Author Interviews, Cancer Research, JAMA, Lung Cancer, Smoking, USPSTF / 16.03.2021
USPSTF: New Recommendations Double Number of People Eligible for Lung Cancer Screening
MedicalResearch.com Interview with:
[caption id="attachment_56962" align="alignleft" width="133"] Dr. Wong[/caption]
John B. Wong, M.D.
Chief Scientific Officer,
Vice Chair for Clinical Affairs
Chief of the Division of Clinical Decision Making
Primary Care Clinician
Department of Medicine
Tufts Medical Center
MedicalResearch.com: What is the background for this study?
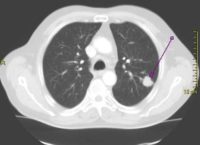
Response: Lung cancer is the leading cause of cancer death for both men and women in the United States. More than 200,000 people are diagnosed with this devastating disease each year. Smoking is the leading cause of lung cancer, resulting in the vast majority of lung cancers in the United States.
Dr. Wong[/caption]
John B. Wong, M.D.
Chief Scientific Officer,
Vice Chair for Clinical Affairs
Chief of the Division of Clinical Decision Making
Primary Care Clinician
Department of Medicine
Tufts Medical Center
MedicalResearch.com: What is the background for this study?
Response: Lung cancer is the leading cause of cancer death for both men and women in the United States. More than 200,000 people are diagnosed with this devastating disease each year. Smoking is the leading cause of lung cancer, resulting in the vast majority of lung cancers in the United States.
 Dr. Wong[/caption]
John B. Wong, M.D.
Chief Scientific Officer,
Vice Chair for Clinical Affairs
Chief of the Division of Clinical Decision Making
Primary Care Clinician
Department of Medicine
Tufts Medical Center
MedicalResearch.com: What is the background for this study?
Response: Lung cancer is the leading cause of cancer death for both men and women in the United States. More than 200,000 people are diagnosed with this devastating disease each year. Smoking is the leading cause of lung cancer, resulting in the vast majority of lung cancers in the United States.
Dr. Wong[/caption]
John B. Wong, M.D.
Chief Scientific Officer,
Vice Chair for Clinical Affairs
Chief of the Division of Clinical Decision Making
Primary Care Clinician
Department of Medicine
Tufts Medical Center
MedicalResearch.com: What is the background for this study?
Response: Lung cancer is the leading cause of cancer death for both men and women in the United States. More than 200,000 people are diagnosed with this devastating disease each year. Smoking is the leading cause of lung cancer, resulting in the vast majority of lung cancers in the United States.