Author Interviews, MRI, Pain Research, Rheumatology / 11.10.2018
Fibromyalgia: Scan Reveals Increased Brain Activation in Pain, Emotion and Affect Areas
MedicalResearch.com Interview with:
Daniel S. Albrecht, Ph.D.
Research Fellow, Department of Radiology
Harvard Medical School
MedicalResearch.com: What is the background for this study? Can you briefly describe what is meant by fibromyalgia?
Response: Fibromyalgia (FM) is a poorly understood chronic condition characterized by widespread musculoskeletal pain, fatigue, unrefreshing sleep, memory deficits and attention difficulties, among other symptoms. FM affects an estimated 4 million adults in the U.S., but despite this prevalence, effective therapies for treating FM are lacking.
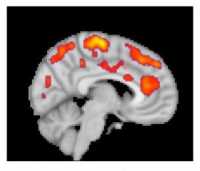
[caption id="attachment_45185" align="aligncenter" width="400"]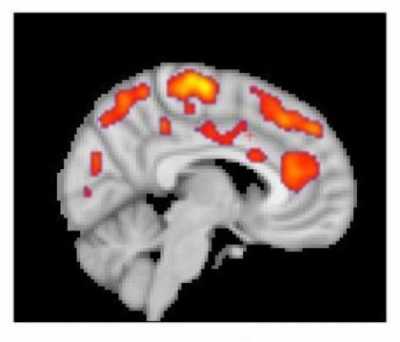 This combined MR/PET image highlights areas of the brain in which patients with fibromyalgia were found to have increased glial activation, compared with unaffected control volunteers.
This combined MR/PET image highlights areas of the brain in which patients with fibromyalgia were found to have increased glial activation, compared with unaffected control volunteers.
Credit: Marco Loggia, PhD, Martinos Center for Biomedical Imaging, Massachusetts General Hospital[/caption] Part of the reason for the paucity of effective therapeutics is insufficient knowledge of the underlying mechanisms contributing to FM. Previous work from co-senior author of the current manuscript, Eva Kosek, MD, PhD, and collaborators at the Karolinska Institute in Sweden found elevated inflammatory molecules in the cerebrospinal fluid of FM patients, which could be reflective of brain neuroinflammation in these patients. However, no study had directly assessed the presence of neuroinflammation in the brain of FM patients. Co-senior author of the study, Marco Loggia, PhD, and collaborators showed in a 2015 Brain publication that individuals with chronic low back pain (cLBP) exhibit evidence of brain neuroinflammation, specifically activation of glial cells. Our team utilized simultaneous MR/PET imaging to image brain levels of the 18 kDa translocator protein (TSPO), which is widely used as a marker of glial activation due to vast upregulation of TSPO in glial cells, e.g. microglia and astrocytes, in preclinical models of inflammation and neurological disease. Dr. Loggia sought to extend these finding in cLBP to FM, hypothesizing that activation of glial cells may also be associated with FM pathology. To this end, we used the same TSPO PET tracer to image 20 FM patients and 16 healthy controls. During a conference where I was presenting preliminary results of the fibromyalgia study, Dr. Loggia met with Dr. Kosek and discovered that, across the Atlantic, her group was performing a very similar study, imaging 11 FM patients and 11 controls with the same TSPO PET compound. They decided to form a collaboration, and logistic talks began to determine the best strategy to combine and analyze the separate datasets. In addition to PET imaging with the TSPO tracer, which is suggested to reflect activated microglia and astrocytes, Dr. Kosek’s group also collected PET scans using a tracer thought to bind specifically to astrocytes rather than microglia. This tracer was used in order to discern the relative contributions of microglia and astrocytes to any observed differences in TSPO PET signal.
 This combined MR/PET image highlights areas of the brain in which patients with fibromyalgia were found to have increased glial activation, compared with unaffected control volunteers.
This combined MR/PET image highlights areas of the brain in which patients with fibromyalgia were found to have increased glial activation, compared with unaffected control volunteers.Credit: Marco Loggia, PhD, Martinos Center for Biomedical Imaging, Massachusetts General Hospital[/caption] Part of the reason for the paucity of effective therapeutics is insufficient knowledge of the underlying mechanisms contributing to FM. Previous work from co-senior author of the current manuscript, Eva Kosek, MD, PhD, and collaborators at the Karolinska Institute in Sweden found elevated inflammatory molecules in the cerebrospinal fluid of FM patients, which could be reflective of brain neuroinflammation in these patients. However, no study had directly assessed the presence of neuroinflammation in the brain of FM patients. Co-senior author of the study, Marco Loggia, PhD, and collaborators showed in a 2015 Brain publication that individuals with chronic low back pain (cLBP) exhibit evidence of brain neuroinflammation, specifically activation of glial cells. Our team utilized simultaneous MR/PET imaging to image brain levels of the 18 kDa translocator protein (TSPO), which is widely used as a marker of glial activation due to vast upregulation of TSPO in glial cells, e.g. microglia and astrocytes, in preclinical models of inflammation and neurological disease. Dr. Loggia sought to extend these finding in cLBP to FM, hypothesizing that activation of glial cells may also be associated with FM pathology. To this end, we used the same TSPO PET tracer to image 20 FM patients and 16 healthy controls. During a conference where I was presenting preliminary results of the fibromyalgia study, Dr. Loggia met with Dr. Kosek and discovered that, across the Atlantic, her group was performing a very similar study, imaging 11 FM patients and 11 controls with the same TSPO PET compound. They decided to form a collaboration, and logistic talks began to determine the best strategy to combine and analyze the separate datasets. In addition to PET imaging with the TSPO tracer, which is suggested to reflect activated microglia and astrocytes, Dr. Kosek’s group also collected PET scans using a tracer thought to bind specifically to astrocytes rather than microglia. This tracer was used in order to discern the relative contributions of microglia and astrocytes to any observed differences in TSPO PET signal.

















 Dr. Ishida[/caption]
Dr. Julie H. Ishida MD
Department of Medicine, Division of Nephrology
University of California, San Francisco and
San Francisco Veterans Affairs Medical Center
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Gabapentin and pregabalin are used for the management of symptoms such as neuropathic pain, itching, and restless leg syndrome in patients receiving hemodialysis. However, hemodialysis patients may be particularly vulnerable to adverse events related to these agents, which are cleared by the kidney, but there is limited data evaluating their risk in this population.
Gabapentin and pregabalin use were associated with risk for altered mental status, fall, and fracture, and in some cases, even at doses that would be considered safe for use in this population.
Dr. Ishida[/caption]
Dr. Julie H. Ishida MD
Department of Medicine, Division of Nephrology
University of California, San Francisco and
San Francisco Veterans Affairs Medical Center
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Gabapentin and pregabalin are used for the management of symptoms such as neuropathic pain, itching, and restless leg syndrome in patients receiving hemodialysis. However, hemodialysis patients may be particularly vulnerable to adverse events related to these agents, which are cleared by the kidney, but there is limited data evaluating their risk in this population.
Gabapentin and pregabalin use were associated with risk for altered mental status, fall, and fracture, and in some cases, even at doses that would be considered safe for use in this population.