Author Interviews, Cancer Research, Dermatology, NEJM / 20.07.2021
KLISYRI® (tirbanibulin): Shorter Topical Treatment for Common Precancerous Actinic Keratoses
MedicalResearch.com Interview with:
Jane Fang, MD
Clinical
Athenex, Inc.
MedicalResearch.com: What is the background for this study?
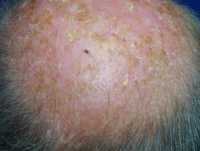
Response: Tirbanibulin is a first-in-class synthetic molecule that has potent anti-proliferative activity by inhibiting tubulin polymerization and disrupting src kinase signaling. It has been formulated as an ointment for the treatment of actinic keratosis, a very common precancerous condition of UV-damaged skin that affects over 50 million people in the US.
The most commonly adopted management approach is to remove AK lesions as it is hard to predict which lesion will become cancerous. Lesion-directed treatment like cryotherapy can effectively remove lesions one at a time but does not treat larger field of cancerization. Also, it is limited by associated pain and long term complication such as scarring. Currently approved topical treatments involve cumbersome application courses of weeks or months, and induce considerable local skin reactions that were not well tolerated by patients. The Phase 3 studies demonstrated that a short 5-day once daily course of tirbanibulin ointment 1% is an efficacious and safe topical treatment of actinic keratosis.






 Prof Jeffrey S Tobias, Prof Jayant S Vaidya, Prof Max Bulsara and Prof Michael BaumMedicalResearch.com Interview with:[/caption]
Professor Jayant S Vaidya
MBBS MS DNB FRCS PhD
Professor of Surgery and Oncology
University College London
MedicalResearch.com: What is the background for this study? What type of single dose radiation is used?
Response: The new paper published in the British Journal of Cancer (
Prof Jeffrey S Tobias, Prof Jayant S Vaidya, Prof Max Bulsara and Prof Michael BaumMedicalResearch.com Interview with:[/caption]
Professor Jayant S Vaidya
MBBS MS DNB FRCS PhD
Professor of Surgery and Oncology
University College London
MedicalResearch.com: What is the background for this study? What type of single dose radiation is used?
Response: The new paper published in the British Journal of Cancer (
 Professor Marie-Aleth Richard
EADV Communications Committee Chair
Professor, University Hospital of La Timone
Marseille, France
MedicalResearch.com: What is the background for this Roundtable event? Would you describe the mission of the European Commission’s Beating Cancer Plan?
Response: Europe’s Beating Cancer Plan is the first, comprehensive EU strategy on cancer, aimed at tackling the disease through all key stages: prevention; early detection; diagnosis and treatment; and quality of life of cancer patients and survivors. The Plan also aims to create opportunities to improve cancer care through research and innovative projects, such as artificial intelligence, and to promote equal access to knowledge and treatments in cancer care across Europe.
The EADV seeks to create a bridge between the EU health policy agenda and scientific research, by engaging with policymakers, patient organisations and other stakeholders to support a patient centric-approach; tackling melanoma and non-melanoma skin cancers (NMSC) at all stages of the pathway, from prevention to follow-up care.
Through our Roundtable event, the EADV brought together these key stakeholders to evaluate the effectiveness of the EBCP on preventing both melanoma and NMSC, as well as identify joint recommendations that step-up measures towards this goal.
Professor Marie-Aleth Richard
EADV Communications Committee Chair
Professor, University Hospital of La Timone
Marseille, France
MedicalResearch.com: What is the background for this Roundtable event? Would you describe the mission of the European Commission’s Beating Cancer Plan?
Response: Europe’s Beating Cancer Plan is the first, comprehensive EU strategy on cancer, aimed at tackling the disease through all key stages: prevention; early detection; diagnosis and treatment; and quality of life of cancer patients and survivors. The Plan also aims to create opportunities to improve cancer care through research and innovative projects, such as artificial intelligence, and to promote equal access to knowledge and treatments in cancer care across Europe.
The EADV seeks to create a bridge between the EU health policy agenda and scientific research, by engaging with policymakers, patient organisations and other stakeholders to support a patient centric-approach; tackling melanoma and non-melanoma skin cancers (NMSC) at all stages of the pathway, from prevention to follow-up care.
Through our Roundtable event, the EADV brought together these key stakeholders to evaluate the effectiveness of the EBCP on preventing both melanoma and NMSC, as well as identify joint recommendations that step-up measures towards this goal.




