Author Interviews, BMJ, Breast Cancer, Cancer Research, Radiation Therapy / 10.11.2020
Single Dose Radiotherapy (TARGIT-IORT) As Good As Conventional Radiotherapy For Most Women With Early Breast Cancer
MedicalResearch.com Interview with:
Professor Jayant S Vaidya
MBBS MS DNB FRCS PhD
Professor of Surgery and Oncology
University College London
MedicalResearch.com: What is the background for this study? What type of single dose radiation is used?
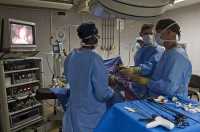
[caption id="attachment_55895" align="alignleft" width="500"] Prof Jeffrey S Tobias, Prof Jayant S Vaidya, Prof Max Bulsara. and Prof Michael Baum[/caption]
Response: The findings of the large international randomised trial (TARGIT-A trial), published in the British Medical Journal (BMJ 2020;370:m2836), confirm the long-term effectiveness of Targeted Intraoperative Radiotherapy (TARGIT-IORT): a breast cancer treatment which is increasingly available throughout the world. For most women with early breast cancer, a single dose of targeted radiotherapy during surgery is just as effective as conventional radiotherapy, which requires several visits to hospital after surgery.
Conventional external beam radiotherapy (EBRT) is delivered from outside the body via a radiotherapy machine (linear accelerator), and consists of a daily treatment session (known as fractions) to the whole breast, over a period between three to six weeks. Each of these treatments is given over a few minutes, but requires 15 to 30 hospital visits, which could be a significant distance from where the patient lives.
TARGIT-IORT is delivered immediately after lumpectomy (tumour removal), via a small ball-shaped device placed inside the breast, directly where the cancer had been. The single-dose treatment lasts for around 20 to 30 minutes and replaces the need for extra hospital visits, benefiting both patient safety and well-being. The device used is called INTRABEAM.
More details are described on the BMJ and UCL webpages:
https://www.bmj.com/company/newsroom/single-dose-radiotherapy-as-good-as-conventional-radiotherapy-for-most-women-with-early-breast-cancer/
https://www.ucl.ac.uk/news/2020/aug/single-dose-radiotherapy-effective-treating-breast-cancer
https://blogs.bmj.com/bmj/2020/08/20/targeted-intraoperative-radiotherapy-for-early-breast-cancer-new-evidence/
Prof Jeffrey S Tobias, Prof Jayant S Vaidya, Prof Max Bulsara. and Prof Michael Baum[/caption]
Response: The findings of the large international randomised trial (TARGIT-A trial), published in the British Medical Journal (BMJ 2020;370:m2836), confirm the long-term effectiveness of Targeted Intraoperative Radiotherapy (TARGIT-IORT): a breast cancer treatment which is increasingly available throughout the world. For most women with early breast cancer, a single dose of targeted radiotherapy during surgery is just as effective as conventional radiotherapy, which requires several visits to hospital after surgery.
Conventional external beam radiotherapy (EBRT) is delivered from outside the body via a radiotherapy machine (linear accelerator), and consists of a daily treatment session (known as fractions) to the whole breast, over a period between three to six weeks. Each of these treatments is given over a few minutes, but requires 15 to 30 hospital visits, which could be a significant distance from where the patient lives.
TARGIT-IORT is delivered immediately after lumpectomy (tumour removal), via a small ball-shaped device placed inside the breast, directly where the cancer had been. The single-dose treatment lasts for around 20 to 30 minutes and replaces the need for extra hospital visits, benefiting both patient safety and well-being. The device used is called INTRABEAM.
More details are described on the BMJ and UCL webpages:
https://www.bmj.com/company/newsroom/single-dose-radiotherapy-as-good-as-conventional-radiotherapy-for-most-women-with-early-breast-cancer/
https://www.ucl.ac.uk/news/2020/aug/single-dose-radiotherapy-effective-treating-breast-cancer
https://blogs.bmj.com/bmj/2020/08/20/targeted-intraoperative-radiotherapy-for-early-breast-cancer-new-evidence/
 Prof Jeffrey S Tobias, Prof Jayant S Vaidya, Prof Max Bulsara. and Prof Michael Baum[/caption]
Response: The findings of the large international randomised trial (TARGIT-A trial), published in the British Medical Journal (BMJ 2020;370:m2836), confirm the long-term effectiveness of Targeted Intraoperative Radiotherapy (TARGIT-IORT): a breast cancer treatment which is increasingly available throughout the world. For most women with early breast cancer, a single dose of targeted radiotherapy during surgery is just as effective as conventional radiotherapy, which requires several visits to hospital after surgery.
