Author Interviews, Biomarkers, Heart Disease, NEJM / 03.03.2022
NEJM Study Evaluates Levels of hs Troponin after Cardiac Surgery and Risk of Death
MedicalResearch.com Interview with:
[caption id="attachment_58828" align="alignleft" width="150"] Dr. PJ.Devereaux[/caption]
PJ Devereaux MD PhD
Professor of Medicine, and
of Health Research Methods, Evidence and Impact
McMaster University
President of the Society of Perioperative Research and Care
MedicalResearch.com: What is the background for this study?
Response: More than 1 million patients undergo cardiac surgery in the United States and Europe annually. Although cardiac surgery has the potential to improve and prolong a patient’s quality and duration of life, it is associated with complications. Prognostically important heart injury – detected by an elevated blood concentration of either cardiac troponin or creatine kinase myocardial MB isoform (CK-MB) – is one of the most common complications after cardiac surgery and is associated with increased mortality.
Although elevated CK-MB was historically used to define heart injury after cardiac surgery, this assay is no longer available in many hospitals worldwide, and consensus statements have recommended high-sensitivity cardiac troponin assays as the preferred biomarker. Based on expert opinion, the Fourth Universal Definition of Myocardial Infarction suggested that a cardiac troponin concentration >10 times the upper reference limit, in patients with a normal baseline measurement, should be the threshold used in the diagnosis of heart attack along with evidence of ischemia (e.g., ischemic ST changes on an ECG) in the first 48 hours after coronary artery bypass grafting (CABG). Although the Academic Research Consortium-2 Consensus stated there was no evidence-based threshold for cardiac troponin after CABG, they endorsed a threshold for the diagnosis of heart attack of ≥35 times the upper reference limit together with new evidence of ischemia, based on expert opinion. They also defined a threshold of ≥70 times the upper reference limit as a stand-alone criterion for clinically important periprocedural myocardial injury.
Globally, many hospitals now use high-sensitivity cardiac troponin assays; however, limited data are available to define a prognostically important degree of myocardial injury after cardiac surgery based on these assays. We undertook the Vascular Events in Surgery Patients Cohort Evaluation (VISION) Cardiac Surgery Study to examine clinical outcomes after cardiac surgery.
A primary objective was to determine the relationship between postoperative levels of high-sensitivity cardiac troponin I and the risk of death 30 days after cardiac surgery.
Dr. PJ.Devereaux[/caption]
PJ Devereaux MD PhD
Professor of Medicine, and
of Health Research Methods, Evidence and Impact
McMaster University
President of the Society of Perioperative Research and Care
MedicalResearch.com: What is the background for this study?
Response: More than 1 million patients undergo cardiac surgery in the United States and Europe annually. Although cardiac surgery has the potential to improve and prolong a patient’s quality and duration of life, it is associated with complications. Prognostically important heart injury – detected by an elevated blood concentration of either cardiac troponin or creatine kinase myocardial MB isoform (CK-MB) – is one of the most common complications after cardiac surgery and is associated with increased mortality.
Although elevated CK-MB was historically used to define heart injury after cardiac surgery, this assay is no longer available in many hospitals worldwide, and consensus statements have recommended high-sensitivity cardiac troponin assays as the preferred biomarker. Based on expert opinion, the Fourth Universal Definition of Myocardial Infarction suggested that a cardiac troponin concentration >10 times the upper reference limit, in patients with a normal baseline measurement, should be the threshold used in the diagnosis of heart attack along with evidence of ischemia (e.g., ischemic ST changes on an ECG) in the first 48 hours after coronary artery bypass grafting (CABG). Although the Academic Research Consortium-2 Consensus stated there was no evidence-based threshold for cardiac troponin after CABG, they endorsed a threshold for the diagnosis of heart attack of ≥35 times the upper reference limit together with new evidence of ischemia, based on expert opinion. They also defined a threshold of ≥70 times the upper reference limit as a stand-alone criterion for clinically important periprocedural myocardial injury.
Globally, many hospitals now use high-sensitivity cardiac troponin assays; however, limited data are available to define a prognostically important degree of myocardial injury after cardiac surgery based on these assays. We undertook the Vascular Events in Surgery Patients Cohort Evaluation (VISION) Cardiac Surgery Study to examine clinical outcomes after cardiac surgery.
A primary objective was to determine the relationship between postoperative levels of high-sensitivity cardiac troponin I and the risk of death 30 days after cardiac surgery.