 Dr. Jinhee Hur[/caption]
Jinhee Hur, PhD
Research Fellow
Department of Nutrition
Harvard T.H. Chan School of Public Health
Boston, MA 02115
MedicalResearch.com: What is the background for this study?
Response: Early-onset colorectal cancer (EO-CRC, age <50 years at diagnosis) is rapidly rising in the US since the mid-1980s, with an unclear understanding of its etiology and contributors to the rise. Sugar-sweetened beverages (SSBs) exert adverse metabolic repercussions throughout the life course, including insulin resistance and inflammation. Higher SSB intake can induce obesity, which has been linked to risk of EO-CRC. A recent experimental study also suggests that high fructose corn syrup, a primary sweetener in SSBs, may promote colon tumor growth, independent of metabolic dysregulation. In the US, SSB consumption has dramatically increased during the 2nd half of the 20th century, and adolescents and young adults have been the heaviest SSB drinkers across all age groups. Thus, we expect SSBs may be an emerging risk factor for EO-CRC and likely contribute to the rising incidence of EO-CRC.
Dr. Jinhee Hur[/caption]
Jinhee Hur, PhD
Research Fellow
Department of Nutrition
Harvard T.H. Chan School of Public Health
Boston, MA 02115
MedicalResearch.com: What is the background for this study?
Response: Early-onset colorectal cancer (EO-CRC, age <50 years at diagnosis) is rapidly rising in the US since the mid-1980s, with an unclear understanding of its etiology and contributors to the rise. Sugar-sweetened beverages (SSBs) exert adverse metabolic repercussions throughout the life course, including insulin resistance and inflammation. Higher SSB intake can induce obesity, which has been linked to risk of EO-CRC. A recent experimental study also suggests that high fructose corn syrup, a primary sweetener in SSBs, may promote colon tumor growth, independent of metabolic dysregulation. In the US, SSB consumption has dramatically increased during the 2nd half of the 20th century, and adolescents and young adults have been the heaviest SSB drinkers across all age groups. Thus, we expect SSBs may be an emerging risk factor for EO-CRC and likely contribute to the rising incidence of EO-CRC.

 Dr. Navlakha[/caption]
Saket Navlakha PhD
Simons Center for Quantitative Biology
Cold Spring Harbor Laboratory
Cold Spring Harbor, NY
MedicalResearch.com: What is the background for this algorithm? How does it aide in patient care?
Response: The machine learning algorithm helps to predict if and when a patient will develop severe COVID symptoms, based on information on how the patient presents on the day of infection. This could lead to improved patient outcomes, by getting a “heads up” on what may happen in the near future.
Dr. Navlakha[/caption]
Saket Navlakha PhD
Simons Center for Quantitative Biology
Cold Spring Harbor Laboratory
Cold Spring Harbor, NY
MedicalResearch.com: What is the background for this algorithm? How does it aide in patient care?
Response: The machine learning algorithm helps to predict if and when a patient will develop severe COVID symptoms, based on information on how the patient presents on the day of infection. This could lead to improved patient outcomes, by getting a “heads up” on what may happen in the near future.



 Dr. Ulhas Nair[/caption]
Nishanth Ulhas Nair, Ph.D.
Affiliation: Staff Scientist at Cancer Data Science Laboratory, Center for Cancer Research
National Cancer Institute (NCI), National Institutes of Health (NIH)
Bethesda, Maryland, USA. Date: April 22, 2021
Dr. Raffit Hassan and Dr. Eytan Ruppin at the National Cancer Institute (NCI) are the senior authors of this study.
MedicalResearch.com: What is the background for this study?
Response: Malignant mesothelioma is an aggressive cancer with limited treatment options and poor prognosis. An in-depth knowledge of genetic, transcriptomic and immunogenic events involved in mesothelioma is critical for successful development of prognostics and therapeutic modalities. In this study we aim to address this by exploring a new large scale patient tumor dataset of 122 mesothelioma patients, called NCI mesothelioma patient data, along with their genomic, transcriptomic, and phenotypic information. Unlike previous large-scale studies which have been focused on malignant pleural mesothelioma patients, our dataset contains an approximately equal representation of malignant pleural and peritoneal mesothelioma patients which allows to identify any differences between them.
Dr. Ulhas Nair[/caption]
Nishanth Ulhas Nair, Ph.D.
Affiliation: Staff Scientist at Cancer Data Science Laboratory, Center for Cancer Research
National Cancer Institute (NCI), National Institutes of Health (NIH)
Bethesda, Maryland, USA. Date: April 22, 2021
Dr. Raffit Hassan and Dr. Eytan Ruppin at the National Cancer Institute (NCI) are the senior authors of this study.
MedicalResearch.com: What is the background for this study?
Response: Malignant mesothelioma is an aggressive cancer with limited treatment options and poor prognosis. An in-depth knowledge of genetic, transcriptomic and immunogenic events involved in mesothelioma is critical for successful development of prognostics and therapeutic modalities. In this study we aim to address this by exploring a new large scale patient tumor dataset of 122 mesothelioma patients, called NCI mesothelioma patient data, along with their genomic, transcriptomic, and phenotypic information. Unlike previous large-scale studies which have been focused on malignant pleural mesothelioma patients, our dataset contains an approximately equal representation of malignant pleural and peritoneal mesothelioma patients which allows to identify any differences between them.






