Author Interviews, CDC, HPV, JAMA, Vaccine Studies / 17.09.2021
More Parents Express Concern About HPV Vaccination, Yet Reported Adverse Reactions Fall
MedicalResearch.com Interview with:
[caption id="attachment_58124" align="alignleft" width="133"] Dr. Sonawane[/caption]
Kalyani Sonawane, PhD
Assistant Professor of Management, Policy and Community Health
UTHealth School of Public Health in Houston
MedicalResearch.com: What is the background for this study?
Response: Safety concern regarding the HPV vaccine is one of the most significant barriers to vaccination. Our objective was to determine how many US adolescents did not initiate the HPV vaccine during 2015-2018 because their parents had concerns regarding the vaccine’s safety.
We also analyzed vaccine adverse event reporting data, in parallel, to understand if the public sentiment of HPV vaccine safety is in alignment with evidence from the vaccine safety surveillance system.
Dr. Sonawane[/caption]
Kalyani Sonawane, PhD
Assistant Professor of Management, Policy and Community Health
UTHealth School of Public Health in Houston
MedicalResearch.com: What is the background for this study?
Response: Safety concern regarding the HPV vaccine is one of the most significant barriers to vaccination. Our objective was to determine how many US adolescents did not initiate the HPV vaccine during 2015-2018 because their parents had concerns regarding the vaccine’s safety.
We also analyzed vaccine adverse event reporting data, in parallel, to understand if the public sentiment of HPV vaccine safety is in alignment with evidence from the vaccine safety surveillance system.
 Dr. Sonawane[/caption]
Kalyani Sonawane, PhD
Assistant Professor of Management, Policy and Community Health
UTHealth School of Public Health in Houston
MedicalResearch.com: What is the background for this study?
Response: Safety concern regarding the HPV vaccine is one of the most significant barriers to vaccination. Our objective was to determine how many US adolescents did not initiate the HPV vaccine during 2015-2018 because their parents had concerns regarding the vaccine’s safety.
We also analyzed vaccine adverse event reporting data, in parallel, to understand if the public sentiment of HPV vaccine safety is in alignment with evidence from the vaccine safety surveillance system.
Dr. Sonawane[/caption]
Kalyani Sonawane, PhD
Assistant Professor of Management, Policy and Community Health
UTHealth School of Public Health in Houston
MedicalResearch.com: What is the background for this study?
Response: Safety concern regarding the HPV vaccine is one of the most significant barriers to vaccination. Our objective was to determine how many US adolescents did not initiate the HPV vaccine during 2015-2018 because their parents had concerns regarding the vaccine’s safety.
We also analyzed vaccine adverse event reporting data, in parallel, to understand if the public sentiment of HPV vaccine safety is in alignment with evidence from the vaccine safety surveillance system. 




 Dr. Blumenthal[/caption]
Kimberly G. Blumenthal, MD, MSc
Massachusetts General Hospital
The Mongan Institute
Boston, MA 02114
[caption id="attachment_57828" align="alignleft" width="100"]
Dr. Blumenthal[/caption]
Kimberly G. Blumenthal, MD, MSc
Massachusetts General Hospital
The Mongan Institute
Boston, MA 02114
[caption id="attachment_57828" align="alignleft" width="100"] Dr. Krantz[/caption]
Matthew S. Krantz, MD
Division of Allergy, Pulmonary and Critical Care Medicine
Department of Medicine,
Vanderbilt University Medical Center,
Nashville, Tennessee
MedicalResearch.com: What is the background for this study?
Response: During the initial COVID-19 vaccine campaign with healthcare workers in December 2020, there was an unexpected higher than anticipated rate of immediate allergic reactions after Pfizer and Moderna mRNA vaccines. This prompted both patient and provider concerns, particularly in those with underlying allergic histories, on the associated risks for immediate allergic reactions with the mRNA vaccines.
Because of the significantly improved effectiveness of two doses of an mRNA vaccine compared to one dose, it was important to determine if those who experienced immediate allergic reaction symptoms after their first dose could go on to tolerate a second dose safely.
Dr. Krantz[/caption]
Matthew S. Krantz, MD
Division of Allergy, Pulmonary and Critical Care Medicine
Department of Medicine,
Vanderbilt University Medical Center,
Nashville, Tennessee
MedicalResearch.com: What is the background for this study?
Response: During the initial COVID-19 vaccine campaign with healthcare workers in December 2020, there was an unexpected higher than anticipated rate of immediate allergic reactions after Pfizer and Moderna mRNA vaccines. This prompted both patient and provider concerns, particularly in those with underlying allergic histories, on the associated risks for immediate allergic reactions with the mRNA vaccines.
