Weight Research / 19.12.2024
Ozempic is All the Rage: But is it Safe?
 In recent years, Ozempic has become a cultural phenomenon, capturing headlines and transforming conversations about weight loss and diabetes management. What are the possible side effects of Ozempic? This question looms large as the medication continues to gain popularity among celebrities, social media influencers, and individuals struggling with weight management. Originally developed as a treatment for type 2 diabetes, Ozempic (semaglutide) has emerged as a powerful weight loss solution, sparking both excitement and concern within medical and public circles. The drug's rapid rise to prominence has created a complex narrative of hope, skepticism, and potential medical breakthrough.
The medication belongs to a class of drugs called glucagon-like peptide-1 (GLP-1) receptor agonists, which work by mimicking a hormone that targets areas of the brain involved in appetite regulation. By slowing down gastric emptying and reducing hunger, Ozempic helps patients consume fewer calories and lose significant amounts of weight. Clinical studies have demonstrated remarkable results, with patients experiencing substantial weight loss and improved metabolic health markers that extend far beyond traditional weight management approaches. The precision of its metabolic intervention has captured the attention of endocrinologists and weight management specialists worldwide.
In recent years, Ozempic has become a cultural phenomenon, capturing headlines and transforming conversations about weight loss and diabetes management. What are the possible side effects of Ozempic? This question looms large as the medication continues to gain popularity among celebrities, social media influencers, and individuals struggling with weight management. Originally developed as a treatment for type 2 diabetes, Ozempic (semaglutide) has emerged as a powerful weight loss solution, sparking both excitement and concern within medical and public circles. The drug's rapid rise to prominence has created a complex narrative of hope, skepticism, and potential medical breakthrough.
The medication belongs to a class of drugs called glucagon-like peptide-1 (GLP-1) receptor agonists, which work by mimicking a hormone that targets areas of the brain involved in appetite regulation. By slowing down gastric emptying and reducing hunger, Ozempic helps patients consume fewer calories and lose significant amounts of weight. Clinical studies have demonstrated remarkable results, with patients experiencing substantial weight loss and improved metabolic health markers that extend far beyond traditional weight management approaches. The precision of its metabolic intervention has captured the attention of endocrinologists and weight management specialists worldwide.






















 Dr. Magruder[/caption]
Matthew Magruder, MD PGY3
Orthopaedic Residency Program
Department of Orthopaedic Surgery and Rehabilitation
Maimonides Medical Center
MedicalResearch.com: What is the background for this study?
Response: The prevalence of obesity and diabetes mellitus has reached epidemic proportions. Approximately 37.3 million people in the United States, accounting for 11.3% of the total population, have diabetes, and 100.1 million, or 41.9%, of all US citizens are obese. Furthermore, these numbers are only projected to increase in the coming decades. This is an issue for orthopaedic surgeons because diabetes and obesity have consistently been demonstrated to be risk factors for complications following total joint replacements, especially total hip replacements. Therefore, we are in desperate need of new and more effective tools in mitigating the risk of poor outcomes in our joint replacement patients.
Semaglutide, and other GLP-1 agonists, are potentially a new tool that can be used to help decrease the risks following joint replacement surgery. Initially a medication to treat diabetes, semaglutide has recently been approved by the FDA to treat obesity as well, as randomized controlled trials have consistently demonstrated significant weight loss with minimal side effects. The purpose of our study was to see what effect the use of semaglutide had on total hip arthroplasty patient outcomes.
Dr. Magruder[/caption]
Matthew Magruder, MD PGY3
Orthopaedic Residency Program
Department of Orthopaedic Surgery and Rehabilitation
Maimonides Medical Center
MedicalResearch.com: What is the background for this study?