 Dr. PJ.Devereaux[/caption]
PJ Devereaux MD PhD
Professor of Medicine, and
of Health Research Methods, Evidence and Impact
McMaster University
President of the Society of Perioperative Research and Care
MedicalResearch.com: What is the background for this study?
Response: More than 1 million patients undergo cardiac surgery in the United States and Europe annually. Although cardiac surgery has the potential to improve and prolong a patient’s quality and duration of life, it is associated with complications. Prognostically important heart injury – detected by an elevated blood concentration of either cardiac troponin or creatine kinase myocardial MB isoform (CK-MB) – is one of the most common complications after cardiac surgery and is associated with increased mortality.
Although elevated CK-MB was historically used to define heart injury after cardiac surgery, this assay is no longer available in many hospitals worldwide, and consensus statements have recommended high-sensitivity cardiac troponin assays as the preferred biomarker. Based on expert opinion, the Fourth Universal Definition of Myocardial Infarction suggested that a cardiac troponin concentration >10 times the upper reference limit, in patients with a normal baseline measurement, should be the threshold used in the diagnosis of heart attack along with evidence of ischemia (e.g., ischemic ST changes on an ECG) in the first 48 hours after coronary artery bypass grafting (CABG). Although the Academic Research Consortium-2 Consensus stated there was no evidence-based threshold for cardiac troponin after CABG, they endorsed a threshold for the diagnosis of heart attack of ≥35 times the upper reference limit together with new evidence of ischemia, based on expert opinion. They also defined a threshold of ≥70 times the upper reference limit as a stand-alone criterion for clinically important periprocedural myocardial injury.
Globally, many hospitals now use high-sensitivity cardiac troponin assays; however, limited data are available to define a prognostically important degree of myocardial injury after cardiac surgery based on these assays. We undertook the Vascular Events in Surgery Patients Cohort Evaluation (VISION) Cardiac Surgery Study to examine clinical outcomes after cardiac surgery.
A primary objective was to determine the relationship between postoperative levels of high-sensitivity cardiac troponin I and the risk of death 30 days after cardiac surgery.
Dr. PJ.Devereaux[/caption]
PJ Devereaux MD PhD
Professor of Medicine, and
of Health Research Methods, Evidence and Impact
McMaster University
President of the Society of Perioperative Research and Care
MedicalResearch.com: What is the background for this study?
Response: More than 1 million patients undergo cardiac surgery in the United States and Europe annually. Although cardiac surgery has the potential to improve and prolong a patient’s quality and duration of life, it is associated with complications. Prognostically important heart injury – detected by an elevated blood concentration of either cardiac troponin or creatine kinase myocardial MB isoform (CK-MB) – is one of the most common complications after cardiac surgery and is associated with increased mortality.
Although elevated CK-MB was historically used to define heart injury after cardiac surgery, this assay is no longer available in many hospitals worldwide, and consensus statements have recommended high-sensitivity cardiac troponin assays as the preferred biomarker. Based on expert opinion, the Fourth Universal Definition of Myocardial Infarction suggested that a cardiac troponin concentration >10 times the upper reference limit, in patients with a normal baseline measurement, should be the threshold used in the diagnosis of heart attack along with evidence of ischemia (e.g., ischemic ST changes on an ECG) in the first 48 hours after coronary artery bypass grafting (CABG). Although the Academic Research Consortium-2 Consensus stated there was no evidence-based threshold for cardiac troponin after CABG, they endorsed a threshold for the diagnosis of heart attack of ≥35 times the upper reference limit together with new evidence of ischemia, based on expert opinion. They also defined a threshold of ≥70 times the upper reference limit as a stand-alone criterion for clinically important periprocedural myocardial injury.
Globally, many hospitals now use high-sensitivity cardiac troponin assays; however, limited data are available to define a prognostically important degree of myocardial injury after cardiac surgery based on these assays. We undertook the Vascular Events in Surgery Patients Cohort Evaluation (VISION) Cardiac Surgery Study to examine clinical outcomes after cardiac surgery.
A primary objective was to determine the relationship between postoperative levels of high-sensitivity cardiac troponin I and the risk of death 30 days after cardiac surgery. 


 Dr. Mapara[/caption]
Markus Y Mapara, MD
Professor of Medicine
Director of the Blood and Marrow Transplantation
Columbia University Medical Center
MedicalResearch.com: What is the background for this study? What are the main findings?
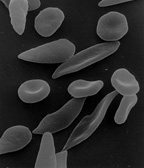
Response: Sickle cell disease is caused by a point mutation in the beta-globin gene of hemoglobin resulting in the production of abnormal hemoglobin which leads to formation of sickle-shaped RBC under conditions of low oxygen. Sickle cell disease affects about 100,000 patients in the US which are predominantly African American. The only curative approach is to perform an allogeneic bone marrow transplant which is however fraught with significant treatment-related risks if a matched sibling donor is not available.
