Author Interviews, Infections, OBGYNE, Sexual Health, STD / 13.05.2020
Trichomoniasis: Results of Phase 3 Trial to Assess Efficacy and Safety of Single-Dose Solosec® (secnidazole) in Women
MedicalResearch.com Interview with:
[caption id="attachment_54202" align="alignleft" width="200"] Dr. Kaufman[/caption]
Gregory Kaufman, M.D.
Senior Vice President
Global Clinical and Medical Affairs
Specialty at Lupin
MedicalResearch.com: What is the background for this study? Would you briefly explain what is meant by trichomoniasis? How common is this infection?
Response: The Phase 3 trial evaluated the effectiveness and safety of a single oral dose of Solosec® (secnidazole) 2g oral granules for the treatment of trichomoniasis in adult women. Top-line results were positive and showed that Solosec was generally well-tolerated.
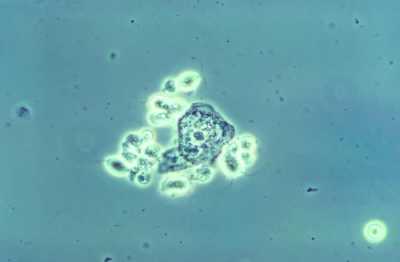
Trichomoniasis is the most common non-viral sexually transmitted infection (STI) in the U.S., and is caused by a protozoan parasite called Trichomonas vaginalis.[i] Trichomoniasis affects 3 to 5 million people in the U.S.,[ii] and is four- to five-times more prevalent in in women, compared to men.[iii].
Dr. Kaufman[/caption]
Gregory Kaufman, M.D.
Senior Vice President
Global Clinical and Medical Affairs
Specialty at Lupin
MedicalResearch.com: What is the background for this study? Would you briefly explain what is meant by trichomoniasis? How common is this infection?
Response: The Phase 3 trial evaluated the effectiveness and safety of a single oral dose of Solosec® (secnidazole) 2g oral granules for the treatment of trichomoniasis in adult women. Top-line results were positive and showed that Solosec was generally well-tolerated.
Trichomoniasis is the most common non-viral sexually transmitted infection (STI) in the U.S., and is caused by a protozoan parasite called Trichomonas vaginalis.[i] Trichomoniasis affects 3 to 5 million people in the U.S.,[ii] and is four- to five-times more prevalent in in women, compared to men.[iii].
 Dr. Kaufman[/caption]
Gregory Kaufman, M.D.
Senior Vice President
Global Clinical and Medical Affairs
Specialty at Lupin
MedicalResearch.com: What is the background for this study? Would you briefly explain what is meant by trichomoniasis? How common is this infection?
Response: The Phase 3 trial evaluated the effectiveness and safety of a single oral dose of Solosec® (secnidazole) 2g oral granules for the treatment of trichomoniasis in adult women. Top-line results were positive and showed that Solosec was generally well-tolerated.
Trichomoniasis is the most common non-viral sexually transmitted infection (STI) in the U.S., and is caused by a protozoan parasite called Trichomonas vaginalis.[i] Trichomoniasis affects 3 to 5 million people in the U.S.,[ii] and is four- to five-times more prevalent in in women, compared to men.[iii].
Dr. Kaufman[/caption]
Gregory Kaufman, M.D.
Senior Vice President
Global Clinical and Medical Affairs
Specialty at Lupin
MedicalResearch.com: What is the background for this study? Would you briefly explain what is meant by trichomoniasis? How common is this infection?
Response: The Phase 3 trial evaluated the effectiveness and safety of a single oral dose of Solosec® (secnidazole) 2g oral granules for the treatment of trichomoniasis in adult women. Top-line results were positive and showed that Solosec was generally well-tolerated.
Trichomoniasis is the most common non-viral sexually transmitted infection (STI) in the U.S., and is caused by a protozoan parasite called Trichomonas vaginalis.[i] Trichomoniasis affects 3 to 5 million people in the U.S.,[ii] and is four- to five-times more prevalent in in women, compared to men.[iii].