Conventional external beam radiotherapy (EBRT) is delivered from outside the body via a radiotherapy machine (linear accelerator), and consists of a daily treatment session (known as fractions) to the whole breast, over a period between three to six weeks. Each of these treatments is given over a few minutes, but requires 15 to 30 hospital visits, which could be a significant distance from where the patient lives.
TARGIT-IORT is delivered immediately after lumpectomy (tumour removal), via a small ball-shaped device placed inside the breast, directly where the cancer had been. The single-dose treatment lasts for around 20 to 30 minutes and replaces the need for extra hospital visits, benefiting both patient safety and well-being. The device used is called INTRABEAM.
More details are described on the BMJ and UCL webpages:
https://www.bmj.com/company/newsroom/single-dose-radiotherapy-as-good-as-conventional-radiotherapy-for-most-women-with-early-breast-cancer/
https://www.ucl.ac.uk/news/2020/aug/single-dose-radiotherapy-effective-treating-breast-cancer
https://blogs.bmj.com/bmj/2020/08/20/targeted-intraoperative-radiotherapy-for-early-breast-cancer-new-evidence/
Prof Jeffrey S Tobias, Prof Jayant S Vaidya, Prof Max Bulsara. and Prof Michael Baum[/caption]
Response: The findings of the large international randomised trial (TARGIT-A trial), published in the British Medical Journal (BMJ 2020;370:m2836), confirm the long-term effectiveness of Targeted Intraoperative Radiotherapy (TARGIT-IORT): a breast cancer treatment which is increasingly available throughout the world. For most women with early breast cancer, a single dose of targeted radiotherapy during surgery is just as effective as conventional radiotherapy, which requires several visits to hospital after surgery.
Conventional external beam radiotherapy (EBRT) is delivered from outside the body via a radiotherapy machine (linear accelerator), and consists of a daily treatment session (known as fractions) to the whole breast, over a period between three to six weeks. Each of these treatments is given over a few minutes, but requires 15 to 30 hospital visits, which could be a significant distance from where the patient lives.
TARGIT-IORT is delivered immediately after lumpectomy (tumour removal), via a small ball-shaped device placed inside the breast, directly where the cancer had been. The single-dose treatment lasts for around 20 to 30 minutes and replaces the need for extra hospital visits, benefiting both patient safety and well-being. The device used is called INTRABEAM.
More details are described on the BMJ and UCL webpages:
https://www.bmj.com/company/newsroom/single-dose-radiotherapy-as-good-as-conventional-radiotherapy-for-most-women-with-early-breast-cancer/
https://www.ucl.ac.uk/news/2020/aug/single-dose-radiotherapy-effective-treating-breast-cancer
https://blogs.bmj.com/bmj/2020/08/20/targeted-intraoperative-radiotherapy-for-early-breast-cancer-new-evidence/



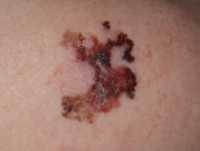
![MedicalResearch.com Interview with: Selin Tokez, PhD Student Department of Dermatology Erasmus MC, Rotterdam MedicalResearch.com: What is the background for this study? Response: Cutaneous squamous cell carcinoma (cSCC) is the second most common skin cancer worldwide with still increasing incidence rates. Given these high incidence rates together with the associated health costs and possibility of fatal progression, it is extremely important to have accurate and complete data on the epidemiology of this disease. Nevertheless, national cancer registries in many countries do not routinely record cSCC cases and therefore currently known numbers are mainly based on incomplete data sources. Additionally, if cSCC cases are registered, this usually only concerns the first cSCC per patient while we know that, contrary to many other malignant neoplasms, patients may develop numerous cSCCs over time. MedicalResearch.com: What are the main findings? Response: In the present study, we analyzed Dutch nationwide data comprising about 145,000 patients with a first invasive cSCC diagnosis between the years 1989 and 2017. We found that the incidence rates of a first cSCC per patient almost tripled in male patients and increased about fivefold in female patients in this 30-year time period. Also, we had data on all cSCCs per patient for the year 2017 and could therefore compare this with the data on only the first cSCC per patient: incidence rates increased by 58% for men and 35% for women when multiple cSCCs were considered. In absolute numbers, this resulted in an increase of 45% in cSCC diagnoses in 2017. Lastly, we extended our analyses by