Because of the significantly improved effectiveness of two doses of an mRNA vaccine compared to one dose, it was important to determine if those who experienced immediate allergic reaction symptoms after their first dose could go on to tolerate a second dose safely. 
 Dr. Blankson[/caption]
Joel N. Blankson, MD, PhD
Department of Infectious Diseases
Associate Professor
Cellular and Molecular Medicine Program
Johns Hopkins
MedicalResearch.com: What is the background for this study? Which vaccines did you evaluate?
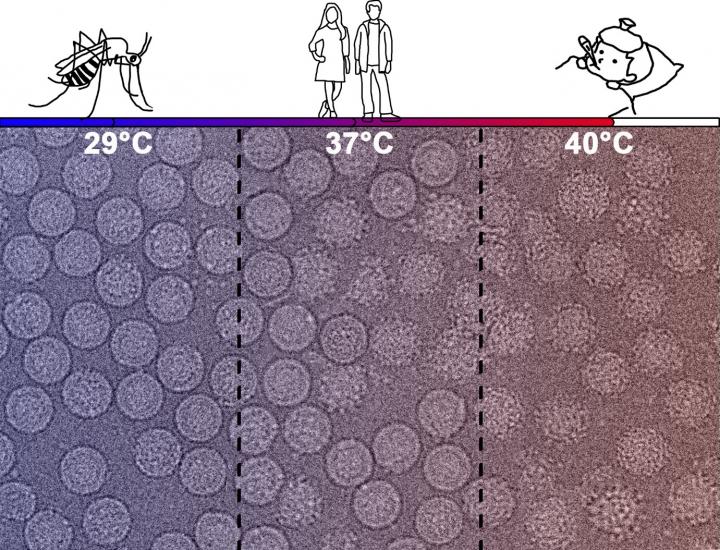
Response: Prior studies from several groups including our own have found T cell cross-recognition of peptides from SARS-CoV-2 and the common cold coronaviruses.
We asked whether as a result of this cross-reactivity, immunization with the SARS-CoV-2 vaccine would also enhance T cell responses to the common cold coronaviruses.
Prior studies also suggested that antibodies elicited from the mRNA vaccines had a reduced ability to neutralize the emerging variants of concern.
Most of the study participants had received the Pfizer vaccine, but a few had received the Moderna vaccine.
Dr. Blankson[/caption]
Joel N. Blankson, MD, PhD
Department of Infectious Diseases
Associate Professor
Cellular and Molecular Medicine Program
Johns Hopkins
MedicalResearch.com: What is the background for this study? Which vaccines did you evaluate?
Response: Prior studies from several groups including our own have found T cell cross-recognition of peptides from SARS-CoV-2 and the common cold coronaviruses.
We asked whether as a result of this cross-reactivity, immunization with the SARS-CoV-2 vaccine would also enhance T cell responses to the common cold coronaviruses.
Prior studies also suggested that antibodies elicited from the mRNA vaccines had a reduced ability to neutralize the emerging variants of concern.
Most of the study participants had received the Pfizer vaccine, but a few had received the Moderna vaccine. 
 Dr. Meyer, J.D.[/caption]
Michelle N. Meyer, PhD, JD
Assistant Professor & Associate Director, Research Ethics, Center for Translational Bioethics & Health Care Policy
Faculty Co-Director, Behavioral Insights Team, Steele Institute for Health Innovation
Assistant Professor of Bioethics
Geisinger Commonwealth School of Medicine
Geisinger, PA
MedicalResearch.com: What is the background for this study?
Response: Earlier research had found people are less likely to say they'll receive a COVID-19 vaccine offered to them under an Emergency Use Authorization (EUA) than one offered to them following full FDA approval. Earlier surveys had also found that only around 30% of health care workers intended to receive a COVID-19 vaccine. Because the public often looks to local health care workers for health advice, and in most prioritization schemes they were slated to be offered vaccines first, this was quite concerning for the prospect of achieving population immunity. Commenters had warned that if the FDA chose to make COVID-19 vaccines available under EUAs, that substantial efforts would need to be made to ensure trust. On Dec. 4, 2020, an announcement about anticipated vaccine availability was emailed to all 23,784 Geisinger employees, who were asked to indicate their intention to receive a vaccine when one was available to them and the reasons for any hesitation they might have.
Dr. Meyer, J.D.[/caption]
Michelle N. Meyer, PhD, JD
Assistant Professor & Associate Director, Research Ethics, Center for Translational Bioethics & Health Care Policy
Faculty Co-Director, Behavioral Insights Team, Steele Institute for Health Innovation
Assistant Professor of Bioethics
Geisinger Commonwealth School of Medicine
Geisinger, PA
MedicalResearch.com: What is the background for this study?
