Author Interviews, Heart Disease, JACC, Race/Ethnic Diversity, Transplantation, Yale / 17.05.2021
Heart Transplantation Outcomes: Racial Disparities Exist But Narrowing
MedicalResearch.com Interview with:
[caption id="attachment_57306" align="alignleft" width="107"] Dr. Fuery[/caption]
Michael Fuery, MD
Department of Internal Medicine
Yale School of Medicine
[caption id="attachment_57307" align="alignleft" width="100"]
Dr. Fuery[/caption]
Michael Fuery, MD
Department of Internal Medicine
Yale School of Medicine
[caption id="attachment_57307" align="alignleft" width="100"] Dr. Clarke[/caption]
Katherine Clark, MD MBA
Division of Cardiovascular Medicine
Department of Internal Medicine
Yale School of Medicine
MedicalResearch.com: What is the background for this study?
Response: Racial and ethnic disparities affect cardiac transplantation outcomes. In cohort analyses of racial and ethnic groups from the previous three decades, Black patients were constantly at a higher risk of mortality after cardiac transplantation. In 2018, the United Network for Organ Sharing (UNOS) revised the allocation system to expand access to organs for the most medically urgent patients and reduce disparities and regional differences. We sought to evaluate contemporary trends and impact of the new 2018 allocation system.
Dr. Clarke[/caption]
Katherine Clark, MD MBA
Division of Cardiovascular Medicine
Department of Internal Medicine
Yale School of Medicine
MedicalResearch.com: What is the background for this study?
Response: Racial and ethnic disparities affect cardiac transplantation outcomes. In cohort analyses of racial and ethnic groups from the previous three decades, Black patients were constantly at a higher risk of mortality after cardiac transplantation. In 2018, the United Network for Organ Sharing (UNOS) revised the allocation system to expand access to organs for the most medically urgent patients and reduce disparities and regional differences. We sought to evaluate contemporary trends and impact of the new 2018 allocation system.
 Dr. Fuery[/caption]
Michael Fuery, MD
Department of Internal Medicine
Yale School of Medicine
[caption id="attachment_57307" align="alignleft" width="100"]
Dr. Fuery[/caption]
Michael Fuery, MD
Department of Internal Medicine
Yale School of Medicine
[caption id="attachment_57307" align="alignleft" width="100"] Dr. Clarke[/caption]
Katherine Clark, MD MBA
Division of Cardiovascular Medicine
Department of Internal Medicine
Yale School of Medicine
MedicalResearch.com: What is the background for this study?
Response: Racial and ethnic disparities affect cardiac transplantation outcomes. In cohort analyses of racial and ethnic groups from the previous three decades, Black patients were constantly at a higher risk of mortality after cardiac transplantation. In 2018, the United Network for Organ Sharing (UNOS) revised the allocation system to expand access to organs for the most medically urgent patients and reduce disparities and regional differences. We sought to evaluate contemporary trends and impact of the new 2018 allocation system.
Dr. Clarke[/caption]
Katherine Clark, MD MBA
Division of Cardiovascular Medicine
Department of Internal Medicine
Yale School of Medicine
MedicalResearch.com: What is the background for this study?
Response: Racial and ethnic disparities affect cardiac transplantation outcomes. In cohort analyses of racial and ethnic groups from the previous three decades, Black patients were constantly at a higher risk of mortality after cardiac transplantation. In 2018, the United Network for Organ Sharing (UNOS) revised the allocation system to expand access to organs for the most medically urgent patients and reduce disparities and regional differences. We sought to evaluate contemporary trends and impact of the new 2018 allocation system.












 Dr. Jordan[/caption]
Stanley C. Jordan, M.D
Director, Division of Nephrology
Medical Director, Kidney Transplant Program
Medical Director, Human Leukocyte Antigen and Transplant Immunology Laboratory
Cedars-Sinai, Los Angeles, CA
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: The background for this study is as follows: Patients who are highly HLA sensitized have antibodies to transplant targets create an immunologic barrier to transplant. Currently, there are no approved therapies for elimination of these antibodies. Desensitization is available but is not always successful and most desensitized patients are still transplanted with a positive crossmatch. Thus, many patients are not able to receive life-saving kidney transplants unless newer therapies to remove antibodies are found.
The findings of our study published in the New England Journal of Medicine revealed that the use of the enzyme from streptococcal pyogenes called IdeS® (IgG endopeptidase) is very effective in eliminating donor specific antibodies and allowing transplantation to occur. Antibodies were eliminated from one week up to two months after one treatment with Ides® allowing a safe environment for the transplant to occur. Rejections episodes did occur in some of the patients but were generally mild and easily treatable. Only one patient of 25 lost his allograft during the study. Thus, the study shows promising results for a new approach for elimination of pathogenic antibodies that did not exist before.
