Author Interviews, NYU/NYMC, Race/Ethnic Diversity, Rheumatology / 21.01.2021
Lupus Affects Women and Minorities More Than Other Groups
MedicalResearch.com Interview with:
[caption id="attachment_56448" align="alignleft" width="200"] Dr. Izmirly[/caption]
Peter Izmirly, M.D.
Associate Professor of Medicine, NYU School of Medicine
Director of Inpatient Rheumatology, Bellevue Hospital Center
co-Director, NYU-Hospital for Joint Diseases Lupus Clinic
Research Office Address:
NYU School of Medicine
New York, NY 10016
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Knowing how many people have systemic lupus erythematosus (SLE) is limited, particularly for racial/ethnic subgroups in the United States.
Our work provides accurate estimates of who has (SLE) among the major racial/ethnic groups in the United States and that our estimates for SLE approach the FDA’s definition or a rare disease.
Dr. Izmirly[/caption]
Peter Izmirly, M.D.
Associate Professor of Medicine, NYU School of Medicine
Director of Inpatient Rheumatology, Bellevue Hospital Center
co-Director, NYU-Hospital for Joint Diseases Lupus Clinic
Research Office Address:
NYU School of Medicine
New York, NY 10016
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Knowing how many people have systemic lupus erythematosus (SLE) is limited, particularly for racial/ethnic subgroups in the United States.
Our work provides accurate estimates of who has (SLE) among the major racial/ethnic groups in the United States and that our estimates for SLE approach the FDA’s definition or a rare disease.
 Dr. Izmirly[/caption]
Peter Izmirly, M.D.
Associate Professor of Medicine, NYU School of Medicine
Director of Inpatient Rheumatology, Bellevue Hospital Center
co-Director, NYU-Hospital for Joint Diseases Lupus Clinic
Research Office Address:
NYU School of Medicine
New York, NY 10016
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Knowing how many people have systemic lupus erythematosus (SLE) is limited, particularly for racial/ethnic subgroups in the United States.
Our work provides accurate estimates of who has (SLE) among the major racial/ethnic groups in the United States and that our estimates for SLE approach the FDA’s definition or a rare disease.
Dr. Izmirly[/caption]
Peter Izmirly, M.D.
Associate Professor of Medicine, NYU School of Medicine
Director of Inpatient Rheumatology, Bellevue Hospital Center
co-Director, NYU-Hospital for Joint Diseases Lupus Clinic
Research Office Address:
NYU School of Medicine
New York, NY 10016
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: Knowing how many people have systemic lupus erythematosus (SLE) is limited, particularly for racial/ethnic subgroups in the United States.
Our work provides accurate estimates of who has (SLE) among the major racial/ethnic groups in the United States and that our estimates for SLE approach the FDA’s definition or a rare disease.



 Dr. LaMoreaux[/caption]
Brian LaMoreaux, M.D., M.S.
Medical Director, Medical Affairs
Horizon Therapeutics
MedicalResearch.com: What is the background for this study?
Response: Hyperuricemia is associated with non-alcoholic fatty liver disease (NAFLD) but the relationship to fibrosis remains uncertain. Moreover, it is not known whether lowering serum urate will affect the course of NAFLD.
Dr. LaMoreaux[/caption]
Brian LaMoreaux, M.D., M.S.
Medical Director, Medical Affairs
Horizon Therapeutics
MedicalResearch.com: What is the background for this study?
Response: Hyperuricemia is associated with non-alcoholic fatty liver disease (NAFLD) but the relationship to fibrosis remains uncertain. Moreover, it is not known whether lowering serum urate will affect the course of NAFLD.