Response: Earlier research had found people are less likely to say they'll receive a COVID-19 vaccine offered to them under an Emergency Use Authorization (EUA) than one offered to them following full FDA approval. Earlier surveys had also found that only around 30% of health care workers intended to receive a COVID-19 vaccine. Because the public often looks to local health care workers for health advice, and in most prioritization schemes they were slated to be offered vaccines first, this was quite concerning for the prospect of achieving population immunity. Commenters had warned that if the FDA chose to make COVID-19 vaccines available under EUAs, that substantial efforts would need to be made to ensure trust. On Dec. 4, 2020, an announcement about anticipated vaccine availability was emailed to all 23,784 Geisinger employees, who were asked to indicate their intention to receive a vaccine when one was available to them and the reasons for any hesitation they might have.
 Laura M. Bogart, PhD
Senior Behavioral Scientist
RAND Corporation
Santa Monica, CA 90407-2138
MedicalResearch.com: What is the background for this study?
Response: Recent media polls continue to show that Black Americans are less likely to intend to get the COVID-19 vaccine than White Americans, and initial state data show a similar racial/ethnic disparity in vaccination rates. Initial uptake of the vaccine has been significantly affected by inequities in vaccine access and supply. In addition to these challenges, other factors contribute to hesitancy around vaccination, including self-perceived risk of infection, trust in the vaccine itself, trust in healthcare systems, healthcare providers, and policymakers who support the vaccine, and trust in the pharmaceutical industry and clinical research. In this study, we conducted a survey of a nationally representative sample of 207 Black Americans in late 2020, after initial COVID-19 vaccine effectiveness and safety data were released to the public. We also did in-depth interviews with a subsample of those surveyed who said that they would not get vaccinated. In addition, we engaged with a stakeholder advisory committee comprised of individuals who represent different subgroups and organizations in Black communities in the U.S., in order to discuss the results and make recommendations for policies to increase COVID-19 vaccination among Black Americans.
Laura M. Bogart, PhD
Senior Behavioral Scientist
RAND Corporation
Santa Monica, CA 90407-2138
MedicalResearch.com: What is the background for this study?
Response: Recent media polls continue to show that Black Americans are less likely to intend to get the COVID-19 vaccine than White Americans, and initial state data show a similar racial/ethnic disparity in vaccination rates. Initial uptake of the vaccine has been significantly affected by inequities in vaccine access and supply. In addition to these challenges, other factors contribute to hesitancy around vaccination, including self-perceived risk of infection, trust in the vaccine itself, trust in healthcare systems, healthcare providers, and policymakers who support the vaccine, and trust in the pharmaceutical industry and clinical research. In this study, we conducted a survey of a nationally representative sample of 207 Black Americans in late 2020, after initial COVID-19 vaccine effectiveness and safety data were released to the public. We also did in-depth interviews with a subsample of those surveyed who said that they would not get vaccinated. In addition, we engaged with a stakeholder advisory committee comprised of individuals who represent different subgroups and organizations in Black communities in the U.S., in order to discuss the results and make recommendations for policies to increase COVID-19 vaccination among Black Americans.

 Dr. Kriner[/caption]
Douglas L. Kriner, PhD
The Clinton Rossiter Professor in American Institutions
Department of Government
Cornell University
MedicalResearch.com: What is the background for this study?
Response: When a safe and effective vaccine for COVID-19 reaches the market, the world will not change overnight. Rather, government and public health individuals will have to develop a comprehensive plan to distribute the vaccine and to convince potentially wary Americans to take it.
Our study examined the influence of both specific vaccine characteristics and the politics surrounding it on public willingness to vaccinate. Both matter in important ways. For example, efficacy is unsurprisingly a major driver of public opinion; Americans are more willing to take a vaccine that is more efficacious.
Dr. Kriner[/caption]
Douglas L. Kriner, PhD
The Clinton Rossiter Professor in American Institutions
Department of Government
Cornell University
MedicalResearch.com: What is the background for this study?
Response: When a safe and effective vaccine for COVID-19 reaches the market, the world will not change overnight. Rather, government and public health individuals will have to develop a comprehensive plan to distribute the vaccine and to convince potentially wary Americans to take it.
Our study examined the influence of both specific vaccine characteristics and the politics surrounding it on public willingness to vaccinate. Both matter in important ways. For example, efficacy is unsurprisingly a major driver of public opinion; Americans are more willing to take a vaccine that is more efficacious.



 Dr. Hesse[/caption]
Dr. Elisabeth Hesse, MD
Epidemic Intelligence Service (EIS Officer)
Centers for Disease Control and Prevention
MedicalResearch.com: What is the background for this study?