The current study describes the successful application of a novel gene therapy to treat patients with sickle cell disease. The strategy is based on a gene-addition approach to introduce the genetic information for a Hemoglobin F-like molecule termed HgAT87Q into hematopoietic stem cells. The expression of this novel hemoglobin prevents polymerization of HgbS and has now been demonstrated to prevent the occurrence of vaso-occlusive pain crises in sickle cell disease patients.
Dr. Mapara[/caption]
Markus Y Mapara, MD
Professor of Medicine
Director of the Blood and Marrow Transplantation
Columbia University Medical Center
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Sickle cell disease is caused by a point mutation in the beta-globin gene of hemoglobin resulting in the production of abnormal hemoglobin which leads to formation of sickle-shaped RBC under conditions of low oxygen. Sickle cell disease affects about 100,000 patients in the US which are predominantly African American. The only curative approach is to perform an allogeneic bone marrow transplant which is however fraught with significant treatment-related risks if a matched sibling donor is not available.
The current study describes the successful application of a novel gene therapy to treat patients with sickle cell disease. The strategy is based on a gene-addition approach to introduce the genetic information for a Hemoglobin F-like molecule termed HgAT87Q into hematopoietic stem cells. The expression of this novel hemoglobin prevents polymerization of HgbS and has now been demonstrated to prevent the occurrence of vaso-occlusive pain crises in sickle cell disease patients.


 Dr. Dickerman[/caption]
Dr. Barbra Dickerman, PhD
CAUSALab investigator and instructor
Department of Epidemiology
Harvard T.H. Chan School of Public Health
MedicalResearch.com: What is the background for this study?
Response: Early randomized trials showed that the BNT162b2 (Pfizer-BioNTech) and mRNA-1273 (Moderna) vaccines were both remarkably effective at preventing symptomatic disease, when comparing each vaccine with no vaccine. However, head-to-head comparisons of these vaccines have been lacking, leaving open the question of which vaccine is more effective.
In this study, we analyzed the VA’s high-quality databases in a way that emulated the design of the hypothetical trial that would have answered this question. Specifically, we used the findings from the original trials to benchmark our methods and then extended them to provide novel evidence for the comparative effectiveness of these two vaccines in a real-world setting and across diverse subgroups and different time periods.
Dr. Dickerman[/caption]
Dr. Barbra Dickerman, PhD
CAUSALab investigator and instructor
Department of Epidemiology
Harvard T.H. Chan School of Public Health
MedicalResearch.com: What is the background for this study?
Response: Early randomized trials showed that the BNT162b2 (Pfizer-BioNTech) and mRNA-1273 (Moderna) vaccines were both remarkably effective at preventing symptomatic disease, when comparing each vaccine with no vaccine. However, head-to-head comparisons of these vaccines have been lacking, leaving open the question of which vaccine is more effective.
In this study, we analyzed the VA’s high-quality databases in a way that emulated the design of the hypothetical trial that would have answered this question. Specifically, we used the findings from the original trials to benchmark our methods and then extended them to provide novel evidence for the comparative effectiveness of these two vaccines in a real-world setting and across diverse subgroups and different time periods.



 Dr. Tian[/caption]
MedicalResearch.com Interview with:
Maoyi TIAN PhD
Program Head, Digital Health and Head, Injury & Trauma
Senior Research Fellow
The George Institute
MedicalResearch.com: What is the background for this study?
Response: There is clear evidence from the literature that sodium reduction or potassium supplementation can reduce blood pressure. Reduced blood pressure can also lead to a risk reduction for cardiovascular diseases.
Salt substitute is a reduced sodium added potassium product combined those effects. Previous research of salt substitute focus on the blood pressure outcome. There is no evidence if salt substitute can reduce the risk of cardiovascular diseases or pre-mature death. This study provided a definitive evidence for this unaddressed question.
MedicalResearch.com: What are the main findings?
The main findings of the research were:
Dr. Tian[/caption]
MedicalResearch.com Interview with:
Maoyi TIAN PhD
Program Head, Digital Health and Head, Injury & Trauma
Senior Research Fellow
The George Institute
MedicalResearch.com: What is the background for this study?
Response: There is clear evidence from the literature that sodium reduction or potassium supplementation can reduce blood pressure. Reduced blood pressure can also lead to a risk reduction for cardiovascular diseases.