Response: The prevalence of obesity and diabetes mellitus has reached epidemic proportions. Approximately 37.3 million people in the United States, accounting for 11.3% of the total population, have diabetes, and 100.1 million, or 41.9%, of all US citizens are obese. Furthermore, these numbers are only projected to increase in the coming decades. This is an issue for orthopaedic surgeons because diabetes and obesity have consistently been demonstrated to be risk factors for complications following total joint replacements, especially total hip replacements. Therefore, we are in desperate need of new and more effective tools in mitigating the risk of poor outcomes in our joint replacement patients.
Semaglutide, and other GLP-1 agonists, are potentially a new tool that can be used to help decrease the risks following joint replacement surgery. Initially a medication to treat diabetes, semaglutide has recently been approved by the FDA to treat obesity as well, as randomized controlled trials have consistently demonstrated significant weight loss with minimal side effects. The purpose of our study was to see what effect the use of semaglutide had on total hip arthroplasty patient outcomes.

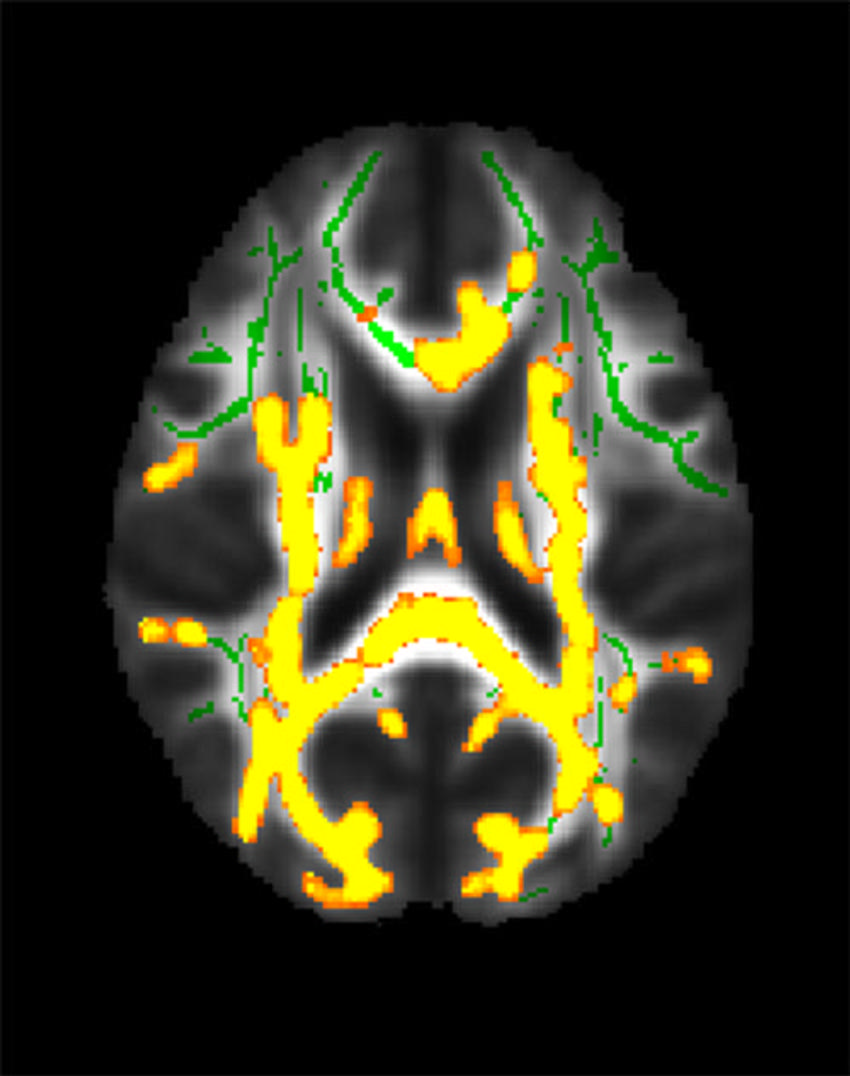
 Dr. Dolatshahi[/caption]
Mahsa Dolatshahi, M.D., M.P.H.
Post-doctoral research fellow
Mallinckrodt Institute of Radiology (MIR)
Washington University School of Medicine
St. Louis
MedicalResearch.com: What is the background for this study?
