Author Interviews, Cannabis, JAMA, Pulmonary Disease / 22.04.2020
Fewer EVALI Cases in States with Recreational Marijuana
MedicalResearch.com Interview with:
[caption id="attachment_53964" align="alignleft" width="200"] Dr. Hollingsworth[/caption]
Alex Hollingsworth PhD
Assistant Professor
O'Neill School of Public and Environmental Affairs
Indiana University
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: I've been working with Coady Wing and Ashley Bradford on a few different studies of the effects of recreational marijuana laws on drug and alcohol use.
Soon after EVALI became a major issue, the prevailing theory from the CDC and others was that EVALI was caused by the use of vitamin E acetate in illegal THC vaping products.
Our group read about this and we thought about some of the things that often happen in black markets for illegal drugs. For instance, during the alcohol prohibition era, bootleg alcohol producers often made and sold alcohol products that were not that safe to drink. In more recent years, there are cases where black market sellers of illegal drugs like heroin try to increase profit margins by adding other substances, which can be harmful.
We thought that maybe something like that could be happening in EVALI. Perhaps people in states where recreational marijuana is legal tended to purchase marijuana products from the legal market and the legal market was not selling any marijuana vaping products that included vitamin E acetate.
Dr. Hollingsworth[/caption]
Alex Hollingsworth PhD
Assistant Professor
O'Neill School of Public and Environmental Affairs
Indiana University
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: I've been working with Coady Wing and Ashley Bradford on a few different studies of the effects of recreational marijuana laws on drug and alcohol use.
Soon after EVALI became a major issue, the prevailing theory from the CDC and others was that EVALI was caused by the use of vitamin E acetate in illegal THC vaping products.
Our group read about this and we thought about some of the things that often happen in black markets for illegal drugs. For instance, during the alcohol prohibition era, bootleg alcohol producers often made and sold alcohol products that were not that safe to drink. In more recent years, there are cases where black market sellers of illegal drugs like heroin try to increase profit margins by adding other substances, which can be harmful.
We thought that maybe something like that could be happening in EVALI. Perhaps people in states where recreational marijuana is legal tended to purchase marijuana products from the legal market and the legal market was not selling any marijuana vaping products that included vitamin E acetate.
 Dr. Hollingsworth[/caption]
Alex Hollingsworth PhD
Assistant Professor
O'Neill School of Public and Environmental Affairs
Indiana University
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: I've been working with Coady Wing and Ashley Bradford on a few different studies of the effects of recreational marijuana laws on drug and alcohol use.
Soon after EVALI became a major issue, the prevailing theory from the CDC and others was that EVALI was caused by the use of vitamin E acetate in illegal THC vaping products.
Our group read about this and we thought about some of the things that often happen in black markets for illegal drugs. For instance, during the alcohol prohibition era, bootleg alcohol producers often made and sold alcohol products that were not that safe to drink. In more recent years, there are cases where black market sellers of illegal drugs like heroin try to increase profit margins by adding other substances, which can be harmful.
We thought that maybe something like that could be happening in EVALI. Perhaps people in states where recreational marijuana is legal tended to purchase marijuana products from the legal market and the legal market was not selling any marijuana vaping products that included vitamin E acetate.
Dr. Hollingsworth[/caption]
Alex Hollingsworth PhD
Assistant Professor
O'Neill School of Public and Environmental Affairs
Indiana University
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: I've been working with Coady Wing and Ashley Bradford on a few different studies of the effects of recreational marijuana laws on drug and alcohol use.
Soon after EVALI became a major issue, the prevailing theory from the CDC and others was that EVALI was caused by the use of vitamin E acetate in illegal THC vaping products.
Our group read about this and we thought about some of the things that often happen in black markets for illegal drugs. For instance, during the alcohol prohibition era, bootleg alcohol producers often made and sold alcohol products that were not that safe to drink. In more recent years, there are cases where black market sellers of illegal drugs like heroin try to increase profit margins by adding other substances, which can be harmful.
We thought that maybe something like that could be happening in EVALI. Perhaps people in states where recreational marijuana is legal tended to purchase marijuana products from the legal market and the legal market was not selling any marijuana vaping products that included vitamin E acetate.

 Aurika Savickaite[/caption]
Aurika Savickaite RN
Adult Gerontology Acute Care Nurse Practitioner
Bulletproof Coach
University of Chicago Medicine
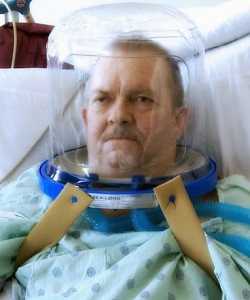
MedicalResearch.com: Would you briefly explain what is meant by helmet-based ventilation? How does it work?
Response: For patients in respiratory failure, noninvasive positive pressure ventilation (NIPPV) is usually delivered through a nasal mask or facemask. Many patients develop pain, discomfort – even claustrophobia -- from using NIPPV systems. The transparent helmet was developed to improve the tolerance of noninvasive ventilation. It allows the patient to see, read, speak and drink without interrupting noninvasive positive-pressure ventilation (NPPV).
The helmet has a sealed connection and a soft collar that adheres to the neck which helps prevent the air leaks that are very common with nasal- or face masks. High positive end-expiratory pressure (PEEP) is vital in treating patients in respiratory failure and thanks to helmets “none to minimum air leak” system, PEEP can be set high (up to 25). NIPPV via a nasal- or full-face mask typically begins to show air leaks when the required pressure exceeds 15-20cm H2O.
Aurika Savickaite[/caption]
Aurika Savickaite RN
Adult Gerontology Acute Care Nurse Practitioner
Bulletproof Coach
University of Chicago Medicine
MedicalResearch.com: Would you briefly explain what is meant by helmet-based ventilation? How does it work?
Response: For patients in respiratory failure, noninvasive positive pressure ventilation (NIPPV) is usually delivered through a nasal mask or facemask. Many patients develop pain, discomfort – even claustrophobia -- from using NIPPV systems. The transparent helmet was developed to improve the tolerance of noninvasive ventilation. It allows the patient to see, read, speak and drink without interrupting noninvasive positive-pressure ventilation (NPPV).
The helmet has a sealed connection and a soft collar that adheres to the neck which helps prevent the air leaks that are very common with nasal- or face masks. High positive end-expiratory pressure (PEEP) is vital in treating patients in respiratory failure and thanks to helmets “none to minimum air leak” system, PEEP can be set high (up to 25). NIPPV via a nasal- or full-face mask typically begins to show air leaks when the required pressure exceeds 15-20cm H2O.