 Prof. Dagna[/caption]
Prof. Lorenzo Dagna MD FACP
Ospedale San Raffaele and
Vita-Salute San Raffaele University
Milan, Italy
MedicalResearch.com: What is the background for this study?
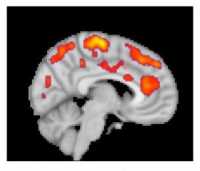
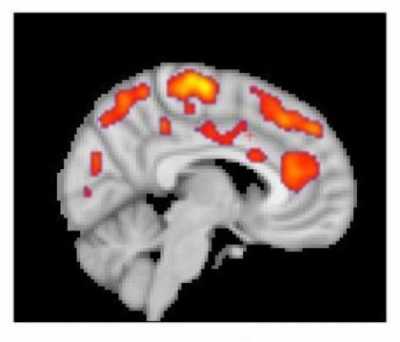
Response: Upon encountering pathogens, our immune system produces pro-inflammatory mediators, called cytokines. Cytokines activate cells from the immune system. In most people, production of cytokines is an appropriate and protective response to infection. However, some individuals develop excessive and detrimental inflammatory responses, which are even more harmful than the pathogen itself to the host organism.
We hypothesized that some patients with COVID-19 might develop excessive and detrimental inflammation, and that treatment with anti-inflammatory agents might be beneficial in this population.
Anakinra is an inhibitor of the pro-inflammatory molecule interleukin 1 (IL-1). It was originally marketed for the treatment of rheumatoid arthritis, but is now mostly used to treat a variety of pediatric inflammatory diseases.
Prof. Dagna[/caption]
Prof. Lorenzo Dagna MD FACP
Ospedale San Raffaele and
Vita-Salute San Raffaele University
Milan, Italy
MedicalResearch.com: What is the background for this study?
Response: Upon encountering pathogens, our immune system produces pro-inflammatory mediators, called cytokines. Cytokines activate cells from the immune system. In most people, production of cytokines is an appropriate and protective response to infection. However, some individuals develop excessive and detrimental inflammatory responses, which are even more harmful than the pathogen itself to the host organism.
We hypothesized that some patients with COVID-19 might develop excessive and detrimental inflammation, and that treatment with anti-inflammatory agents might be beneficial in this population.
Anakinra is an inhibitor of the pro-inflammatory molecule interleukin 1 (IL-1). It was originally marketed for the treatment of rheumatoid arthritis, but is now mostly used to treat a variety of pediatric inflammatory diseases.















 Dr. Emamifar[/caption]
Dr. Amir Emamifar, MD
Department of Rheumatology
Odense university Hospital
Svendborg Hospital, Denmark
[caption id="attachment_23869" align="alignleft" width="139"]
Dr. Emamifar[/caption]
Dr. Amir Emamifar, MD
Department of Rheumatology
Odense university Hospital
Svendborg Hospital, Denmark
[caption id="attachment_23869" align="alignleft" width="139"] Dr. Hansen[/caption]
Associate Professor
Dr. Inger Marie Jensen Hansen, PhD, DMsci
Department of Rheumatology
Odense university Hospital
Svendborg Hospital
University of Southern Denmark
MedicalResearch.com: What is the background for this study?
Response: Rheumatoid arthritis is a systemic, inflammatory disease that affects 1% of the general population. Apart from main articular manifestations, rheumatoid arthritis may involve other organs including heart, lung, skin, and eye. The auditory system can be affected during the course of the disease as well; however the association between rheumatoid arthritis and hearing impairment has not been clearly defined. It seems that hearing impairment in rheumatoid arthritis is a multifactorial disease affecting by environmental factors and disease and patient characteristics. We did a comprehensive review of all published data to reveal the potential link between rheumatoid arthritis and hearing impairment.
Dr. Hansen[/caption]
Associate Professor
Dr. Inger Marie Jensen Hansen, PhD, DMsci
Department of Rheumatology
Odense university Hospital
Svendborg Hospital
University of Southern Denmark
MedicalResearch.com: What is the background for this study?
Response: Rheumatoid arthritis is a systemic, inflammatory disease that affects 1% of the general population. Apart from main articular manifestations, rheumatoid arthritis may involve other organs including heart, lung, skin, and eye. The auditory system can be affected during the course of the disease as well; however the association between rheumatoid arthritis and hearing impairment has not been clearly defined. It seems that hearing impairment in rheumatoid arthritis is a multifactorial disease affecting by environmental factors and disease and patient characteristics. We did a comprehensive review of all published data to reveal the potential link between rheumatoid arthritis and hearing impairment.