Response: Over the last decade, there has been increasing attention given to shoulder injuries diagnosed after intramuscular vaccinations, with multiple publications of case reports and case series. However, to the best of our knowledge, there haven’t been any robust studies to determine how frequently this happens and what may make some people more likely than others to have shoulder injuries after vaccination. The Vaccine Safety Datalink (VSD) is an ideal system to use for such a study, because it contains medical and vaccination records of over 10 million people across the United States. We found that out of the 2.9 million people over the age of 3 who received an injectable flu vaccine (specifically, inactivated influenza vaccine) during the 2016-2017 flu season, fewer than 8 people per million vaccinated developed shoulder bursitis that can be attributed to the vaccination.
Dr. Hesse[/caption]
Dr. Elisabeth Hesse, MD
Epidemic Intelligence Service (EIS Officer)
Centers for Disease Control and Prevention
MedicalResearch.com: What is the background for this study?
Response: Over the last decade, there has been increasing attention given to shoulder injuries diagnosed after intramuscular vaccinations, with multiple publications of case reports and case series. However, to the best of our knowledge, there haven’t been any robust studies to determine how frequently this happens and what may make some people more likely than others to have shoulder injuries after vaccination. The Vaccine Safety Datalink (VSD) is an ideal system to use for such a study, because it contains medical and vaccination records of over 10 million people across the United States. We found that out of the 2.9 million people over the age of 3 who received an injectable flu vaccine (specifically, inactivated influenza vaccine) during the 2016-2017 flu season, fewer than 8 people per million vaccinated developed shoulder bursitis that can be attributed to the vaccination. Dr. Kempe[/caption]
Allison Kempe, MD, MPH
Ergen Family Endowed Chair in Pediatric Outcomes Research
Professor of Pediatrics, University of Colorado School of Medicine
Director of ACCORDS (Adult and Child Consortium for Health Outcomes Research and Delivery Science)
University of Colorado School of Medicine | Children’s Hospital Colorado
MedicalResearch.com: What is the background for this study?
Response: In 2019 the WHO designated vaccine hesitancy as one of the ten leading threats to global health. Although studies have assessed parental vaccine hesitancy in different localities and estimated vaccine refusals nationally, there is little recent US national data on the prevalence of hesitancy about routine childhood vaccines and national hesitancy rates for influenza vaccine have never been assessed. We used a hesitancy scale developed by the WHO to estimate levels of parental hesitancy for both routine childhood and childhood influenza vaccination
Dr. Kempe[/caption]
Allison Kempe, MD, MPH
Ergen Family Endowed Chair in Pediatric Outcomes Research
Professor of Pediatrics, University of Colorado School of Medicine
Director of ACCORDS (Adult and Child Consortium for Health Outcomes Research and Delivery Science)
University of Colorado School of Medicine | Children’s Hospital Colorado
MedicalResearch.com: What is the background for this study?
Response: In 2019 the WHO designated vaccine hesitancy as one of the ten leading threats to global health. Although studies have assessed parental vaccine hesitancy in different localities and estimated vaccine refusals nationally, there is little recent US national data on the prevalence of hesitancy about routine childhood vaccines and national hesitancy rates for influenza vaccine have never been assessed. We used a hesitancy scale developed by the WHO to estimate levels of parental hesitancy for both routine childhood and childhood influenza vaccination 

 Dr. Emily Parker Hyle[/caption]
Emily Parker Hyle, M.D.
Assistant Professor of Medicine
Massachusetts General Hospital
MedicalResearch.com: What is the background for this study?
Response: We found that many children who were planning to travel internationally were eligible for MMR vaccination prior to departure but often did not receive it - especially if they were aged 6 months to 6 years. That is because most children do not routinely receive their first dose of MMR till 12-15 months of age and their second dose of MMR till 4-6 years of age. However, ACIP recommendations are different for children who are traveling internationally. The risk of being infected with measles is much higher outside of the US, so it is recommended that children older than 1 year have had 2 MMR vaccinations and that children 6-12 months receive 1 MMR vaccination prior to travel. MMR vaccination is a safe and effective way to greatly reduce the risk of measles infection.
Dr. Emily Parker Hyle[/caption]
Emily Parker Hyle, M.D.
Assistant Professor of Medicine
Massachusetts General Hospital
MedicalResearch.com: What is the background for this study?
Response: We found that many children who were planning to travel internationally were eligible for MMR vaccination prior to departure but often did not receive it - especially if they were aged 6 months to 6 years. That is because most children do not routinely receive their first dose of MMR till 12-15 months of age and their second dose of MMR till 4-6 years of age. However, ACIP recommendations are different for children who are traveling internationally. The risk of being infected with measles is much higher outside of the US, so it is recommended that children older than 1 year have had 2 MMR vaccinations and that children 6-12 months receive 1 MMR vaccination prior to travel. MMR vaccination is a safe and effective way to greatly reduce the risk of measles infection.