 Anne Cust[/caption]
Anne Cust | PhD, Professor of Cancer Epidemiology
The University of Sydney
Faculty of Medicine and Health
Sydney School of Public Health
MedicalResearch.com: What is the background for this study? Are the screeners specially trained, use full body photographs, dermoscopy etc?
Response: The Melanoma High-risk Clinic Study was developed to optimise the early detection of new melanomas in people at high risk of developing melanoma. A previous single-centre study observed fewer excisions and healthcare costs, thinner melanomas and better quality of life when surveillance of high risk patients was conducted in a melanoma dermatology clinic with a structured surveillance protocol involving 6-monthly full body examinations aided by total body photography (TBP) and sequential digital dermoscopy imaging (SDDI). The initial pilot study was performed in a single tertiary referral specialist centre using trained dermatologists who routinely used the diagnostic interventions.
Our objective was to examine longer-term sustainability and expansion of the program to multiple practices including a primary care skin cancer clinic setting. The hypothesis was that the outcomes would be similar if using the same protocol and diagnostic tools. The participating doctors were trained to follow the protocol, which included instruction on how to respond and interpret changing lesions, but not in use of dermoscopy or skin examinations, which were routinely and consistently used in all clinics prior to the study commencing. There were 593 participants assessed as very high risk of melanoma who participated in the Melanoma High-risk Clinic Study from 2012-2018. Nearly all of the participants had had a previous melanoma and had additional melanoma risk factors. 57% were male and the median age at study entry was 58 years.
Anne Cust[/caption]
Anne Cust | PhD, Professor of Cancer Epidemiology
The University of Sydney
Faculty of Medicine and Health
Sydney School of Public Health
MedicalResearch.com: What is the background for this study? Are the screeners specially trained, use full body photographs, dermoscopy etc?
Response: The Melanoma High-risk Clinic Study was developed to optimise the early detection of new melanomas in people at high risk of developing melanoma. A previous single-centre study observed fewer excisions and healthcare costs, thinner melanomas and better quality of life when surveillance of high risk patients was conducted in a melanoma dermatology clinic with a structured surveillance protocol involving 6-monthly full body examinations aided by total body photography (TBP) and sequential digital dermoscopy imaging (SDDI). The initial pilot study was performed in a single tertiary referral specialist centre using trained dermatologists who routinely used the diagnostic interventions.
Our objective was to examine longer-term sustainability and expansion of the program to multiple practices including a primary care skin cancer clinic setting. The hypothesis was that the outcomes would be similar if using the same protocol and diagnostic tools. The participating doctors were trained to follow the protocol, which included instruction on how to respond and interpret changing lesions, but not in use of dermoscopy or skin examinations, which were routinely and consistently used in all clinics prior to the study commencing. There were 593 participants assessed as very high risk of melanoma who participated in the Melanoma High-risk Clinic Study from 2012-2018. Nearly all of the participants had had a previous melanoma and had additional melanoma risk factors. 57% were male and the median age at study entry was 58 years.


 Nadia Koyratty[/caption]
Nadia Koyratty PhD student
Department of Epidemiology and Environmental Health
University at Buffalo
State University of New York
MedicalResearch.com: What is the background for this study?
Response: The literature suggests that sugars contribute to the incidence of breast cancer, but few exists on the prognosis after a breast cancer diagnosis.
MedicalResearch.com: What are the main findings?
Response: Compared to breast cancer patients who never or rarely drank non-diet soda, those who reported drinking non-diet soda five times or more per week had a 62% higher likelihood of dying from any causes, and were 85% more likely to die from breast cancer specifically.
Nadia Koyratty[/caption]
Nadia Koyratty PhD student
Department of Epidemiology and Environmental Health
University at Buffalo
State University of New York
MedicalResearch.com: What is the background for this study?
Response: The literature suggests that sugars contribute to the incidence of breast cancer, but few exists on the prognosis after a breast cancer diagnosis.
MedicalResearch.com: What are the main findings?
Response: Compared to breast cancer patients who never or rarely drank non-diet soda, those who reported drinking non-diet soda five times or more per week had a 62% higher likelihood of dying from any causes, and were 85% more likely to die from breast cancer specifically.