predicting future cSCC incidence rates up to 2027. Given that no substantially effective measures are undertaken in the near future, current cSCC incidence rates will increase with 23% in males and 29% in females in the next decade. MedicalResearch.com: What should readers take away from your report? Response: We could summarize our main message in two points: while people generally know that cSCC is a very common disease with increasing incidence rates, it is not taken into account that these numbers are often based on incomplete data registries and that the real numbers are even higher. In this paper, we provided these numbers for a period of 30 years based on highly accurate data from the Netherlands Cancer Registry. On top of that, as a second main finding, we showed that the real burden caused by cSCC is approximately 50% higher (taken males and females together) when all cSCC diagnoses in 1 year are registered instead of only the first tumor per patient. Together with our prediction analyses that showed an on average 26% further increase for the coming decade, this will have enormous implications for the dermato-oncological health care planning and cost management. Our results urgently call for revision of skin cancer health policies to be able to cope with this rising burden of cSCC management. Ultimately, primary prevention will remain the key strategy to halt the increasing trend in cSCC incidence and the occurrence of multiple cSCCs per patient, which we hope to further stimulate with our paper as well. MedicalResearch.com: What recommendations do you have for future research as a result of this work? Response: As we had data on multiple cSCCs for only one year, we would suggest to perform trend analyses for multiple cSCCs per patient as well when a longer follow-up duration has been reached. Furthermore, it would be very relevant to identify high-risk cSCCs or patients at risk for multiple cSCCs in order to be able to establish efficient follow-up regimens and take these high risk groups into account when revising skin cancer health policies. MedicalResearch.com: Is there anything else you would like to add? Response: The authors of this study have no conflicts of interest that are relevant to this article. Citation: Tokez S, Hollestein L, Louwman M, Nijsten T, Wakkee M. Incidence of Multiple vs First Cutaneous Squamous Cell Carcinoma on a Nationwide Scale and Estimation of Future Incidences of Cutaneous Squamous Cell Carcinoma. JAMA Dermatol. Published online October 28, 2020. doi:10.1001/jamadermatol.2020.3677 [subscribe] Last Modified: [last-modified] The information on MedicalResearch.com is provided for educational purposes only, and is in no way intended to diagnose, cure, or treat any medical or other condition. Always seek the advice of your physician or other qualified health and ask your doctor any questions you may have regarding a medical condition. In addition to all other limitations and disclaimers in this agreement, service provider and its third party providers disclaim any liability or loss in connection with the content provided on this website.](https://medicalresearch.com/wp-content/uploads/2020/10/squamous-cell-cancer-dermnetnz-image.jpg)




 Dr. Bandi[/caption]
Priti Bandi PhD
Principal Scientist, Risk Factors Surveillance Research
American Cancer Society, Inc.
Atlanta, GA 30303
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Trends in e-cigarette prevalence and population count of users according to cigarette smoking histories are unknown. These data are needed to inform public health actions against a rapidly changing U.S. e-cigarette market.
Dr. Bandi[/caption]
Priti Bandi PhD
Principal Scientist, Risk Factors Surveillance Research
American Cancer Society, Inc.
Atlanta, GA 30303
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Trends in e-cigarette prevalence and population count of users according to cigarette smoking histories are unknown. These data are needed to inform public health actions against a rapidly changing U.S. e-cigarette market.





 Dr. Lara-Corrales[/caption]
Irene Lara-Corrales, MD
Associate Professor of Pediatrics at the University of Toronto
Staff physician in Pediatric Dermatology at the
Hospital for Sick Children in Toronto, Canada
She is a member of the
Dr. Lara-Corrales[/caption]
Irene Lara-Corrales, MD
Associate Professor of Pediatrics at the University of Toronto
Staff physician in Pediatric Dermatology at the
Hospital for Sick Children in Toronto, Canada
She is a member of the 
 Dr. Huang[/caption]
Jennifer Huang, MD
Dr. Huang is a pediatric dermatologist at Boston Children’s Hospital and Dana-Farber Cancer Institute.
