MedicalResearch.com Interview with:
[caption id="attachment_36326" align="alignleft" width="92"]

Dr. Huml[/caption]
Dr. Anne Huml MD
Center for Reducing Health Disparities
Case Western Reserve University
MetroHealth Medical Center
Cleveland, Ohio
MedicalResearch.com: What is the background for this study? What are the main findings?
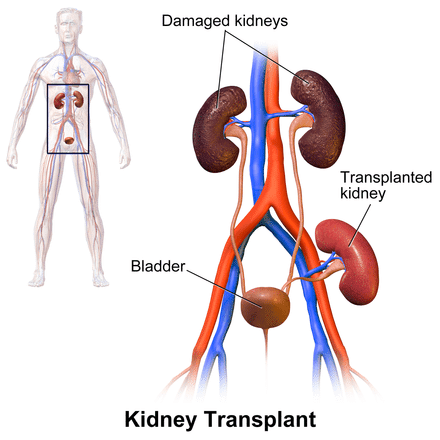
Response: Overall, about 600,000 Americans have end stage renal disease and require chronic dialysis treatment or a kidney transplant to survive. Compared to chronic dialysis, kidney transplantation results in better survival and quality of life and lower health care costs. Approximately 100,000 patients are listed for a kidney transplant. However, only 17,000 transplants occur per year with two-thirds of these coming from deceased donor organs. Annually, over 8,000 patients either die waiting for a kidney transplant or are removed from the waiting list for being too ill. Waiting times vary based on geography, but it is not unusual for patients to wait upwards of 5 years for a kidney transplant. There are sizeable race, gender, and socioeconomic disparities in access to kidney transplantation
.
In this study, we evaluated the outcomes of deceased donor kidney offers and their association with donor and waitlisted patient characteristics. Differences in kidney offer outcomes to patients at the top of the waiting list may contribute to disparities in transplantation.
When a deceased donor organ becomes available, a match run list is created that ranks potential recipients in priority order based upon several characteristics, including waiting time and immunologic criteria. At the discretion of the transplant center, organ offers to patients on their waiting list can be accepted for transplant, or refused for a particular patient. The offers continue down the match run list in sequential order. For each potential recipient in whom the organ is not transplanted, a refusal code is generated and catalogued with the United Network of Organ Sharing, or UNOS. UNOS identifies 37 unique refusal codes and categorizes them into donor-related, transplant center bypassed for pre-specified criteria, recipient-related, histocompatibility-related, program-related, or other reasons for refusal.


 Jim Gleason, National President of Transplant Recipients International Organization (TRIO), discusses what organ transplant patients are experiencing during the pandemic, and some tips they ought to consider to help ease some of the burden.
Mr. Gleason also discusses his role in the development of the recently released AlloCare app is in the AppStore --
Jim Gleason, National President of Transplant Recipients International Organization (TRIO), discusses what organ transplant patients are experiencing during the pandemic, and some tips they ought to consider to help ease some of the burden.
Mr. Gleason also discusses his role in the development of the recently released AlloCare app is in the AppStore -- 






 Rebecca Ivy Hartman, M.D
Instructor in Dermatology
Brigham and Women's Hospital
Boston MA 02115
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Organ transplant recipients (OTR) are at 100-fold higher risk to develop certain skin cancers compared to the general population due to immunosuppression, and thus preventing skin cancer in this population is critical.
Our study found that in a high-risk Australian OTR population, only half of patients practiced multiple measures of sun protection regularly.
However, after participating in a research study that required dermatology visits, patients were over 4-times more likely to report using multiple measures of sun protection regularly. Patients were more likely to have a positive behavioral change if they did not already undergo annual skin cancer screening prior to study participation.
Rebecca Ivy Hartman, M.D
Instructor in Dermatology
Brigham and Women's Hospital
Boston MA 02115
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Organ transplant recipients (OTR) are at 100-fold higher risk to develop certain skin cancers compared to the general population due to immunosuppression, and thus preventing skin cancer in this population is critical.
Our study found that in a high-risk Australian OTR population, only half of patients practiced multiple measures of sun protection regularly.
However, after participating in a research study that required dermatology visits, patients were over 4-times more likely to report using multiple measures of sun protection regularly. Patients were more likely to have a positive behavioral change if they did not already undergo annual skin cancer screening prior to study participation.












 Dr. Joseph Alvarnas[/caption]
Joseph Alvarnas, MD
Associate clinical professor
Department of hematology and Director of value-based analytics
City of Hope National Medical Center
Duarte, CA
MedicalResearch.com: What is the background for this study?
Dr. Alvarnas: Patients with HIV infection have a significantly increased risk of non-Hodgkin lymphoma and Hodgkin lymphoma. Prior to the availability of effective anti-retroviral therapy, HIV-infected patients with lymphoma had very poor treatment outcomes. Following the availability of effective anti-HIV therapy, patient outcomes for HIV-infected patients now parallel those of non-infected patients. Historically, however, HIV infection has been used as a criterion for not offering patients autologous blood stem cell transplantation outside of centers with unique expertise. The purpose of this trial was to evaluate outcomes, complication rates, and immunological reconstitution of HIV-infected patients following autologous blood stem cell transplantation.
Dr. Joseph Alvarnas[/caption]
Joseph Alvarnas, MD
Associate clinical professor
Department of hematology and Director of value-based analytics
City of Hope National Medical Center
Duarte, CA
MedicalResearch.com: What is the background for this study?
Dr. Alvarnas: Patients with HIV infection have a significantly increased risk of non-Hodgkin lymphoma and Hodgkin lymphoma. Prior to the availability of effective anti-retroviral therapy, HIV-infected patients with lymphoma had very poor treatment outcomes. Following the availability of effective anti-HIV therapy, patient outcomes for HIV-infected patients now parallel those of non-infected patients. Historically, however, HIV infection has been used as a criterion for not offering patients autologous blood stem cell transplantation outside of centers with unique expertise. The purpose of this trial was to evaluate outcomes, complication rates, and immunological reconstitution of HIV-infected patients following autologous blood stem cell transplantation.