Salt substitute is a reduced sodium added potassium product combined those effects. Previous research of salt substitute focus on the blood pressure outcome. There is no evidence if salt substitute can reduce the risk of cardiovascular diseases or pre-mature death. This study provided a definitive evidence for this unaddressed question.
MedicalResearch.com: What are the main findings?
The main findings of the research were:



 Prof. Reich[/caption]
Prof. Kristian Reich, MD, PhD
Professor for Translational Research in Inflammatory Skin Diseases
Institute for Health Services Research in Dermatology and Nursing
University Medical Center Hamburg-Eppendorf
MedicalResearch.com: What is the background for this study? What are the main findings?
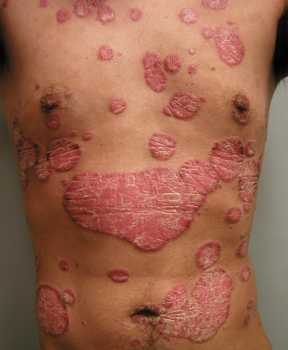
Response: Complete skin clearance is an important treatment goal for patients with psoriasis and is closely associated with treatment satisfaction and improved quality of life. However, it remains an unmet need for many patients.
The interleukin (IL)-17 isoforms IL-17A and IL-17F play central roles in psoriasis pathophysiology and are overexpressed in psoriatic tissues. Existing biologic therapies, such as secukinumab, inhibit IL-17A only. However, increasing evidence indicates that IL-17F contributes independently to the pathobiology of plaque psoriasis, and that blocking both IL-17A and IL-17F may lead to more complete suppression of inflammation and superior clinical outcomes, compared with blocking IL‑17A alone.
Bimekizumab is a humanized monoclonal IgG1 antibody that has been designed to selectively inhibit IL-17F in addition to IL-17A.
Prof. Reich[/caption]
Prof. Kristian Reich, MD, PhD
Professor for Translational Research in Inflammatory Skin Diseases
Institute for Health Services Research in Dermatology and Nursing
University Medical Center Hamburg-Eppendorf
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Complete skin clearance is an important treatment goal for patients with psoriasis and is closely associated with treatment satisfaction and improved quality of life. However, it remains an unmet need for many patients.
The interleukin (IL)-17 isoforms IL-17A and IL-17F play central roles in psoriasis pathophysiology and are overexpressed in psoriatic tissues. Existing biologic therapies, such as secukinumab, inhibit IL-17A only. However, increasing evidence indicates that IL-17F contributes independently to the pathobiology of plaque psoriasis, and that blocking both IL-17A and IL-17F may lead to more complete suppression of inflammation and superior clinical outcomes, compared with blocking IL‑17A alone.
Bimekizumab is a humanized monoclonal IgG1 antibody that has been designed to selectively inhibit IL-17F in addition to IL-17A.


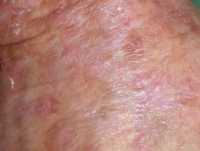
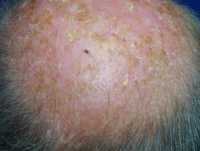
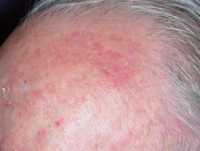 One example of actinic keratoses on scalp
One example of actinic keratoses on scalp







 Dr. Trudo[/caption]
Frank Trudo, MD MBA
Vice President, US Medical Affairs
Respiratory & Immunology
AstraZeneca
MedicalResearch.com: What is the background for this study?
Response: ETHOS is a randomized, double-blinded, multi-center, parallel-group, 52-week trial to assess the efficacy and safety of PT010 in symptomatic patients with moderate to very severe COPD and a history of exacerbation(s) in the previous year. Outcomes in the ETHOS trial included, as a primary endpoint, the rate of moderate or severe exacerbations.
MedicalResearch.com: How does PT010 differ from other treatments for COPD?
Dr. Trudo[/caption]
Frank Trudo, MD MBA
Vice President, US Medical Affairs
Respiratory & Immunology
AstraZeneca
MedicalResearch.com: What is the background for this study?
Response: ETHOS is a randomized, double-blinded, multi-center, parallel-group, 52-week trial to assess the efficacy and safety of PT010 in symptomatic patients with moderate to very severe COPD and a history of exacerbation(s) in the previous year. Outcomes in the ETHOS trial included, as a primary endpoint, the rate of moderate or severe exacerbations.
MedicalResearch.com: How does PT010 differ from other treatments for COPD?