Response: Obesity at midlife is recognized as a risk factor for developing Alzheimer disease decades afterwards. However, body mass index on its own does not adequately represent the risks associated with obesity.
In this study, we went beyond BMI and considered anatomical distribution of body fat, including the metabolically active visceral fat in the belly, and showed its association with Alzheimer pathology in the form of amyloid proteins. In addition, visceral fat along with obesity and insulin resistance were associated with thinning of brain cortex, as early as midlife.
Dr. Dolatshahi[/caption]
Mahsa Dolatshahi, M.D., M.P.H.
Post-doctoral research fellow
Mallinckrodt Institute of Radiology (MIR)
Washington University School of Medicine
St. Louis
MedicalResearch.com: What is the background for this study?
Response: Obesity at midlife is recognized as a risk factor for developing Alzheimer disease decades afterwards. However, body mass index on its own does not adequately represent the risks associated with obesity.
In this study, we went beyond BMI and considered anatomical distribution of body fat, including the metabolically active visceral fat in the belly, and showed its association with Alzheimer pathology in the form of amyloid proteins. In addition, visceral fat along with obesity and insulin resistance were associated with thinning of brain cortex, as early as midlife.



 Dr. GALBIATI[/caption]
Francesca Galbiati, MD
Clinical/Research fellow in Endocrinology
Massachusetts General Hospital
MedicalResearch.com: What is the background for this study?
Response: Arginine-vasopressin (AVP) is a neurohormone well known for its role in water balance regulation. It promotes renal water absorption in the kidney, to maintain normal sodium levels in the blood via a tightly controlled osmotic regulation. Besides AVP classical role, data have shown that AVP effects extend beyond water balance regulation. Animal studies have shown that AVP has metabolic effects, including reducing food intake, inducing lipolysis, and promoting muscle regeneration in male mice.
Furthermore, AVP is regulated differently in males and females, and affects cognition differently across sexes, a phenomenon called sexual dimorphism. However, it is unknown whether its dimorphism translates to metabolism. Also, findings on AVP metabolic role are inconsistent, possibly due to the opposing effects of AVP at different receptor subtypes, which regulation is still largely unknown. We performed this study to better investigate AVP metabolic role, and explore sex differences. We hypothesized that AVP would be positively associated with BMI, adiposity, and lean mass (acting as a signal of energy availability). We also predicted that relationships between AVP and body composition measures would differ by sex. We used the AVP area under the curve around a standardized meal to better capture repeated measures in response to food intake (that directly impacts energy availability). This also allowed to avoid the possible risk of fluctuating AVP levels due to possible pulsatile secretion.
Dr. GALBIATI[/caption]
Francesca Galbiati, MD
Clinical/Research fellow in Endocrinology
Massachusetts General Hospital
MedicalResearch.com: What is the background for this study?
Response: Arginine-vasopressin (AVP) is a neurohormone well known for its role in water balance regulation. It promotes renal water absorption in the kidney, to maintain normal sodium levels in the blood via a tightly controlled osmotic regulation. Besides AVP classical role, data have shown that AVP effects extend beyond water balance regulation. Animal studies have shown that AVP has metabolic effects, including reducing food intake, inducing lipolysis, and promoting muscle regeneration in male mice.
Furthermore, AVP is regulated differently in males and females, and affects cognition differently across sexes, a phenomenon called sexual dimorphism. However, it is unknown whether its dimorphism translates to metabolism. Also, findings on AVP metabolic role are inconsistent, possibly due to the opposing effects of AVP at different receptor subtypes, which regulation is still largely unknown. We performed this study to better investigate AVP metabolic role, and explore sex differences. We hypothesized that AVP would be positively associated with BMI, adiposity, and lean mass (acting as a signal of energy availability). We also predicted that relationships between AVP and body composition measures would differ by sex. We used the AVP area under the curve around a standardized meal to better capture repeated measures in response to food intake (that directly impacts energy availability). This also allowed to avoid the possible risk of fluctuating AVP levels due to possible pulsatile secretion.