Dr. Jordan[/caption]
Stanley C. Jordan, M.D
Director, Division of Nephrology
Medical Director, Kidney Transplant Program
Medical Director, Human Leukocyte Antigen and Transplant Immunology Laboratory
Cedars-Sinai, Los Angeles, CA
MedicalResearch.com: What is the background for this study? What are the main findings?
Response: The background for this study is as follows: Patients who are highly HLA sensitized have antibodies to transplant targets create an immunologic barrier to transplant. Currently, there are no approved therapies for elimination of these antibodies. Desensitization is available but is not always successful and most desensitized patients are still transplanted with a positive crossmatch. Thus, many patients are not able to receive life-saving kidney transplants unless newer therapies to remove antibodies are found.
The findings of our study published in the New England Journal of Medicine revealed that the use of the enzyme from streptococcal pyogenes called IdeS® (IgG endopeptidase) is very effective in eliminating donor specific antibodies and allowing transplantation to occur. Antibodies were eliminated from one week up to two months after one treatment with Ides® allowing a safe environment for the transplant to occur. Rejections episodes did occur in some of the patients but were generally mild and easily treatable. Only one patient of 25 lost his allograft during the study. Thus, the study shows promising results for a new approach for elimination of pathogenic antibodies that did not exist before.




 Dr. Jaimin Trivedi[/caption]
Jaimin Trivedi, MD, MPH
Instructor
Department of Cardiovascular and Thoracic Surgery
University of Louisville
Louisville, KY 40202
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Trivedi: There is a donor heart shortage in United States and certain donor hearts are likely to be turned down because the donors required cardiopulmonary resuscitation (CPR) prior to procurement. The rationale behind conducting the study was to identify impact of donor CPR and its duration on recipient survival after transplantation.
Our findings show that presence of CPR and duration of CPR does not adversely impact the post heart transplant survival. The study also shows that ejection fraction and peak cardiac troponins between the CPR and non-CPR donors were comparable at time of transplant suggesting recovery of cardiac function.
Dr. Jaimin Trivedi[/caption]
Jaimin Trivedi, MD, MPH
Instructor
Department of Cardiovascular and Thoracic Surgery
University of Louisville
Louisville, KY 40202
MedicalResearch.com: What is the background for this study? What are the main findings?
Dr. Trivedi: There is a donor heart shortage in United States and certain donor hearts are likely to be turned down because the donors required cardiopulmonary resuscitation (CPR) prior to procurement. The rationale behind conducting the study was to identify impact of donor CPR and its duration on recipient survival after transplantation.
Our findings show that presence of CPR and duration of CPR does not adversely impact the post heart transplant survival. The study also shows that ejection fraction and peak cardiac troponins between the CPR and non-CPR donors were comparable at time of transplant suggesting recovery of cardiac function.



 Pritesh Karia[/caption]
MedicalResearch.com Interview with:
Pritesh Karia[/caption]
MedicalResearch.com Interview with: Dr. Sergio Acuna[/caption]
MedicalResearch.com Interview with:
Sergio A. Acuna, MD
Graduate Student at St. Michael's Hospital and IHPME
University of Toronto
Medical Research: What is the background for this study?
Dr. Acuna: Solid organ transplant recipients are known to be at greater risk of developing cancers compared to the general population; however, because they are also at high increased risk of mortality from non-cancer causes, the risk of cancer morality in this population is unclear. As previous studies on this topic have reported disparate findings, the cancer mortality risk in this population remained uncertain.
Medical Research: What are the main findings?
Dr. Acuna: Our study provides conclusive evidence that solid organ transplant recipients are at increased risk of cancer mortality. Our findings demonstrate that solid organ transplant recipients are at increased risk of cancer death compared to the general population regardless of age, transplanted organ, and year of transplantation, and indicate cancer is a substantial cause of death in this population.
Dr. Sergio Acuna[/caption]
MedicalResearch.com Interview with:
Sergio A. Acuna, MD
Graduate Student at St. Michael's Hospital and IHPME
University of Toronto
Medical Research: What is the background for this study?
Dr. Acuna: Solid organ transplant recipients are known to be at greater risk of developing cancers compared to the general population; however, because they are also at high increased risk of mortality from non-cancer causes, the risk of cancer morality in this population is unclear. As previous studies on this topic have reported disparate findings, the cancer mortality risk in this population remained uncertain.
Medical Research: What are the main findings?
