young people under age 50 are skyrocketing and have been for the last 3-4 decades. We really don’t understand why because most cases (probably around 70%) are not genetic or hereditary, just random, unfortunate events. We suspect that it is some exposure(s) like excess consumption of red meat, processed foods, sugar-sweetened beverages, excess antibiotic use altering the microbiome, rising incidence of obesity or some other factors. We really don’t know why yet.
Our study used a technology called metabolomics, the study of breakdown products and production building blocks for our bodies, to look for differences in colorectal cancer in young people versus people that are older that developed colorectal cancer. Because metabolomics measures how each individual interacts with the exposures in our environment like diet, air quality, etc., it is a way to bridge the gap between our nature (determined by genetics) and nurture (determined by our exposures).



 Dr. Lova Sun[/caption]
Lova L. Sun, MD, MSCE
Medical Oncology
Assistant Professor of Medicine
Hospital of the University of Pennsylvania
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: An common clinical question for patients with metastatic non-small cell lung cancer with long-term response to immunotherapy-based treatment is how long to continue treatment. The major clinical trials stopped immunotherapy at a maximum of 2 years, but in clinical practice many patients and clinicians continue treatment beyond this time point.
We conducted a retrospective study of lung cancer patients across the US with long-term response to immunotherapy, to compare survival between those who stopped treatment at 2 years vs those who continued beyond 2 years. We found that there was no statistically significant difference in survival between the two groups.
Dr. Lova Sun[/caption]
Lova L. Sun, MD, MSCE
Medical Oncology
Assistant Professor of Medicine
Hospital of the University of Pennsylvania
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: An common clinical question for patients with metastatic non-small cell lung cancer with long-term response to immunotherapy-based treatment is how long to continue treatment. The major clinical trials stopped immunotherapy at a maximum of 2 years, but in clinical practice many patients and clinicians continue treatment beyond this time point.
We conducted a retrospective study of lung cancer patients across the US with long-term response to immunotherapy, to compare survival between those who stopped treatment at 2 years vs those who continued beyond 2 years. We found that there was no statistically significant difference in survival between the two groups.



 Dr. Ruiz[/caption]
John M. Ruiz, Ph.D
Associate Professor of Clinical Psychology
Department of Psychology
University of Arizona
Dr. Ruiz is the incoming editor-in-chief of the American Psychological Association (APA) journal, Health Psychology
Dr. Ruiz joined the U.S. Preventive Services Task Force in January 2022
MedicalResearch.com: What is the background for this study? What are the main findings?
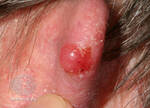
Response: Skin cancer is the most common type of cancer in the United States, but it often does not cause serious complications or death. The Task Force’s recommendation on screening for skin cancer focuses on the effectiveness of visual skin exams for children and adults who do not have any symptoms. When reviewing the latest research, we found that there is currently not enough evidence to tell us whether or not screening people without signs or symptoms is beneficial. This is an I statement.
Dr. Ruiz[/caption]
John M. Ruiz, Ph.D
Associate Professor of Clinical Psychology
Department of Psychology
University of Arizona
Dr. Ruiz is the incoming editor-in-chief of the American Psychological Association (APA) journal, Health Psychology
Dr. Ruiz joined the U.S. Preventive Services Task Force in January 2022
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Skin cancer is the most common type of cancer in the United States, but it often does not cause serious complications or death. The Task Force’s recommendation on screening for skin cancer focuses on the effectiveness of visual skin exams for children and adults who do not have any symptoms. When reviewing the latest research, we found that there is currently not enough evidence to tell us whether or not screening people without signs or symptoms is beneficial. This is an I statement.
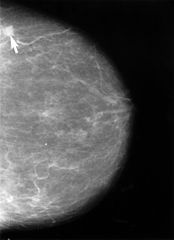
 Dr. Fallah[/caption]
Mahdi Fallah, MD, PhD
Dr. Fallah[/caption]
Mahdi Fallah, MD, PhD