She is an Associate Professor of Dermatology at Harvard Medical School.
Dr. Huang is a member of the
Dr. Huang[/caption]
Jennifer Huang, MD
Dr. Huang is a pediatric dermatologist at Boston Children’s Hospital and Dana-Farber Cancer Institute.
She is an Associate Professor of Dermatology at Harvard Medical School.
Dr. Huang is a member of the 




 Dr. Gerstung[/caption]
Moritz Gerstung PhD
Group Leader: Computational cancer biology
EMBL-European Bioinformatics Institute
MedicalResearch.com: What is the background for this study?
Response: We have learned a lot in the last ten years about the molecular nature about various cancers thanks to the resources created by TCGA, ICGC and many other initiatives. Similarly, digital pathology has progressed hugely due to new AI algorithms. Yet it hasn’t been explored deeply how a cancer’s genetic makeup and its histopathological appearance are related. Here computers can be very helpful as they can process large amounts of digital microscopy slide images and test whether there are any recurrent histopathological patterns in relation to hundreds or thousands of genetic and other molecular abnormalities.
Dr. Gerstung[/caption]
Moritz Gerstung PhD
Group Leader: Computational cancer biology
EMBL-European Bioinformatics Institute
MedicalResearch.com: What is the background for this study?
Response: We have learned a lot in the last ten years about the molecular nature about various cancers thanks to the resources created by TCGA, ICGC and many other initiatives. Similarly, digital pathology has progressed hugely due to new AI algorithms. Yet it hasn’t been explored deeply how a cancer’s genetic makeup and its histopathological appearance are related. Here computers can be very helpful as they can process large amounts of digital microscopy slide images and test whether there are any recurrent histopathological patterns in relation to hundreds or thousands of genetic and other molecular abnormalities. 




 Dr. Lang[/caption]
Dr. Kathryn Lang
VP, Outcomes and Evidence
Guardant Health
MedicalResearch.com: What is the background for this study?
Response: Despite a wide variety of screening methods available and increasing public awareness of the value of early detection, colorectal cancer (CRC) remains a leading cause of cancer-related deaths. However nearly 1 in 3 adults in the United States is not compliant with screening recommendations, with most citing that current screening methods are time consuming, unpleasant (stool-based testing), and in the case of colonoscopy, invasive. A blood-based CRC screening test could improve compliance rates by providing physicians with an opportunistic, in-office screening modality. However, demonstrating the clinical utility of blood-based cell-free circulating tumor DNA (ctDNA) fractions for the detection of cancer in asymptomatic individuals has thus far been challenged by the failure to achieve clinically meaningful sensitivity and specificity thresholds due to significantly lower tumor cell-free free DNA fractions and the increasing relevance of biological confounders.
The multi-modal approach of Guardant Health’s LUNAR-2 assay (genomics, methylation and fragmentomics) coupled with advanced bioinformatic analysis and a focused approach of honing in on the unique signals of CRC has been shown in previously reported cohorts to perform with sensitivity and specificity which satisfies the needs of clinicians in screening for CRC.
Dr. Lang[/caption]
Dr. Kathryn Lang
VP, Outcomes and Evidence
Guardant Health
MedicalResearch.com: What is the background for this study?
Response: Despite a wide variety of screening methods available and increasing public awareness of the value of early detection, colorectal cancer (CRC) remains a leading cause of cancer-related deaths. However nearly 1 in 3 adults in the United States is not compliant with screening recommendations, with most citing that current screening methods are time consuming, unpleasant (stool-based testing), and in the case of colonoscopy, invasive. A blood-based CRC screening test could improve compliance rates by providing physicians with an opportunistic, in-office screening modality. However, demonstrating the clinical utility of blood-based cell-free circulating tumor DNA (ctDNA) fractions for the detection of cancer in asymptomatic individuals has thus far been challenged by the failure to achieve clinically meaningful sensitivity and specificity thresholds due to significantly lower tumor cell-free free DNA fractions and the increasing relevance of biological confounders.
The multi-modal approach of Guardant Health’s LUNAR-2 assay (genomics, methylation and fragmentomics) coupled with advanced bioinformatic analysis and a focused approach of honing in on the unique signals of CRC has been shown in previously reported cohorts to perform with sensitivity and specificity which satisfies the needs of clinicians in screening for CRC.