Dr. Acuna: Our study provides conclusive evidence that solid organ transplant recipients are at increased risk of cancer mortality. Our findings demonstrate that solid organ transplant recipients are at increased risk of cancer death compared to the general population regardless of age, transplanted organ, and year of transplantation, and indicate cancer is a substantial cause of death in this population.
 Dr. Cassie Kennedy[/caption]
MedicalResearch.com Interview with:
Cassie Kennedy, M.D.
Pulmonology and Critical Care Medicine
Mayo Clinic
Medical Research: What is the background for this study?
Dr. Kennedy: Lung transplant is a surgical procedure that can offer extended life expectancy and improved quality of life to selected patients with end-stage lung disease. However there are about 1700 patients awaiting lung transplant at any given time in the United States because transplant recipients far exceed potential donors. In addition, even with carefully chosen candidates, lung transplant recipients live on average about 5.5 years. It is therefore very important for transplant physicians to choose patients who will receive the most benefit from their lung transplant.
Frailty (defined as an increased vulnerability to adverse health outcomes) has typically been a subjective consideration by transplant physicians when choosing lung transplant candidates. The emergence of more objective and reproducible frailty measures from the geriatric literature present an opportunity to study the prevalence of frailty in lung transplant (despite that subjective screening) and to determine whether the presence of frailty has any impact on patient outcomes.
Medical Research: What are the main findings?
Dr. Kennedy: Frailty is quite common --46 percent of our patient cohort was frail by the Frailty Deficit Index. We also saw a significant association between frailty and worsened survival following lung transplantation: one-year survival rate for frail patients was 71.7 percent, compared to 92.9 percent for patients who were not frail. At three years this difference in survival persisted--the survival rate for frail patients was 41.3 percent, compared to 66.1 percent for patients who were not frail.
Dr. Cassie Kennedy[/caption]
MedicalResearch.com Interview with:
Cassie Kennedy, M.D.
Pulmonology and Critical Care Medicine
Mayo Clinic
Medical Research: What is the background for this study?
Dr. Kennedy: Lung transplant is a surgical procedure that can offer extended life expectancy and improved quality of life to selected patients with end-stage lung disease. However there are about 1700 patients awaiting lung transplant at any given time in the United States because transplant recipients far exceed potential donors. In addition, even with carefully chosen candidates, lung transplant recipients live on average about 5.5 years. It is therefore very important for transplant physicians to choose patients who will receive the most benefit from their lung transplant.
Frailty (defined as an increased vulnerability to adverse health outcomes) has typically been a subjective consideration by transplant physicians when choosing lung transplant candidates. The emergence of more objective and reproducible frailty measures from the geriatric literature present an opportunity to study the prevalence of frailty in lung transplant (despite that subjective screening) and to determine whether the presence of frailty has any impact on patient outcomes.
Medical Research: What are the main findings?
Dr. Kennedy: Frailty is quite common --46 percent of our patient cohort was frail by the Frailty Deficit Index. We also saw a significant association between frailty and worsened survival following lung transplantation: one-year survival rate for frail patients was 71.7 percent, compared to 92.9 percent for patients who were not frail. At three years this difference in survival persisted--the survival rate for frail patients was 41.3 percent, compared to 66.1 percent for patients who were not frail.
 Dr. Schlansky[/caption]
MedicalResearch.com Interview with:
Barry Schlansky, M.D., M.P.H
Assistant Professor of Medicine
Oregon Health & Science University
Medical Research: What are the main findings and significance of this study?
Dr. Schlansky: This study examines how obese patients fare before and after liver transplantation. Similar to other researchers, we found that obese patients do just as well as normal weight patients after liver transplantation. We were surprised, however, to find that very obese patients died more often while on the wait list before liver transplant.
Dr. Schlansky[/caption]
MedicalResearch.com Interview with:
Barry Schlansky, M.D., M.P.H
Assistant Professor of Medicine
Oregon Health & Science University
Medical Research: What are the main findings and significance of this study?
Dr. Schlansky: This study examines how obese patients fare before and after liver transplantation. Similar to other researchers, we found that obese patients do just as well as normal weight patients after liver transplantation. We were surprised, however, to find that very obese patients died more often while on the wait list before liver transplant.
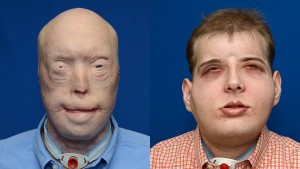 Patrick Hardison before surgery (left) and in November 2015, nearly three months after the surgery.[/caption]
Mr. Hardison was referred to
Patrick Hardison before surgery (left) and in November 2015, nearly three months after the surgery.[/caption]
Mr. Hardison was referred to  Dr. Givens
Dr. Givens